Escolar Documentos
Profissional Documentos
Cultura Documentos
Dehydration of Methanol To Dimethyl Ether, Ethylene and Propylene Over Silica-Doped Sulfated Zirconia
Enviado por
krishna_nimeshTítulo original
Direitos autorais
Formatos disponíveis
Compartilhar este documento
Compartilhar ou incorporar documento
Você considera este documento útil?
Este conteúdo é inapropriado?
Denunciar este documentoDireitos autorais:
Formatos disponíveis
Dehydration of Methanol To Dimethyl Ether, Ethylene and Propylene Over Silica-Doped Sulfated Zirconia
Enviado por
krishna_nimeshDireitos autorais:
Formatos disponíveis
1844
Bull. Korean Chem. Soc. 2006, Vol. 27, No. 11
Syed T. Hussain et al.
Dehydration of Methanol to Dimethyl ether, Ethylene and Propylene over Silica-Doped Sulfated Zirconia
Syed T. Hussain,* M. Mazhar, Sheraz Gul, Karl T Chuang, and Alan R. Sanger
Department of Chemistry, Quaid-e-Azam University, Islamabad 45320, Pakistan. *E-mail: dr_tajammul@yahoo.ca Department of Chemical Engineering, University of Alberta, Alberta, Canada T6G 2G6 Received August 28, 2006
Two types of catalyst samples were prepared, one sulfated zirconia and the other silica doped sulfated zirconia. The acidity tests indicate that sulfated zirconia doped with silica has higher concentration and strength of acidic catalyst sites than undoped sulfated zirconia. The acidic surface sites have been characterized using FTIR, NMR, pyridine adsorption, TPD, XRD and nitrogen adsorption. Doping with silica increased the concentration of surface Lewis and Brnsted acid sites and resulted in generation of proximate acid sites. The activity test indicates that doping sulfated zirconia with silica increases both the acidity and catalytic activity for liquid phase dehydration of methanol at 413-453 K. Methanol is sequentially dehydrated to dimethyl ether and ethylene over both catalysts. Significant amounts of propylene are also formed over the silica-doped catalyst, but not over the undoped catalyst. Key Words : Sulfated zirconia, Dehydration of methanol, Dimethyl ether, Ethylene, Propylene
Introduction
Solid acid catalysts play an important role in hydrocarbon conversion reactions in the petroleum and chemical industries. In particular, the solid super-acid properties and activity of sulfated zirconia were first described over a decade ago, which led to intense research in this area. Rational design of solid acid catalysts for a specific application requires control of acid site type (Lewis, Brnsted), density, and strength. To this end, there are two general strategies for modification of the acidic properties of catalytic oxides. One approach is to dope the oxide with anions. Anionic dopants can increase the electron-withdrawing capability of the doped surface, thereby enhancing the ability of neighboring hydroxyl groups to act as proton donors. A second approach is to dope an oxide catalyst with a second oxide. Modeling of the performance and acidity of mixed oxides show that strongly acidic sites form when large localized charge imbalances develop at M-O-M' linkages (M and M' are different cations). The high catalytic activity of sulfated zirconia has been attributed to superacidity. The goal of the present research is formulation of highly active acidic catalytic materials. To this end we are preparing, characterizing and testing modified zirconium sulfate based catalysts having Lewis and Brnsted acid sites with different concentrations and strengths. We have now found that doping zirconium sulfate with silica increases both the strength and density of surface acidic sites, and their catalytic activity. Herein we will compare the catalytic activity of silica-doped sulfated zirconia (SZS) with that of undoped sulfated zirconia (SZ) using, as a probe reaction, methanol dehydration to sequentially form dimethyl ether and ethylene. We will show that significant amounts of propylene are also formed when the catalyst is SZS, but not when it is SZ. A
1 2 3,4 5-7
detailed comparison of the physical properties of SZS and SZ and the respective surface acidic sites using spectroscopic and microscopic techniques and chemical probes has also been described.
Experimental Details Catalyst Preparation. Zirconium oxynitrate [ZrO(NO ) ] was dissolved in 10 N nitric acid solution. Aqueous ammonia was then slowly added to the stirred solution to precipitate zirconium hydroxide, until the pH was ca. 11. The precipitate was recovered by filtration, washed with water and calcined at 573 K for 4 hours under atmospheric conditions to form zirconia. Sulfated zirconia (SZ) was prepared by suspending 5 g zirconia in 100 mL of an aqueous solution containing 50 wt% sulfuric acid. The solution was then heated to evaporate water using a hotplate. The solid formed by drying the mixture over the hot plate for ca. 2 h was calcined at 923 K for 4 hours under atmospheric conditions. Silica-doped sulfated zirconia (SZS) was prepared using substantially the same procedure as described above, except that a weighed amount of silica was added to the stirred reaction mixture before heating. Catalyst Characterization. BET surface areas of fresh and used catalysts were determined by nitrogen adsorption at 77 K (Table 1). Crystallite sizes of fresh and used samples were determined using the Scherer equation and presented in Table 2. Pyridine adsorption was used to determine the acidic sites using FTIR, TPD and NMR and then their consequent effect on the catalytic activity was discussed. The amount of sulfate leached from SZ or SZS during catalytic tests was determined by acid-base titration of the residual liquid reaction mixture. Catalytic Activity Testing. Reactions were performed in
3 2
Dehydration of Methanol to Dimethyl ether, Ethylene, and Propylene
Table 1
Bull. Korean Chem. Soc. 2006, Vol. 27, No. 11
1845
. BET area and pore volume for fresh and used catalysts Catalyst Surface area Surface area Pore Volume Pore Volume sample m2/g (Fresh) m2/g (Used) mL/g (Fresh) mL/g (Used) SZ 191.58 146.83 0.1082 0.1744 SZS 223.14 172.98 0.09231 0.1443 . Crystallite size (XRD data) Catalyst Crystallite size (nm) sample (Fresh) SZ 14.60 SZS 13.60
Crystallite size (nm) (Used) 31.5 30.0
Table 2
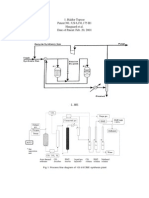
a batch slurry reactor system (Figure 1). The reactor was a high pressure SS-316 reactor with a volume of 320 mL (model 4841, Parr Instruments Inc. USA), equipped with an impeller. A J-type thermocouple was immersed into the liquid phase to measure the reactor temperature, and was connected to a heater/controller to maintain the reactor temperature within 1 K. Liquid samples were collected using a 1/8 inch o.d. stainless steel tube connected to a stainless steel sampling valve. A pressure transducer (Foxboro electronic transmitter, Model 84 GM-D) was used to measure pressure in the reactor with an accuracy of 4.0 kPa. Analyses of Reaction Mixtures. Liquid and gas samples were analyzed using a Hewlett Packard 5890 Series II Gas Chromatograph (GC) equipped with a TCD detector and a 0.914 m long column with Poropak R packing (50-80 mesh), operated at a constant column temperature of 423 K. The GC was calibrated for ethylene, propylene, dimethyl ether (DME), water and methanol using standard mixtures having different concentrations of ethylene, propylene and dimethyl ether (DME). The exit gasses were analyzed for DME, ethylene and propylene. Liquid samples (1 mL) were taken hourly and analyzed using GC. Analyses of the vapor phase were performed online. Compositions of gas and liquid phase product mixtures were determined by comparison of GC data with
Figure 1
. Schematic diagram of the equipment.
standard mixtures. Procedure. First, 150 mL of methanol (analytical grade, BDH) and 2.0 g of catalyst were loaded into the reactor, so that initially about one half of the vessel was filled with liquid. The liquid expanded at high temperatures; hence space was allocated to accommodate this expansion. The reactor was then purged with helium to remove any air initially present in the reactor, and to provide an inert atmosphere. The inlet and outlet of the reactor were then closed. The amount of helium initially present in the reactor prior to heating was determined from the initial reactor temperature, pressure and vapor volume after sealing the reactor, and was used as an internal standard to calculate material balance in kinetic studies. The reactor contents were then heated to a reaction temperature pre-selected from 373, 413, 433 and 453 K. The range 373-453 K was selected for two reasons. First, the reaction rate was sufficiently fast to allow experiments to be completed within hours. Second, pressures developed within this range could be reproducibly determined with good accuracy using the present experimental equipment. The pressure in the closed reactor rose from 260 psig at 413 K to 375 psig at 433 K and 575 psig at 453 K. The temperature range 413-453 K was then selected for more detailed investigation because a significant amount of alkene reaction product was produced in this range. Negligible amounts of reaction products were produced during the time required to heat the reaction mixture to the pre-selected temperature as monitored by on line GC during the ramp. The reaction mixture was stirred at 1080 rpm for the duration of the reaction (1-3 hours) to minimize the influence of external mass transfer. Selectivity and conversion were calculated from GC analytical results. After the reaction, the residual product liquid was filtered and the catalyst was recovered, dried and could be used again for further reaction. Catalyst Characterization Using XRD and BET. The increase in surface area of fresh SZS compared to fresh SZ was attributed to modification of the catalyst structure as a consequence of doping with silica. SZS had a higher surface area than SZ, but had a slightly smaller pore volume. Doping SZ with silica reduced the average pore size. Figure 2 shows the crystalline XRD for fresh and used samples of SZS and SZ, respectively. The XRD data showed that the physical structure of SZS differed from that of SZ. Comparison of the XRD of fresh and used samples of each catalyst showed that the structure of SZS was more stable than that of SZ. The XRD of SZS showed much more intense peaks for 2 at 30o and 34o in comparison with SZ. This suggested that SiO2 doping not only resulted in a more stable crystal structure, but also in modification of catalyst physical characteristics (Figure 2b). The differences between the XRD data for the two fresh samples were consistent with a combination of a change in crystal structure resulting from doping SZ with SiO2, and a minor change in size of crystallites. There were similar differences between used samples
1846
Bull. Korean Chem. Soc. 2006, Vol. 27, No. 11
Syed T. Hussain et al.
. XRD spectra of fresh and used samples of (a) silica doped and (b) undoped sulfated zirconia.
Figure 2
. FTIR spectra of undoped and silica doped catalysts before and after adsorption of pyridine.
Figure 3
of SZ and SZS. However, the surface areas and crystallite sizes of used SZ and SZS were both significantly larger than those for fresh catalysts. The combination of an increase in crystallite size, coupled with an increase in surface area during the methanol dehydration reaction, suggested that the crystallites agglomerated and became more porous during the reaction. Although the cause for this combination of effects cannot be determined unambiguously from the present data, it may in part have been a consequence of leaching of a portion of sulfate during that reaction. From data and Figure 2 it can be seen that the catalyst phase, surface area, pore volume and crystallite size changed appreciably during use as a catalyst for methanol dehydration. However, the changes did not appear to affect catalytic activity or selectivity when the catalyst was used in one or more catalyst runs up to 3 h in duration. The data suggest that the changes in the catalyst structure did not significantly affect catalyst sites, and that the catalysts had acceptable physical stability. The amount of sulfate ions leached over the period of the methanol dehydration experiments was not sufficient to induce any major change in the catalyst sites on SZ and SZS catalysts over a small number of runs. However, prolonged leaching resulted in catalyst deactivation. It is noteworthy that the crystal stability of SZS was greater than that for SZ (XRD, Figure 2), and the amount of sulfate leached from SZS was less than that from SZ. The combination of the above results indicated that both the stability of active sites and the crystal structures of SZ
and SZS depended on retention of a significant portion of the original sulfate content. Pyridine Adsorption. Typical IR spectra obtained before and after pyridine adsorption on SZ and SZS samples at 100 o C are presented in Figure 3a (before pyridine adsorption) and 3b (after pyridine adsorption). Two diagnostic peaks characteristic of pyridine interacting with Brnsted acid sites to form pyridinium ions (PyH+) were observed at 1540 cm1 and 1445 cm1. Additional peaks assigned to PyH+ were located at 1630 cm1, 1600 cm1 and 1489 cm1. Peaks at 1637 cm1 and 1488 cm1 have been attributed to pyridine molecules adsorbed on Lewis acid sites. Other peak observed in the spectra corresponded to physisorbed pyridine and pyridine that was hydrogen-bonded to silanol groups. The spectrum for pyridine adsorbed on SZS (Figure 3b) showed an intense pyridinium (PyH+) peak at 1445 cm1. Thus, SZS had a higher concentration of Brnsted acid sites than SZ. However, only small shoulders were observed at 1488 and 1637 cm1, that corresponded to a trace amount of pyridine adsorbed at Lewis acid sites. When the sample was exposed to further pyridine, only the peaks at 1445 and 1637 cm1 increased in intensity, indicating that the Brnsted and Lewis acid sites were already saturated. The intensities of the peaks at 1445 cm1 and at 1637 cm1 were used to estimate the concentration of the respective sites. The breadth of the FWHM [full width half maximum] of peaks at 1445, 1530 and 1630 cm1 indicated the presence of additional multiple acid sites that were distinct from both of
Dehydration of Methanol to Dimethyl ether, Ethylene, and Propylene
Bull. Korean Chem. Soc. 2006, Vol. 27, No. 11
1847
. Temperature programmed desorption (TPD) of pyridine pre-adsorped on undoped and silica-doped catalysts (numbers are temperature (K) corresponding to maxima).
Figure 4
Figure 5
. FTIR analysis of spent SZS sample.
isolated Brnsted and Lewis acid sites. Both the IR peak at 1375 cm1 and a 1H NMR signal at 5.75 ppm, assigned to super-acid protons, lost intensity after pyridine adsorption. Thus, Brnsted, Lewis and combinations of proximate acid sites were all available as potentially active acidic catalyst sites. The concentration of Brnsted acid sites determined for the SZS sample, in accordance with the method of reference,8 was found to be 1.1 1020 ions.g1. Both SZ and SZS samples displayed a prominent feature (Figure 3a) at 1375 cm1, arising from increased S=O double bond character for sulfate ions at high temperature. Figure 4 shows the TPD of pyridine on SZ and SZS. It was found that the strength of adsorption of pyridine on SZS was higher than on SZ. SZ did not adsorb pyridine at Brnsted acid sites at 423 K. The acidity of Brnsted acid sites of SZS was stronger after activation at 923 K. The TPD of pyridine on SZS displayed two features at 650 and 720 K that were not present in TPD of pyridine on SZ. Detection of these additional features indicated that combinations of proximate acid sites were present only on SZS. In addition, SZS was found to have a higher total acid sties density, 2.4 mmolm2, compared with 1.8 mmolm2 on SZ. Thus, the overall concentration of surface acidic sites, their strengths, and the distribution of types of site were all enhanced by doping SZ with silica to form SZS. Characterization of Acid Sites Using NMR. 1H NMR data of SZ and SZS before and after pyridine adsorption are presented in Table 5. Three distinct NMR resonances were detected, one at 5.75 ppm and two other signals at 2.1 and 1.2 ppm. The signal at 5.75 ppm was in the range attributable to super-acid protons. The intensity of the signal at 5.75 ppm was much more in the spectrum of SZS when compared with the spectrum of SZ, indicating that the concentration of strongly acid sites was higher on SZS. Pyridine adsorption on either SZ or SZS reduced the intensity of the signal at 5.75 ppm. The reduction in intensity was greater for SZ than for SZS (Table 5). The spectrum of
SZS treated with pyridine showed a new signal at 1.9 ppm that was assigned to pyridine adsorption at a combination of proximate acid sites. Together with the FTIR and TPD data above, the NMR data supported the proposal that SZS catalyst had complex proximate acid sites in addition to Brnsted and Lewis acid sites, and that these complex sites on SZS were more acidic than the acid sites on SZ.
Results and Discussions
Methanol dehydration was used as a model reaction for comparing the catalytic properties of SZ and SZS for reactions in the temperature range 373-453 K. The results are presented in Table 3. Methanol was dehydrated to DME at all temperatures 373-453 K over either SZ or SZS. Ethylene and propylene were produced only in reactions at 413-453 K. Formation of propylene occurred only over SZS catalyst. No other hydrocarbon products were detected. Dehydration of Methanol to DME. Direct dehydration of methanol produced DME as the sole primary product at 373-453 K (Eq. 1). When alkenes were formed as secondary products, the concentration of DME did not increase linearly with time, as it was consumed to form ethylene or propylene. 2CH3OH (CH3)2O + H2O (1) Table 3 shows the compositions of the reaction mixture after reaction for 1-3 h at different temperatures. Conversion to dehydration products over SZS catalyst was higher at all temperatures than reaction over SZ. This effect showed that the acidity of SZS was higher than that of SZ. Dehydration of DME to Ethylene. Conversion of DME to ethylene was a secondary reaction (Eq. 2; * represents a surface site). The characteristics of catalytic sites for conversion of alcohols to olefins have been studied intensely. The mechanism for elimination of water from a C2+ alcohol, in which the C-C bond already exists, differs from dehydration of methanol to ethylene, or to propylene. Nevertheless, similar intermediates may be formed at the catalyst surface, and so studies of conversion of aliphatic primary C2+ alcohols to olefins on catalysts possessing acidic centers provide useful insights.9-11 It is well known that Brnsted
Temperature Programmed Desorption of Pyridine.
1848
Bull. Korean Chem. Soc. 2006, Vol. 27, No. 11
18 Berg, CH3OH *-OCH3
et al.
Syed T. Hussain et al.
. Methanol conversion and product distribution over SZ and SZS catalysts as a function of temperature Product selectivity (%) Reaction Methanol temp. (K) conversion (%) Dimethyl ether Ethene Propene Over SZ catalyst 413 32.5 15.6 84.4 0 433 41.2 6.4 93.6 0 453 28.0 7.5 92.5 0 Over SZS catalyst 413 42.7 12.6 75.6 11.7 433 52.1 3.3 86.4 10.4 453 43.2 0.4 64.6 35.0
Table 3
acid sites catalyze dehydration of alcohols to olefins. However, Brnsted acid catalysis is not necessarily the sole cause of olefin formation. It has been shown that alcohols also can dissociatively adsorb on various metal oxides to form surface alkoxide intermediates (*-OCnHm)12,13; and these can then undergo further reaction to eliminate alkene. The details of the alkoxide pathway have been shown to depend on factors such as the structure14 and the electronic nature15 of the reacting alcohol. Lahousse, .16 showed that catalytic redox ability is required for activity for dehydration of 2-propanol to propylene. (CH3)2O *-OC2H5 C2H4 (2) Formation of Propylene. Propylene was also formed as a secondary product over SZS, but not SZ (Table 3). Coupling of both carbon atoms from a single molecule of DME to form ethylene conceivably could have occurred by reaction at either one surface site or a small number of proximate sites. However, formation of propylene required participation of a third carbon atom, probably as a *-CHx (* represents a surface site) species, either from methanol or from a second molecule of DME (Eq. 3). Our argument is supported by the IR data (Figure 5) of spent SZS catalyst which shows a peak at 3013 cm1 corresponding to CH bonding and a peak at 1593 cm1 representing C=C bonding. 3CH3OH C3H6 + 3H2O (3) There has been debate as to whether ethylene is the only hydrocarbon product containing a C-C bond formed directly from DME or whether propylene should also be considered as directly formed from DME.17,18 Two distinct mechanisms have been proposed. In the first mechanism, ethylene is initially formed -elimination from ethoxy species, and is subsequently methylated by reaction with a methoxy group (Eq. 4). Chain propagation proceeds a methylation process. In the second mechanism, it has been suggested that methoxy groups interact with methanol and/or DME to form an iso-propoxy group, by a process analogous to that described for the formation of ethoxy species. Propylene then is formed by -elimination from the isopropoxy species (Eq. 5). We suggest that the second propagation pathway is an essential feature of the mechanistic proposal of Van der
et al via via
*-OC3H7 C3H6 (4) *-OC2H5 C2H4 (CH3)2O + *-OCH3 *-OC3H7 C3H6 (5) The present data provide further insight into the process, and suggest that intermediate surface species *-CHx were stabilized at highly acidic sites. The intermediate species then react with either C2H4, in a mechanism similar to Eq. (4), or with an intermediate such as an alkoxy species preceding formation of C2H4 (Eq. 6 a & b) to form the C3 backbone of propylene. CH3OH *-CHx (6a) *-OC2H5 + *-CHx *-OC3H7 C3H6 (6b) We proposed a mechanism in which a combination of pathway comprising parallel and consecutive reactions, involving nucleophilic and electrophilic species, is required for formation of DME, ethylene and propylene from methanol dehydration on acidic catalysts. In the present work, formation of propylene only occurred when surface sites had sufficiently high acidity to form *-CHx intermediate moieties, and these intermediates were themselves sufficiently reactive and proximate to react with *-OC2H5 intermediates and form the telomerized intermediate *-OC3H7. Thus, formation of propylene required that there was a high concentration of surface sites, and that several of these sites were highly acidic. SZS has a higher density and stronger multiple acidic sties than SZ. Thus, formation of propylene on SZS, and not on SZ, was consistent with the requirement that the more strongly acidic sites on SZS were available in high concentration for formation of C3 surface intermediate species. It is noteworthy that no hydrocarbon products higher than propylene were formed, even though the selectivity to propylene increased to 35% at 453 K over SZS. Three inferences can be drawn from these results. First, there was no C4 product, and so dimerization of C2 intermediates or products does not occur to a detectable amount. The second inference is that chain growth only formation and assembly of C1 intermediates probably was not a significant component of the mechanism. If the mechanism involved a significant amount of assembly of C1 units to form the carbon backbone chain, it would be expected that the distribution of products would more closely resemble an Anderson-Schultz-Flory distribution. In that case, one would anticipate that a detectable amount of C4 product also should have been formed when the selectivity to propylene was as high as 35%, but none was found. Thus, it is most probable that the mechanism involved reaction of a Cn species, most likely an alkoxy intermediate, with a *-CHx intermediate moiety to form a Cn+1 chain, rather than assembly of the chain from only C1 units.
via
Dehydration of Methanol to Dimethyl ether, Ethylene, and Propylene
Bull. Korean Chem. Soc. 2006, Vol. 27, No. 11
1849
The third inference is that if, indeed, the mechanism required chain growth through reaction of a Cn chain with *-CHx to form a Cn+1 chain, then formation of a surface bonded C4 intermediate was unfavoured. A limitation of this nature would be consistent with a mechanism in which the chain growth process occurred by nucleophilic attack at a secondary carbon in the present chain (CnH2n or *-OCnH2n+1) to form a branched product, and that there was steric constraint to formation of *-OC4H9 as a branched chain intermediate (Eq. 7). *OC3H7 (or C3H6) + *-CHx *-OC4H9 (7) Apparent Rate Constants. Methanol conversion reaction data for all SZ and SZS catalyst tests were found to fit the Bassett-Habgood rate equation (Eq. 8) for first order reactions in which the reactant partial pressure is in the range 260-575 psig. The adsorption rate is faster than the rate of reaction, the latter being the rate-determining step. ln[1/(1X)] = kaRT(W/F) (8) Where X is total conversion, ka is the apparent rate constant, W is the weight of catalyst, and F is the weight of methanol in the reactor. Plots of ln[1/(1-X)] Vs F1 are linear for either SZ or SZS catalysts, and pass through the origin, as expected from Eq. (8). Therefore, the reaction is first order in methanol. Apparent rate constants, ka, obtained from the slopes of the linear plots of ln[1/(1-X)] Vs F1 and activation parameters calculated using the rate law equation (Eq. 8) are shown in Table 4. Gas phase dehydration of alcohols to the corresponding olefins proceeds on acid catalysts via the reaction scheme presented in reference.19 The mechanism proposed above proceeds via alkene elimination from surface alkoxy species. The data obtained in this study are consistent with a mechanism of product formation that is substantially similar to that proposed for reaction in the gas phase. On the basis of discussions presented above we proposed that Lewis and Brnsted acid sites are in equilibrium on SZ and SZS, as shown in Eq. (9). Dehydration of an alcohol to an alkene includes elimina. Apparent rate constants (ka) and activation parameters (Ea and ln A) for methanol conversion on SZ and SZS catalysts, as derived from the Bassett-Habgood rate equation [20] rate Ea (Activation Temperature Ka (Apparent constant) energy) ln A (K) (106 mol/atm.g.s.) (kcal/mol)
Table 4
(9)
SZ catalyst 413 433 453 413 433 453
14.1 16.4 12.0 SZS catalyst 20.3 35.4 18.7
106 3 123 4 100 4 120 3 134 4 110 4
0.2 1.8 0.8 2.0
0.6
1.4
tion of the -hydrogen atom from an adsorbed substrate. The nominally weak acidic OH groups in cation exchanged catalyst are sufficiently active to catalyze alcohol dehydration. Both SZ and SZS catalysts are more active for alcohol dehydration than other acid catalysts screened during a parallel study such as Zeolite 3A, Zeolite 5A dispersed on silica or alumina; sulfuric acid; Ce and Al phosphate catalysts dispersed on silica or alumina; Amberlyst 16 and Amberlyst 36 dispersed on silica or alumina, and aluminasilica catalysts having different alumina-silica ratios ranging between 20-80%. Further reaction to form propylene occurred only over SZS. Thus, higher acidity and increased concentration of acidic sites on SZS compared to SZ enhanced activity for additional C-C bond formation reactions. Correlation of Activity with Surface Properties. Doping of SZ with SiO2 produced a SZS catalyst that was slightly more active for dehydrating methanol to dimethyl ether and ethylene than undoped SZ, and also produced propylene. A slight increase in selectivity to ethylene was also observed with SZS. The TPD of pyridine adsorbed on SZS displayed features that characterized the presence of combinations of proximate acid sites in SZS, which were not present in TPD of pyridine on SZ. In addition, SZS was found to have a higher total acid site density, 2.4 mmolm2, compared with 1.8 mmolm2 on SZ. Our study clearly indicates that concentrations of Brnsted and Lewis acid sites determine catalytic activity and selectivity. Thus, the increase in catalytic activity of SZS compared to SZ, and the differences between the products obtained for the present reaction, were entirely consistent with the presence of a high concentration of proximate and strongly acidic sites only on SZS. Sulfate Ion Leaching and Catalyst Stability. From Tables 1 and 2, it can be seen that the area, pore volume and crystallite size changed appreciably with use of either SZ or SZS as catalyst. However, the changes did not appear to affect catalytic activity or selectivity during a catalyst run. This indicated that the catalysts had acceptable physical stability over the duration of runs under the present experimental conditions. Small amounts of sulfate ions and other oxyanion species within the catalyst phase were leached during the catalytic runs. The amount of sulfate leached from SZ (0.075 g (SO42)/g(cat) and from SZS (0.025 g (SO42)/g(cat) was determined by acid-base titration of the residual liquid reaction mixture. Leaching caused a significant change in crystallite size and, to a lesser degree, change in surface area.
1850
Bull. Korean Chem. Soc. 2006, Vol. 27, No. 11
1
H NMR of undoped and silica-doped sulfated zirconia, before and after pyridine (Py) adsorption
Syed T. Hussain et al.
SZS
Table 5.
Before Py ads. ppm Intensity
SZ
5.75 3.35* 2.51* 2.1 1.2
62.9 1050 260 23.1 9.6
After Py ads. ppm Intensity + 79.3+ 8.58 + 7.81 44.6+ + 7.40 137.5+ 5.75 13.4 3.35* 1049 2.51* 250 2.1 30.0 1.2 9.6
Before Py ads. ppm Intensity
5.75 3.35* 2.51* 2.1 1.2
151.0 1046 258 6.8 2.4
After Py ads. ppm Intensity + 8.58 66.2+ + 7.81 21.9+ + 7.40 100.8+ 5.75 48.4 3.35* 1047 2.51* 261 2.1 93.3 1.9 30.4 1.2 2.9
Remarks
+
Pyridine peaks
Solvent (DMSO) peaks
These data suggested that particles became aggregated during use. The amount of sulfate ions leached over the period of the experiments reported was not sufficient to induce any major change in the concentration or nature of catalyst sites on SZ and SZS catalysts during each run. The lesser amount of sulfate that was leached from SZS than from a same quantity of SZ showed that silica dopant increased the resistance of the catalyst to leaching.
Conclusion
Propylene was formed over sulfated zirconia doped with silica, but not over undoped sulfated zirconia. Formation of propylene is attributed to formation of intermediate *-CHx species at strongly acidic sites, which react with either ethylene, or with an alkoxy intermediate leading to formation of ethylene, in a chain propagation step. The catalytic activity and product selectivity show that doping sulfated zirconia with silica resulted in an increase in both acidity and concentration of surface acidic sites, and formation of proximate acid sites. No hydrocarbon products higher than propylene were formed. This suggested that the mechanism proceeded via nucleophilic attack on the carbon chain of the intermediate, and that there was steric constraint to formation of branched C4 or higher intermediates that would have been formed as a result of such a reaction. The kinetics of ethylene formation are consistent with a elimination mechanism from a surface intermediate alkoxy species derived from methanol. Sulfated zirconia doped with silica was more stable than undoped catalyst, and less prone to leaching of sulfate. FTIR, NMR and pyridine adsorption showed that doping sulfated zirconia with silica resulted in an increase in both acidity and concentration of Brnsted acid sites, and formation of proximate acid sites. Correlation of spectroscopic data with differences between reaction data for dehydration of methanol to dimethyl ether, ethylene and propylene over the doped and undoped
catalysts showed that the highly acidic sites were related to the higher catalytic activity of the doped catalyst. Formation of propylene over sulfated zirconia doped with silica, but not over undoped sulfated zirconia, was attributed to formation of intermediate CHx species at strongly acidic sites. Differences between the combination and relative strengths of Lewis, Brnsted and complex proximate acid sites on the surfaces of undoped sulfated zirconia and sulfated zirconia doped with silica determined their relative catalytic activity and selectivity.
References
1. Baiker, A.; Kijenski, J. Catal. Today , 5, 1. 2. Hino, M.; Kobayashi, M.; Arata, K. J. Am. Chem. Soc. , 108, 6439. 3. Kung, H. H. Solid State Chem. , 52, 191. 4. Tanabe, K.; Sumiyoshi, T.; Shibata, K.; Kiyoura, T. Bull. Chem. Soc. Jpn. , 47, 1064. 5. Tanabe, K.; Itoh, M.; Morishige, K.; Hattori, H. in Preparation of Catalysts; Delmon, B.; Jacobs, P. A.; Poncelet, G., Eds.; Elsevier: Amsterdam, 1976; p 65. 6. Arata, K. Adv. Catal. , 37, 165. 7. Corma, A. Chem. Rev. , 95, 559. 8. Willams, C.; Makarova, M. A.; Malysheva, L. V.; Paukshits, E. A.; Thomas, J. M.; Zamaraev, K. I. J. Chem. Soc. Farady Trans. , 86, 3473. 9. Kanazirev, V.; Tsoncheva, T. Minchev, Chr, Z. Phys. Chem. N.F. , 149, 237. 10. Tanabe, K.; Misono, M.; Ono, Y.; Hattori, H. In New Solid Acids and Bases, Their Catalytic Properties; Delmon, B.; Yates, J. T., Eds.; Elsevier: Amsterdam, 1989; Vol. 51. 11. Gervasini, A.; Auroux, A. J. Catal. , 131, 190. 12. Vohs, J. M.; Barteau, M. A. Surf. Sci. , 221, 5909. 13. Vohs, J. M.; Barteau, M. A. J. Phys. Chem. , 95, 227. 14. Cunningham, J.; Hodnett, B. K.; Illyas, M.; Tobin, J.; Leahy, E. L.; Fierro, J. L. G. Discuss. Farady Soc. , 72, 283. , 15. Parrot, S. L.; Rogers, Jr. J. W.; White, J. M. Appl. Surf. Sci. 1, 443. 16. Lahousse, C.; Bachelier, J.; Lavally, J. C.; Lauron-Pernot, H.; LeGovie, A. M. J. Mol. Cata. , 87, 329. 17. Kannan, S. V.; Pillai, C. N. Curr. Sci. , 23, 665. 18. Bassett, D.; Habgood, H. W. J. Phys. Chem. , 64, 769. 19. Buchang, S.; Burton, H. D. J. Catal. , 57, 359.
1989 1986 1984 1974 1990 1995 1990 1986 1991 1989 1991 1981 1978 1994 1968 1960 1995
Você também pode gostar
- 030-035 225isomerisationDocumento6 páginas030-035 225isomerisationThương ĐơAinda não há avaliações
- Effect of Resorcinol Formaldehyde Resin Gel On TheDocumento8 páginasEffect of Resorcinol Formaldehyde Resin Gel On ThehoseiAinda não há avaliações
- Ref 15Documento7 páginasRef 15giriAinda não há avaliações
- AkineticsandsurfaceWGS-Ross-1973 ImportDocumento8 páginasAkineticsandsurfaceWGS-Ross-1973 ImportleonardoAinda não há avaliações
- A Kella 1984Documento8 páginasA Kella 1984AntonyHurtadoCristobalAinda não há avaliações
- B120432 1279 PDFDocumento6 páginasB120432 1279 PDFCarolina PalacioAinda não há avaliações
- Kinetic Study of Selective Gas-Phase Oxidation of Isopropanol To Acetone UsingDocumento5 páginasKinetic Study of Selective Gas-Phase Oxidation of Isopropanol To Acetone UsingHaroon Ur RashidAinda não há avaliações
- Size Control of Iron Oxide Nanoparticles Using Reverse Microemulsion Method: Morphology, Reduction and Catalytic Activity in CO HydrogenationDocumento28 páginasSize Control of Iron Oxide Nanoparticles Using Reverse Microemulsion Method: Morphology, Reduction and Catalytic Activity in CO HydrogenationNguyễn Thanh TùngAinda não há avaliações
- Zhang 2014Documento5 páginasZhang 2014oviabeautyAinda não há avaliações
- Sharma 2015Documento11 páginasSharma 2015CarolinaQuimbayaAinda não há avaliações
- New Method For Preparing Highly Effective Catalyst For Hydrodesulfurisation (HDS)Documento18 páginasNew Method For Preparing Highly Effective Catalyst For Hydrodesulfurisation (HDS)rancakAinda não há avaliações
- Comparative Studies of Low-Temperature Water-Gas Shift Reaction Over PT Ceo, Au Ceo, and Au Fe O CatalystsDocumento7 páginasComparative Studies of Low-Temperature Water-Gas Shift Reaction Over PT Ceo, Au Ceo, and Au Fe O CatalystsLucas MarchiniAinda não há avaliações
- s10562 019 02678 XDocumento13 páginass10562 019 02678 XYoki YulizarAinda não há avaliações
- Methane Steam ReformingDocumento8 páginasMethane Steam ReformingPanda BoyAinda não há avaliações
- Temperature Invariance of Nacl Solubility in Water: Inferences From Salt Water Cluster Behavior of Nacl, KCL, and NH CLDocumento8 páginasTemperature Invariance of Nacl Solubility in Water: Inferences From Salt Water Cluster Behavior of Nacl, KCL, and NH CLconker4Ainda não há avaliações
- Shahrazad Kalid Ramzy XDocumento23 páginasShahrazad Kalid Ramzy XrawadAinda não há avaliações
- Surface Modification of A Granular Activated Carbon by CitriDocumento9 páginasSurface Modification of A Granular Activated Carbon by CitriAnonymous ggzcpPM4Ainda não há avaliações
- Dry Reforming of CH Over Solid Solutions of Lani Co O: Gustavo Valderrama, A. Kiennemann, M.R. GoldwasserDocumento7 páginasDry Reforming of CH Over Solid Solutions of Lani Co O: Gustavo Valderrama, A. Kiennemann, M.R. GoldwasserMarina CirqueiraAinda não há avaliações
- Qafoku 2006Documento15 páginasQafoku 2006Jaime Jaramillo GutierrezAinda não há avaliações
- AC Catalst PTDocumento5 páginasAC Catalst PTJarretAinda não há avaliações
- 2000 Effects of Oxidant and Solvent On The Liquid Phase Cyclohexane Oxidation Catalyzed by Ce Exchanged Zeolite YDocumento7 páginas2000 Effects of Oxidant and Solvent On The Liquid Phase Cyclohexane Oxidation Catalyzed by Ce Exchanged Zeolite YYash GokaniAinda não há avaliações
- ZR OCl 2Documento9 páginasZR OCl 2Daniel RivaldiAinda não há avaliações
- Effect of Different Potassium Species On The Deactivation of V2O5-WO3/TiO2 SCR Catalyst: Comparison of K2SO4, KCL and K2ODocumento7 páginasEffect of Different Potassium Species On The Deactivation of V2O5-WO3/TiO2 SCR Catalyst: Comparison of K2SO4, KCL and K2OLilyAinda não há avaliações
- The Hydrogenation of Toluene and O-, M-, and P-Xylene Over Palladium I. Kinetic Behavior and O-Xylene IsomerizationDocumento16 páginasThe Hydrogenation of Toluene and O-, M-, and P-Xylene Over Palladium I. Kinetic Behavior and O-Xylene Isomerizationsj singhAinda não há avaliações
- The Kinetics of The Oxidation of Hydrogen Chloride Over Molten Salt CatalystsDocumento10 páginasThe Kinetics of The Oxidation of Hydrogen Chloride Over Molten Salt CatalystsAnne PorterAinda não há avaliações
- Esterification of Oleic Acid With Glycerol in The PresenceDocumento5 páginasEsterification of Oleic Acid With Glycerol in The PresencesapastuchAinda não há avaliações
- Svedov Ve Ark.Documento16 páginasSvedov Ve Ark.Mert GüngörAinda não há avaliações
- Kinetic Study of The Catalytic Reforming of Methane With Carbon Dioxide To Synthesis Gas Over Ni - La2O3 Catalyst PDFDocumento8 páginasKinetic Study of The Catalytic Reforming of Methane With Carbon Dioxide To Synthesis Gas Over Ni - La2O3 Catalyst PDFMarcus NguyễnAinda não há avaliações
- Jae Lee-2008-Ethylbenzene Dehydrogenation IntoDocumento12 páginasJae Lee-2008-Ethylbenzene Dehydrogenation IntogeocottonAinda não há avaliações
- Kinetics of N-Dodecane Dehydrogenation On Promoted Platinum CatalystDocumento11 páginasKinetics of N-Dodecane Dehydrogenation On Promoted Platinum CatalystSoroush KaramianAinda não há avaliações
- Ziegler KJ 2003bDocumento6 páginasZiegler KJ 2003bRiyasath ReihanAinda não há avaliações
- A Comparison of Surface Acidic Features Betweentetragonal and Monoclinic Nanostructured ZirconiaDocumento8 páginasA Comparison of Surface Acidic Features Betweentetragonal and Monoclinic Nanostructured ZirconiarafelAinda não há avaliações
- Highly Sensitive Ethanol Gas Sensor Based On in O Spheres: Ionics (2021) 27:3647-3653Documento7 páginasHighly Sensitive Ethanol Gas Sensor Based On in O Spheres: Ionics (2021) 27:3647-3653MCG GroupAinda não há avaliações
- 8-ACS SusDocumento8 páginas8-ACS Susvenkatesha_3125078Ainda não há avaliações
- h2 O2 ProductionDocumento9 páginash2 O2 ProductionVaibhav ChaudharyAinda não há avaliações
- Articulo ProyectoDocumento9 páginasArticulo ProyectoHenry ArenasAinda não há avaliações
- Energy Conversion and ManagementDocumento43 páginasEnergy Conversion and ManagementnursyifaaAinda não há avaliações
- Acidity and Basicity of Metal Oxide Surfaces 1 II. Determination by Catalytic Decomposition of IsopropanolDocumento9 páginasAcidity and Basicity of Metal Oxide Surfaces 1 II. Determination by Catalytic Decomposition of Isopropanolnguyennha1211Ainda não há avaliações
- Gas-Phase Hydroformylation of Propene Over Silica-Supported PPH - Modified Rhodium CatalystsDocumento9 páginasGas-Phase Hydroformylation of Propene Over Silica-Supported PPH - Modified Rhodium CatalystsIlireaAinda não há avaliações
- A Comparative Study of Water-Gas-Shift Reaction Over Ceria Supported Metallic CatalystsDocumento8 páginasA Comparative Study of Water-Gas-Shift Reaction Over Ceria Supported Metallic CatalystsleonardoAinda não há avaliações
- Active Sites of Ni2PSiO2 Catalyst For Hydrodeoxygenation of Guaiacol A Joint XAFS and DFT Study PDFDocumento9 páginasActive Sites of Ni2PSiO2 Catalyst For Hydrodeoxygenation of Guaiacol A Joint XAFS and DFT Study PDFblackjack_027Ainda não há avaliações
- 5.suchi Dissertation M.phil - Copy EditedDocumento16 páginas5.suchi Dissertation M.phil - Copy EditedDEBABRATA BEHERAAinda não há avaliações
- Wei Li, Shan Tan, Yun Shi, Sujing Li: SciencedirectDocumento8 páginasWei Li, Shan Tan, Yun Shi, Sujing Li: SciencedirectAnindita IndrianaAinda não há avaliações
- Synthesis of Nanosized R-Nickel Hydroxide by A Sonochemical MethodDocumento4 páginasSynthesis of Nanosized R-Nickel Hydroxide by A Sonochemical MethodemediageAinda não há avaliações
- Part-2 Kinetics of ZNS RoastingDocumento7 páginasPart-2 Kinetics of ZNS RoastingakshukAinda não há avaliações
- Tin (II) Hydroxy Chloride - Heterogeneous Catalyst For Condensation ReactionDocumento6 páginasTin (II) Hydroxy Chloride - Heterogeneous Catalyst For Condensation ReactionVijaykumar MarakattiAinda não há avaliações
- Analytical Chemistry Volume 28 Issue 10 1956 (Doi 10.1021/ac60118a012) Pflaum, R. T. Howick, L. C. - Spectrophotometric Determination of Potassium With Sodium TetraphenylborateDocumento3 páginasAnalytical Chemistry Volume 28 Issue 10 1956 (Doi 10.1021/ac60118a012) Pflaum, R. T. Howick, L. C. - Spectrophotometric Determination of Potassium With Sodium TetraphenylboratekhqqxyzAinda não há avaliações
- CoSiO2 For Fischer-Tropsch Synthesis Comparison...Documento5 páginasCoSiO2 For Fischer-Tropsch Synthesis Comparison...Genesis CalderónAinda não há avaliações
- Pyrite PaperDocumento6 páginasPyrite PaperPham Thi HoaAinda não há avaliações
- Ç GüldürDocumento15 páginasÇ GüldürsencanlisAinda não há avaliações
- Synthesis, Characterization and Catalytic Properties of PtCeO2-Al2O3 and PtLa2O3-Al2O3 Sol-Gel Derived CatalystsDocumento9 páginasSynthesis, Characterization and Catalytic Properties of PtCeO2-Al2O3 and PtLa2O3-Al2O3 Sol-Gel Derived CatalystsSangHao NgAinda não há avaliações
- Zaidi 2005Documento5 páginasZaidi 2005daniels calderonAinda não há avaliações
- Effects of Metal Loading and Support For Supported Cobalt CatalystDocumento9 páginasEffects of Metal Loading and Support For Supported Cobalt CatalystThemba MasukuAinda não há avaliações
- Nickel Nanoparticles Catalyse Reversible Hydration of Carbon Dioxide For Mineralization Carbon Capture and StorageDocumento6 páginasNickel Nanoparticles Catalyse Reversible Hydration of Carbon Dioxide For Mineralization Carbon Capture and StorageRahul GogiaAinda não há avaliações
- Catalysis Communications: Ryan M. West, Mark H. Tucker, Drew J. Braden, James A. DumesicDocumento4 páginasCatalysis Communications: Ryan M. West, Mark H. Tucker, Drew J. Braden, James A. Dumesicmagomago87Ainda não há avaliações
- Pyrite Oxidation in Alkaline Solutions: Nature of The Product LayerDocumento14 páginasPyrite Oxidation in Alkaline Solutions: Nature of The Product LayerAlejandro Valenzuela100% (1)
- 1-PbSO4 Leaching in Citric Acid Sodium Citrate Solution and Subsequent ...Documento10 páginas1-PbSO4 Leaching in Citric Acid Sodium Citrate Solution and Subsequent ...Gregorio Antonio Valero VerdeAinda não há avaliações
- Korean Journal of Chemical Engineering, 28 (2), 383-387 (2011)Documento5 páginasKorean Journal of Chemical Engineering, 28 (2), 383-387 (2011)KhangKGBAinda não há avaliações
- Fourth International Conference on Non-Aqueous Solutions: Vienna 1974No EverandFourth International Conference on Non-Aqueous Solutions: Vienna 1974V. GutmannAinda não há avaliações
- Reactive Transport Modeling: Applications in Subsurface Energy and Environmental ProblemsNo EverandReactive Transport Modeling: Applications in Subsurface Energy and Environmental ProblemsYitian XiaoAinda não há avaliações
- Lifestyle DiseasesDocumento14 páginasLifestyle Diseaseskrishna_nimeshAinda não há avaliações
- Constitution of India - Wikipedia, The Free EncyclopediaDocumento14 páginasConstitution of India - Wikipedia, The Free Encyclopediakrishna_nimeshAinda não há avaliações
- Simulation Study For Optimization of LSD Stripper: ObjectiveDocumento4 páginasSimulation Study For Optimization of LSD Stripper: Objectivekrishna_nimeshAinda não há avaliações
- A Guideline For Design Pressure - Part 1 PDFDocumento4 páginasA Guideline For Design Pressure - Part 1 PDFkrishna_nimeshAinda não há avaliações
- Manorama Yearbook 2014Documento3 páginasManorama Yearbook 2014Heena Soni100% (1)
- Syria Profile - Timeline - BBC NewsDocumento15 páginasSyria Profile - Timeline - BBC Newskrishna_nimeshAinda não há avaliações
- Jecetv3i2c 3Documento20 páginasJecetv3i2c 3krishna_nimeshAinda não há avaliações
- Thermal Efficiency Fired HeaterDocumento5 páginasThermal Efficiency Fired Heatermuhammad_asim_10Ainda não há avaliações
- United States: (12) Patent Application Publication (10) Pub. No.: US 2013/0211147 A1Documento8 páginasUnited States: (12) Patent Application Publication (10) Pub. No.: US 2013/0211147 A1krishna_nimeshAinda não há avaliações
- Global Kinetics of Direct Dimethyl Ether Synthesis Process From Syngas in Slurry Reactor Over A Novel Cu ZN Al ZR Slurry CatalystDocumento7 páginasGlobal Kinetics of Direct Dimethyl Ether Synthesis Process From Syngas in Slurry Reactor Over A Novel Cu ZN Al ZR Slurry Catalystkrishna_nimeshAinda não há avaliações
- Outlines of Thesis WritingDocumento20 páginasOutlines of Thesis Writingkrishna_nimeshAinda não há avaliações
- ValIdation of DMEDocumento6 páginasValIdation of DMEkrishna_nimeshAinda não há avaliações
- DME FlowsheetsDocumento4 páginasDME Flowsheetskrishna_nimeshAinda não há avaliações
- Total Site AnalysisDocumento21 páginasTotal Site Analysiskrishna_nimesh100% (1)
- Coal Project Report FinalDocumento42 páginasCoal Project Report Finalkrishna_nimeshAinda não há avaliações
- Potions Round 1 ResultDocumento1 páginaPotions Round 1 Resultkrishna_nimeshAinda não há avaliações
- PROCEDUREDocumento1 páginaPROCEDUREkrishna_nimeshAinda não há avaliações
- Exercise ScheduleDocumento1 páginaExercise Schedulekrishna_nimeshAinda não há avaliações
- Round 1Documento12 páginasRound 1krishna_nimeshAinda não há avaliações
- Round 2Documento3 páginasRound 2krishna_nimeshAinda não há avaliações
- KrishnaDocumento16 páginasKrishnakrishna_nimeshAinda não há avaliações
- Round 1Documento12 páginasRound 1krishna_nimeshAinda não há avaliações
- Pub 302 WI Alcohol Beverage and Tobacco Laws For Retailers - December 2016Documento16 páginasPub 302 WI Alcohol Beverage and Tobacco Laws For Retailers - December 2016Woinlah NuiAinda não há avaliações
- Chemistry LL SyllabusDocumento19 páginasChemistry LL SyllabusRajat Kumar VishwakarmaAinda não há avaliações
- Oxidaciones CrO3Documento16 páginasOxidaciones CrO3COMPAQSR14Ainda não há avaliações
- Acc. Chem. Res. 1992,25, 504 Gif Chemistry PDFDocumento9 páginasAcc. Chem. Res. 1992,25, 504 Gif Chemistry PDFCarlotaAinda não há avaliações
- Carbonyl CompoundsDocumento14 páginasCarbonyl Compoundsyvg95100% (1)
- DiphenylmethanolDocumento2 páginasDiphenylmethanolAbdul Rahman ObaidAinda não há avaliações
- MIDTERM Mix. Vodka 5Documento19 páginasMIDTERM Mix. Vodka 5Amos CalebAinda não há avaliações
- BASF Agrochemical AdditivesDocumento19 páginasBASF Agrochemical AdditivesAlex100% (3)
- Organic Chemistry Delivery GuideDocumento43 páginasOrganic Chemistry Delivery GuideKizzy Anne Boatswain Carbon100% (1)
- Chem 217Documento5 páginasChem 217Subhabrata MabhaiAinda não há avaliações
- Alcohol Laws of IndiaDocumento11 páginasAlcohol Laws of IndiaNabendu SahaAinda não há avaliações
- TSR 140Documento6 páginasTSR 140Yohana MelianthaAinda não há avaliações
- Introduction To Bar and Beverage ManagementDocumento23 páginasIntroduction To Bar and Beverage ManagementLaliene Rosales100% (1)
- Chapter - 3 Chemistry of Natural ProductsDocumento50 páginasChapter - 3 Chemistry of Natural ProductsAnonymous QG3UqD974Ainda não há avaliações
- Ranjeet ShahiDocumento529 páginasRanjeet Shahiयash ᴍᴀɴGalAinda não há avaliações
- Class10 NSO Level2 2012Documento7 páginasClass10 NSO Level2 2012PraveshAinda não há avaliações
- Conversion of Biomass Platform Molecules Into Fuel Additives and Liquid Hydrocarbon FuelsDocumento32 páginasConversion of Biomass Platform Molecules Into Fuel Additives and Liquid Hydrocarbon FuelsJuanFleitesAinda não há avaliações
- Chanakya National Law University: Project of Sociology ON Liquor Ban in BiharDocumento21 páginasChanakya National Law University: Project of Sociology ON Liquor Ban in BiharAntra AzadAinda não há avaliações
- UP Excise BRAND MRPDocumento162 páginasUP Excise BRAND MRPstuti.wahiAinda não há avaliações
- Bairstow RiggingDocumento168 páginasBairstow Riggingswhite336Ainda não há avaliações
- 0001 Assessment On The Safety of Candelilla Wax, Japan Wax, and BeeswaxDocumento41 páginas0001 Assessment On The Safety of Candelilla Wax, Japan Wax, and BeeswaxEvangeline AntonioAinda não há avaliações
- Indian Luquor ScenarioDocumento12 páginasIndian Luquor Scenariokumar bhanushaliAinda não há avaliações
- Chemistry Nov 05Documento18 páginasChemistry Nov 05Bassem HrakiAinda não há avaliações
- Alcohols TheoryDocumento31 páginasAlcohols TheoryChesta MalhotraAinda não há avaliações
- Sadq PDFDocumento3 páginasSadq PDFHans KaufmannAinda não há avaliações
- Https WWW - Sigmaaldrich.com Technical-Documents Articles Biology Ir-Spectrum-Table - Printerview.htmlDocumento1 páginaHttps WWW - Sigmaaldrich.com Technical-Documents Articles Biology Ir-Spectrum-Table - Printerview.htmlnissa0% (1)
- Facile Oxidation of Aldehydes To Acids and Esters With OxoneDocumento4 páginasFacile Oxidation of Aldehydes To Acids and Esters With Oxonejonster2Ainda não há avaliações
- 1-Sentence-Summary: The Unexpected Joy of Being Sober Will Help You Have ADocumento4 páginas1-Sentence-Summary: The Unexpected Joy of Being Sober Will Help You Have ASainolzii GanboldAinda não há avaliações
- E PVAzubereitungenDocumento6 páginasE PVAzubereitungentedakAinda não há avaliações
- MRP 01042020Documento29 páginasMRP 01042020SAS TrainerAinda não há avaliações