Escolar Documentos
Profissional Documentos
Cultura Documentos
Summary of All Anemia
Enviado por
benlarsena93%(14)93% acharam este documento útil (14 votos)
5K visualizações2 páginassummary_of_all anemia
Direitos autorais
© Attribution Non-Commercial (BY-NC)
Formatos disponíveis
DOC ou leia online no Scribd
Compartilhar este documento
Compartilhar ou incorporar documento
Você considera este documento útil?
Este conteúdo é inapropriado?
Denunciar este documentosummary_of_all anemia
Direitos autorais:
Attribution Non-Commercial (BY-NC)
Formatos disponíveis
Baixe no formato DOC ou leia online no Scribd
93%(14)93% acharam este documento útil (14 votos)
5K visualizações2 páginasSummary of All Anemia
Enviado por
benlarsenasummary_of_all anemia
Direitos autorais:
Attribution Non-Commercial (BY-NC)
Formatos disponíveis
Baixe no formato DOC ou leia online no Scribd
Você está na página 1de 2
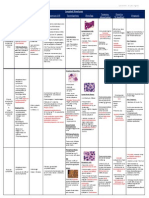
Hemolytic Anemias
Type Type of Mutation/ Cause Pathogenesis Clinical picture and findings
hemolysis
Sickle cell Extravascular Point mutation in beta-globin chain-glu On deoxygenation-polymerizaton and reversible Homozygous: severe anemia, HCT 18-30%,
anemia hemolysis replaced by val- S Hb sickling, continued exposure-ireversible – reticulocytosis, hyper-bilirubinemia, crisis
Can be homozygous- all chains have mononuclear phagocytosis & microvascular Hetero: asymptomatic until exposure to severe
mutation or heterozygous- abt half obstructions. hypoxia
chains have mutation Hypospleenism in adults, spleenomegaly in
Particularly predisposed with salmonella children- infections.
osteomyelitis
Thalasemia intravascular α- thalasemia: 4 genes on ch 16 Excessive β- & γ- chains- form stable tetramers Major:
hemolysis deletions- 1- silent carries, 4- hydrops (Hb H, Hb bart) low damage, ineffective in O2 Microcytic hypochromic + poikilocytosis
fetalis, 3- disease delivery Failure of normal development
β- thalasemia: 2 genes on ch 11 Decrease in β-chains – decreased Hb Skeletal deformities, reticulocytosis.
point mutations on certain regions of More imp: increase in free α-chains that aggregate, Iron overload – leads to cardiac failure (need
gene: in promoter & unsplicing region form inclusion in RBC (reduce plasticity of RBC, iron chelators)
of intron- β+ (reduced β-chains) becomes more susceptible to mononuclear Minor
In exon & splicing region of intron- β0 phagocytosis) destruction also of blasts – Mild microcytic hypochromic
(absence of β-chains) ineffective erythropoisis - iron overload Normal life expectancy
G6PD Intra- & extra- G6PD gene on ch X forms G6PD Exposure to drugs, toxins or infections increases H No symptoms unless exposure oxidative injury.
deficiency vascular enzyme that regenerates GSH after its peroxide so increases oxidation of GSH to GSSG, In males severe oxidant injury
hemolysis oxidation GSH regeneration is impaired so – accum. of H In females asymptomatic
Mutation causes more rapid decay of peroxide which denatures Hb. This ppts causing Heinz bodies (precipitated Hb) and bite cells
enzyme (A- variant) IVhemolysis and also EVhemolysis in spleen (phagocytosis by splenic phagocytes)
Paroxysmal intravascular Acquired membrane defect secondary PIGA deficient BM cells are present in normal PIG-tailed proteins (3 that prevent activation of
nocturnal hemolysis to a mutation that involves myeloid individuals, when there is immune-mediated complement on normal RBC’s) cannot be
hemoglobinuria stem cells. Mutation in X-linked PIGA destruction or suppression of BM cells by expressed so RBC sensitive to lytic activity of
(synthesis of intramembranous recognition of sp. PIG-tailed antigens the PIGA complement, also not expressed on granulocytes
glycoprotein anchor – PIG) deficient cells do not express the targets and so and platelets – susceptibility to infections and
escape the attack and replace BM IVthrombosis
Immuno- Warm: IgG or (rarely) IgA active at 37 Opsonization by IgG and subsequent phagocytosis +ve direct/ indirect coombs test for both
hemolytic deg. 60% idiopathic and 40 % by splenic macrophages. Attempts of phagocytosis Chronic mild anemia with moderate
anemia underlying disease (SLE) or drugs (a- leads to injured bits of CM – decreased SA:V ratio spleenomegaly
methyl dopa, penicillin) – spheroidal cells – sequestration
Extravascular
Cold: IgM binds to CM below 30 deg. Complement most active at 37 deg. so no Acute during recovery from Mycoplasma
hemolysis
(in distal parts) IVhemolysis, when cells go to warmer regions pneumonia and infectious mononucleosis or
IgM is not well bound but leaves behind C3b chronic resulting in transient mild anemia with
opsonin causing phagocytosis by kupffer cells often Raynaud phenomenon
(EV)
Resulting from Intravascular Valve prostheses and microangiopathic Valves cause abnormal pressure gradient and Burr, hemlet and triangle cells
trauma hemolysis (caused by DIC, malignant HT, SLE or turbulent blood flow, and RBCs are squeezed thru
disseminated cancer) narrowed vessels, results in mechanical damage in
both
malaria Intravascular Plasmodium vivax, malariea, ovale & After infecting hepatocytes, merozites infect Spikes of shaking, chills and fever at intervals
hemolysis falciparum (most fatal causing cerebral erythrocytes forming trophozites (characteristic to which coincide with merozite release from RBC.
malaria and blackwater fever) each specie) which divides giving rise to new Brown discoloration of spleen, liver, lymph
merozites that destroy RBCs upon escape nodes & BM, massive spleenomegaly
(hyperplasia of mononuclear phagocytes)
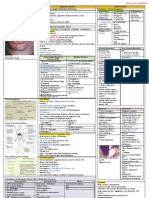
Anemias of Diminished Erythropoiesis
Type Cause Pathogenesis Clinical picture Diagnosis
Iron Deficiency Low dietary intake (rare), Starts with decline in serum ferretin and Mostly asymptomatic with weakness Low: Hb, HCT, MCV, serum
malabsorption (e.g. gastrectomy), stainable amounts of iron in BM. Followed and pallor in severe cases. Long-term ferretin, iron levels, transferring
increased demands (pregnancy, by decrease in circulating iron and rise in severs anemia – thinning, flattening saturation, microcytic hypochromic
infants), chronic blood loss (from transferrin iron-binding capacity – impact and spooning of fingernails, pica, RBCs. HIGH total iron-binding
GIT – peptic ulcer, hemorrhoids - on Hb, myoglobin and other iron increase in platelet count. Sometimes capacity
or female genital tract) compounds. When more severe – impaired develops plummer-vinson syndrome
work performance, brain function and
immunocompetence
Anemia of Occurs in chronic microbial Sequestration of iron from cells from the __ Normocytic normochromic, or
chronic disease infections (osteomyelitis, storage compartment (mononuclear hypocytic hypochromic, low serum
bacterial endocarditis, lung phagocyte storage pool) and suppression of Treatment: iron, (similar to iron deficiency)
abcess), chronic immune erythropoiesis due to inflammatory erythropoietin administration may but: Increased storage iron in
disorders (rheumatoid arthritis, mediators (IL-1, TNF, interferon-alpha) that improve but only treatment of marrow macrophages, increased
regional enteritis) and neoplasms are released from the underlying disease. underlying condition is reliable serum ferretin, decreased total iron-
(Hodgkin, lung & breast cancer) binding capacity
Megaloblastic Deficiency of folic acid from vit B12 required for regeneration of Non-specific symptoms relating to Smear of peripheral blood and bone
anemia poor diet or increased metabolic tetrahydrofolate, Folic acid provides pancytopenia (weakness, pallor, easy marrow. To differentiate between
needs. Or inhibition of folate tetrahydrofolate (carrier of a carbon group) fatigability, petechia, easy infection). vit B12 – serum folate and vit B12
metabolism by methotrexate, so both required for DNA synthesis. Alimentary tract related symptoms are levels and RBC folate levels
acidic foods & beans, phenytoin. Deficiency – delay in nuclear maturation common e.g. sore tonge (rapid dividing Low serum vit B12 levels, normal
pernicious anemia Impaired and cell division of erythroid precursors GIT cells). In vit B12 deficiency only or elevated folate, histamine-fast
absorption of vitamin B12 by: producing megaloblasts (also granulocyte neurological symptoms may take place gastric achlohydria, anti IF
*malabsorption (lack of vit B12 precursors produce giant megakaryocytes) e.g. symmetric numbness, tingling, antibodies, megaloblastic anemia
uncommon), *autoimmune some megaloblasts have very defective burning in feet or hands, unsteadiness findings, leucopenia, schilling test
reaction against parietal cells or DNA that they undergo apoptosis in BM, of gait, loss of position sense (unable to absorb an oral dose of vit
IF, others give mature RBC’s but output is *Cellular morphology: hypercellular B12 but when administered with IF
*surgery and gut disorders diminished. The enlarged RBCs are prone BM, nuclear-cytoplasmic asynchrony, absorption takes place)
(gastrectomy regional enteritis) to premature destruction by mononuclear megaloblasts – delicate finely
or *aging (gastric atrophy & phagocyte system leading to accumulation reticulated nuclear chromatin,
achlohydria) of iron (in mononuclear phagocytes) megakaryocytes – bizarre multi-lobed
nulei
Aplastic Suppression of multipotent Autoreactive T-cells (patients respond to Symptoms relating to pancytopenia Differential diagnosis to
myeloid stem cells caused by: immunosuppressive therapy aimed at T (weakness pallor, dyspnea, differentiate between it and
*idiopathic, *myelotoxic agents cells) where viral antigens, drug-drived petechiae,frequent persistant minor myelophthisic (or other
(irradiation or myelotoxic druds), haptens and genetic damage create infections or sudden onset of chills or pancytopenias) – hypocellular BM
*drugs and chemicals neoantigens within stem cells that are fever) owing to stem cell failure
(antineoplastic drugs, benzene, targets for autoreactive T cells Normocytic, normochromic, *Treatment: BM transplantation v.
chloramphenicol, or sensitivity to sometimes slight macrocytic. effective in patients nontransfused
sulfonamides, phenylbutazone Reticulocytosis is absent and younger than 40. other patients
etc.), *viral infection Spleenomegaly is absent – immunosuppressive therapy
Myelophthisic Associated with metastasis Bone marrow failure caused by extensive Anemia and thrombocytopenia Peripheral blood smear shows
arising from breast, lung, replacement of BM by tumors or other (pancytopenia) immature RBCs (teardrops) ,
prostate or thyroid 1ry lesion or lesions slightly elevated WBC count,
with myelofibrosis leukoerythroblastosis
Você também pode gostar
- AnemiasDocumento2 páginasAnemiasdoktorcoop100% (2)
- Peripheral Smear Examination PDFDocumento91 páginasPeripheral Smear Examination PDFtufis02Ainda não há avaliações
- WBC Neoplasms Review - PathologyDocumento6 páginasWBC Neoplasms Review - Pathologylas100% (6)
- Acute and Chronic LeukemiasDocumento3 páginasAcute and Chronic Leukemiaskaku100% (2)
- Approach To Diagnosis of Haemolytic AnaemiasDocumento2 páginasApproach To Diagnosis of Haemolytic AnaemiasGerardLumAinda não há avaliações
- Anti-Coagulants, Anti-Platelets, FibrinolyticsDocumento1 páginaAnti-Coagulants, Anti-Platelets, FibrinolyticsGerardLum100% (1)
- Leukocytes Benign DisordersDocumento3 páginasLeukocytes Benign DisordersGerardLum100% (3)
- Hematologic DisordersDocumento7 páginasHematologic DisordersCernan Oliveros100% (3)
- Acquired Bleeding DisordersDocumento1 páginaAcquired Bleeding DisordersGerardLumAinda não há avaliações
- Hemotology Pathology PDFDocumento15 páginasHemotology Pathology PDFMorgan PeggAinda não há avaliações
- Mnemonic SDocumento42 páginasMnemonic SWen Jie LauAinda não há avaliações
- Hematology Tables Morphology of RBCsDocumento5 páginasHematology Tables Morphology of RBCsGlydenne Glaire Poncardas GayamAinda não há avaliações
- MUST To KNOW in Immunohematology Blood BankingDocumento42 páginasMUST To KNOW in Immunohematology Blood BankingMerhella Amor Suerte MendozaAinda não há avaliações
- HAEMOPOIESISDocumento6 páginasHAEMOPOIESISDiyana ZahariAinda não há avaliações
- AnemiaDocumento1 páginaAnemiaindriyanti natasya ayu utami kottenAinda não há avaliações
- 22 - ImmunohematologyDocumento6 páginas22 - Immunohematologyhamadadodo7Ainda não há avaliações
- Hematologic Pathology p36-47Documento12 páginasHematologic Pathology p36-47zeroun24Ainda não há avaliações
- H1d - Microcytic Anemias ChartDocumento3 páginasH1d - Microcytic Anemias ChartqselmmAinda não há avaliações
- Full Blood PictureDocumento1 páginaFull Blood PictureGerardLumAinda não há avaliações
- Path Hematology ReviewDocumento19 páginasPath Hematology ReviewChristopher MuellerAinda não há avaliações
- Clinical ChemistryDocumento8 páginasClinical Chemistryleizej29Ainda não há avaliações
- Anemias ChartDocumento14 páginasAnemias ChartM Patel100% (2)
- Blood FilmDocumento2 páginasBlood FilmGerardLum100% (1)
- Role of Blood Vessels in Hemostasis: Villa, M.D. MLS 4CDocumento10 páginasRole of Blood Vessels in Hemostasis: Villa, M.D. MLS 4CMarianne Dennesse100% (1)
- Basic HaematologyDocumento32 páginasBasic HaematologyAhtshamtaeiq AhtshamtariqAinda não há avaliações
- HematologyDocumento42 páginasHematologyadaako100% (8)
- Hematology ReviewerDocumento4 páginasHematology ReviewerAbigail Puno100% (1)
- Hematology Lecture Notes PDFDocumento102 páginasHematology Lecture Notes PDFMarisa IzzaAinda não há avaliações
- Penicillins Pen&G Pen&V: T.%pallidum% (Syphilis)Documento1 páginaPenicillins Pen&G Pen&V: T.%pallidum% (Syphilis)gregoryvo100% (5)
- Clinical Medicine - Lecture: - Topic: - DateDocumento3 páginasClinical Medicine - Lecture: - Topic: - DateqselmmAinda não há avaliações
- HematologyDocumento15 páginasHematologyGilberto GonzalezAinda não há avaliações
- WBC DisordersDocumento114 páginasWBC DisordersNdor BariboloAinda não há avaliações
- AnemiaDocumento10 páginasAnemiaBia Payawal100% (2)
- Virology - Study GuideDocumento5 páginasVirology - Study GuideMatt McGlothlinAinda não há avaliações
- Correctly: IncorrectlyDocumento70 páginasCorrectly: IncorrectlyDjdjjd Siisus100% (1)
- Problems in Bone Marrow PathologyDocumento29 páginasProblems in Bone Marrow PathologymaurocznAinda não há avaliações
- RBC DisordersDocumento70 páginasRBC DisordersNdor Baribolo100% (1)
- Hematology Lecture - AnemiaDocumento10 páginasHematology Lecture - AnemiaKimberly EspaldonAinda não há avaliações
- Hematology NotesDocumento17 páginasHematology NotesEly Sibayan100% (2)
- Clin Path Lab 6 UrinalysisDocumento5 páginasClin Path Lab 6 Urinalysisapi-3743217100% (6)
- Myeloproliferative Disorders PDFDocumento52 páginasMyeloproliferative Disorders PDFBoneyJalgar100% (3)
- Bone Marrow FailureDocumento2 páginasBone Marrow FailureGerardLum100% (1)
- Hematology & OncologyDocumento94 páginasHematology & OncologyDaNy Chiriac100% (1)
- All Objectives HematologyDocumento45 páginasAll Objectives HematologyNursing200980% (5)
- Acute Lymphoblastic Leukemia QuestionsDocumento22 páginasAcute Lymphoblastic Leukemia Questionsđoàn lươngAinda não há avaliações
- HematologyDocumento5 páginasHematologyIvy Jan OcateAinda não há avaliações
- Micro Buzz Words - KEY WordsDocumento8 páginasMicro Buzz Words - KEY WordsKris GulleyAinda não há avaliações
- AntibioticsDocumento6 páginasAntibioticsyezan27100% (8)
- Hematology Slides Step 1 UsmleDocumento1 páginaHematology Slides Step 1 Usmlenreena aslamAinda não há avaliações
- Hematologic Pathology p24-35Documento12 páginasHematologic Pathology p24-35zeroun24100% (6)
- Fast Facts: Myelodysplastic Syndromes: Determining risk, tailoring therapy, supporting patientsNo EverandFast Facts: Myelodysplastic Syndromes: Determining risk, tailoring therapy, supporting patientsAinda não há avaliações
- Types Of Hemolytic Anemia, A Simple Guide To The Condition, Treatment And Related ConditionsNo EverandTypes Of Hemolytic Anemia, A Simple Guide To The Condition, Treatment And Related ConditionsAinda não há avaliações
- Fast Facts: Measurable Residual Disease: A clearer picture for treatment decisionsNo EverandFast Facts: Measurable Residual Disease: A clearer picture for treatment decisionsAinda não há avaliações
- Objective & Scope of ProjectDocumento8 páginasObjective & Scope of ProjectPraveen SehgalAinda não há avaliações
- Quick Help For EDI SEZ IntegrationDocumento2 páginasQuick Help For EDI SEZ IntegrationsrinivasAinda não há avaliações
- IEC ShipsDocumento6 páginasIEC ShipsdimitaringAinda não há avaliações
- Thermally Curable Polystyrene Via Click ChemistryDocumento4 páginasThermally Curable Polystyrene Via Click ChemistryDanesh AzAinda não há avaliações
- De Thi Hoc Ki 1 Mon Tieng Anh Lop 5 Co File NgheDocumento10 páginasDe Thi Hoc Ki 1 Mon Tieng Anh Lop 5 Co File Nghetuyen truongAinda não há avaliações
- Benevisión N15 Mindray Service ManualDocumento123 páginasBenevisión N15 Mindray Service ManualSulay Avila LlanosAinda não há avaliações
- DarcDocumento9 páginasDarcJunior BermudezAinda não há avaliações
- Lesson PlanDocumento2 páginasLesson Plannicole rigonAinda não há avaliações
- Making Effective Powerpoint Presentations: October 2014Documento18 páginasMaking Effective Powerpoint Presentations: October 2014Mariam TchkoidzeAinda não há avaliações
- PMP Question BankDocumento3 páginasPMP Question BankOmerZiaAinda não há avaliações
- Residual Power Series Method For Obstacle Boundary Value ProblemsDocumento5 páginasResidual Power Series Method For Obstacle Boundary Value ProblemsSayiqa JabeenAinda não há avaliações
- Computing of Test Statistic On Population MeanDocumento36 páginasComputing of Test Statistic On Population MeanKristoffer RañolaAinda não há avaliações
- How Drugs Work - Basic Pharmacology For Healthcare ProfessionalsDocumento19 páginasHow Drugs Work - Basic Pharmacology For Healthcare ProfessionalsSebastián Pérez GuerraAinda não há avaliações
- Ito Na Talaga Yung FinalDocumento22 páginasIto Na Talaga Yung FinalJonas Gian Miguel MadarangAinda não há avaliações
- B.SC BOTANY Semester 5-6 Syllabus June 2013Documento33 páginasB.SC BOTANY Semester 5-6 Syllabus June 2013Barnali DuttaAinda não há avaliações
- Unit 16 - Monitoring, Review and Audit by Allan WatsonDocumento29 páginasUnit 16 - Monitoring, Review and Audit by Allan WatsonLuqman OsmanAinda não há avaliações
- 8.ZXSDR B8200 (L200) Principle and Hardware Structure Training Manual-45Documento45 páginas8.ZXSDR B8200 (L200) Principle and Hardware Structure Training Manual-45mehdi_mehdiAinda não há avaliações
- RSA - Brand - Guidelines - 2019 2Documento79 páginasRSA - Brand - Guidelines - 2019 2Gigi's DelightAinda não há avaliações
- Pt3 English Module 2018Documento63 páginasPt3 English Module 2018Annie Abdul Rahman50% (4)
- Biological Beneficiation of Kaolin: A Review On Iron RemovalDocumento8 páginasBiological Beneficiation of Kaolin: A Review On Iron RemovalValentin GnoumouAinda não há avaliações
- Heterogeneity in Macroeconomics: Macroeconomic Theory II (ECO-504) - Spring 2018Documento5 páginasHeterogeneity in Macroeconomics: Macroeconomic Theory II (ECO-504) - Spring 2018Gabriel RoblesAinda não há avaliações
- Waterstop TechnologyDocumento69 páginasWaterstop TechnologygertjaniAinda não há avaliações
- Working With Difficult People Online WorksheetDocumento4 páginasWorking With Difficult People Online WorksheetHugh Fox IIIAinda não há avaliações
- LEIA Home Lifts Guide FNLDocumento5 páginasLEIA Home Lifts Guide FNLTejinder SinghAinda não há avaliações
- Anatomy Anal CanalDocumento14 páginasAnatomy Anal CanalBela Ronaldoe100% (1)
- E MudhraDownload HardDocumento17 páginasE MudhraDownload HardVivek RajanAinda não há avaliações
- Ozone Therapy - A Clinical Review A. M. Elvis and J. S. EktaDocumento5 páginasOzone Therapy - A Clinical Review A. M. Elvis and J. S. Ektatahuti696Ainda não há avaliações
- Iec TR 61010-3-020-1999Documento76 páginasIec TR 61010-3-020-1999Vasko MandilAinda não há avaliações
- The Mantel Colonized Nation Somalia 10 PDFDocumento5 páginasThe Mantel Colonized Nation Somalia 10 PDFAhmad AbrahamAinda não há avaliações
- Dtu Placement BrouchureDocumento25 páginasDtu Placement BrouchureAbhishek KumarAinda não há avaliações