Escolar Documentos
Profissional Documentos
Cultura Documentos
4 - Phytohormones and Elicitor Molecules - ABSCISIC ACID PDF
Enviado por
FCiências0 notas0% acharam este documento útil (0 voto)
181 visualizações20 páginasAbscisic acid (ABA) exerts both short-term (rapid and reversible) and long-term (lasting) controls over plant development. ABA is a ubitiquitous plant hormone in vascular plants, detected also in mosses. The Chemical Structure of ABA determines its physiological activity.
Descrição original:
Título original
4_-_Phytohormones_and_Elicitor_Molecules_-__ABSCISIC_ACID.pdf
Direitos autorais
© © All Rights Reserved
Formatos disponíveis
PDF, TXT ou leia online no Scribd
Compartilhar este documento
Compartilhar ou incorporar documento
Você considera este documento útil?
Este conteúdo é inapropriado?
Denunciar este documentoAbscisic acid (ABA) exerts both short-term (rapid and reversible) and long-term (lasting) controls over plant development. ABA is a ubitiquitous plant hormone in vascular plants, detected also in mosses. The Chemical Structure of ABA determines its physiological activity.
Direitos autorais:
© All Rights Reserved
Formatos disponíveis
Baixe no formato PDF, TXT ou leia online no Scribd
0 notas0% acharam este documento útil (0 voto)
181 visualizações20 páginas4 - Phytohormones and Elicitor Molecules - ABSCISIC ACID PDF
Enviado por
FCiênciasAbscisic acid (ABA) exerts both short-term (rapid and reversible) and long-term (lasting) controls over plant development. ABA is a ubitiquitous plant hormone in vascular plants, detected also in mosses. The Chemical Structure of ABA determines its physiological activity.
Direitos autorais:
© All Rights Reserved
Formatos disponíveis
Baixe no formato PDF, TXT ou leia online no Scribd
Você está na página 1de 20
Abscisic Acid
A Seed Maturation and Stress-Response Hormone
FISIOLOGIA VEGETAL COMPLEMENTAR
(2013/2014)
Phytohormones and Elicitor Molecules
Abscisic acid (ABA) exerts both short-term (rapid
and reversible) and long-term (lasting) controls over
plant development. The hormone has major roles in
plant responses to water stress, drought, low
temperature, and salinity, as well as seed maturation
and bud dormancy.
The extent and timing of plant growth are controlled by coordinated actions
of positive and negative regulators. Some of the most obvious examples of
regulated nongrowth are seed and bud dormancy, adaptative features that
delay growth until environmental conditions are favourable.
A group of growth-inhibiting compounds, including a substance known as
dormin was purified from sycamore leaves collected in early autumn, when
the trees were entering dormancy. Upon discovery that dormin was
chemically identical to a substance that promotes the abscission of cotton
fruits, abscisin II, the compound was renamed abscisic acid (ABA) (Figure
23.1).
ABA is now recognized as an important plant hormone that regulates growth
and stomatal closure, particularly when the plant is under stress. Another
important function is its regulation of seed maturation and dormancy.
Ironically, ABAs effects on abscission remain controversial: In many species,
ABA appears to promote senescence (i.e., the events preceding abscission)
but not abscission itself.
Occurrence, Chemical Structure, and Measurement of ABA
ABA is a ubitiquitous plant hormone in vascular plants, detected also in mosses. Within the
plant, ABA has been detected in every major organ or living tissue from the root cap to the
apical bud. ABA is synthesized in almost all cells that contain chloroplasts or amyloplasts.
The chemical structure of ABA determines its physiological activity.
ABA is a 15-carbon compound that resembles the terminal portion of some carotenoid
molecules, and it exists in cis and trans, as well as S and R forms (Figure 23.1). The
orientation of the carboxyl group at carbon 2 determine the cis and trans isomers of ABA.
Nearly all naturally occurring ABA is in the cis form, and by convention the name abscisic acid
refers to the cis isomer.
ABA can be measured by bioassays, gas
chromatography, high-pressure liquid
chromatography, and immunoassays.
Bioassays for ABA, include inhibition of
coleoptile growth, germination, and
giberelic acid (GA)-induced -amylase
synthesis. Rapid inductive responses such
as promotion of stomatal closure and gene
expression are also associated with ABA..
As with other hormones, the response to ABA depends on its concentration
within the tissue and on the sensitivity of the tissue to the hormone.
The processes of biosynthesis, catabolism, compartmentation, and
transport all contribute to the concentration of active hormone in plant
tissues at any given stage of development.
Biosynthesis, Metabolism, and Transport of ABA
Biosynthesis, Metabolism, and Transport of ABA
ABA is produced from a carotenoid precursor (C
40
)
that is synthesized from isopentenyl diphosphate (IPP)
in plastids, with final steps completed in cytoplasm
(Figure 23.2).
Under stress conditions, trans-violaxanthin, is
converted to another C
40
compound, trans-
neoxanthin, by a reaction dependent on the product
of the Arabidopsis ABA4 locus.
Following isomerization by an as-yet unidentified
enzyme(s), 9-cis-neoxanthin can be cleaved by an
enzyme abreviated NCED
*
to form the C
15
compound
xanthoxin, that has physiological properties similar to
those of ABA .
Xanthoxin moves to the cytoplasm, where it is
converted to ABA, via oxidative steps involving the
intermediate(s) ABA-aldehyde and / or possibly
xanthoxi acid (abscisic alcohol).
*9-cis-epoxycarotenoid dioxygenase (NCED)
Biossynthesis, Metabolism, and Transport of ABA
Mutations that block carotenoid biosynthesis reduce ABA levels
and cause precocious germination (Figure 23.3).
Maize (Zea mays) mutants (termed viviparous, vp) that are blocked
at other steps in the carotenoid pathway also have reduced levels
of ABA and exhibit viviparity the precocious germination of seeds
in the fruit while still attached to the plant (Figure 23.3). Viviparity is
a feature of many ABA-deficient seeds.
Conversion of xanthoxin to ABA is catalyzed by a short-chain dehydrogenase / reductase-like (SDR)
enzyme, encoded, by ABA2 locus of Arabidopsis. The final step is catalyzed by a differentially
regulated family of abscisic aldehyde oxidases (AAOs) that all require a molybdenum cofactor
(Figure 23.2).
ABA levels can change dramatically during
development or in response to environmental
changes including dehydration stress (Figure 23.4).
In developing seeds, for example, ABA levels can
increase 100-fold within few days, reaching
concentrations in the micromolar range, and then
decline to very low levels as maturation proceeds.
Under conditions of water stress, ABA in the leaves
can increase 50-fold within 4 to 8 hours (Figure 23.4)
Part of this increase is due to increased expression
of biosynthetic enzymes, but the specific enzymes
depend on the tissue and the signal.
As with other plant hormones, the concentration of
free ABA in the cytosol is also regulated by
degradation, compartmentation, conjugation, and
transport.
Biossynthesis, Metabolism, and Transport of ABA
ABA concentrations in tissues are highly variable
Biossynthesis, Metabolism, and Transport of ABA
ABA is translocated in vascular tissue
ABA is transported by both the xylem and the
phloem, but it is normally more abundant in the
phloem sap.
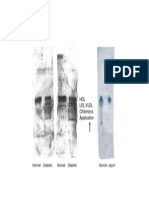
During water stress, ABA accumulates first in
shoot vascular tissue, and only later appears in
roots and guard cells (Figure 23.5).
Biossynthesis, Metabolism, and Transport of ABA
ABA is translocated in vascular tissue
During water stress, pH changes in apoplast and cytosol increase the amount of ABA reaching the
guard cells to stimulate their closure (Figure 23.6).
During the early stages of water stress, the pH of the xylem sap becomes more alkaline, increasing
from about pH 6.3 to about pH 7.2. Stress-induced alkalinization of the apoplast favours formation of
the dissociated form of abscisic acid, ABA
-
. At the same time, dehydration also acidifies the cytosol,
contributing to ABA release from its sites of synthesis and decreasing uptake by mesophyll cells. Both
of these pH changes increase the amount of ABA reaching the guard cells via the transpiration stream
(Figure 23.6).
In this way ABA can be redistributed
in the leaf without any increase in the
total ABA level. Therefore, the
increase in xylem sap pH may
function as an additional root signal
that promotes early closure of the
stomata.
Developmental and Physiological Effects of ABA
ABA has roles in regulating seed development, seed and bud dormancy, germination, vegetative growth,
senescence, stomatal regulation, as well as stress response.
ABA influences many other aspects of plant development by interacting, usually as an antagonist, with
auxin, cytokinin, gibberellin, ethylene, and brassinosteroids.
ABA regulates seed maturation
Seed development can be divided into three phases of approximately equal duration:
1st Phase. Is characterized by cell divisions and tissue differentiation, the zygote undergoes
embryogenesis and the endosperm tissue proliferates.
2nd Phase. Cell divisions cease and storage compounds accumulate.
3rd Phase Embryos of orthodox seeds become tolerant to desiccation, and the seeds dehydrate, losing
up to 90% of their water. As a consequence of dehydration, metabolism comes to a halt and the seed
enters a quescent (resting) sate. In some cases the seed becomes dormant as well. Unlike quiescent
seeds, which will germinate upon rehydration, dormant seeds require additional treatment or signals for
germination occur.. In contrast to orthodox seeds, recalcitrant seeds do not complete this phase, so have a
high moisture content at maturity and are not desiccation tolerant.
Typically, the ABA content of seeds is very low early in embryogenesis, reaches a maximum at about halfway point, and then gradually
falls to low levels as the seed reaches maturity.
Developmental and Physiological Effects of ABA
ABA inhibits precocious germination and viviparity
ABA promotes seed storage reserve accumulation and desiccation tolerance
During mid- to late embryogenesis, when seed ABA levels are highest, seeds accumulate storage
compounds that will support seedling growth at germination.
ABA promote the acquisition of desiccation tolerance. Desiccation can severely damage membranes
and other celular constituents. However, in seeds such damage is prevented as, besides storage of
sugars and storage proteins, ABA promotes the synthesis and accumulation of the so-called Late
embryogenesis-abundant (LEA) proteins. These molecules are thought to interact to form a glassy
state (a highly viscous liquid with very slow diffusion and therefore limited chemical reactions) involved
in desiccation tolerance.
Seed dormancy can be regulated by ABA and environmental factors
Developmental and Physiological Effects of ABA
Seed dormancy can be regulated by ABA and environmental factors
In many cases a viable (living) seed will not germinate even if all the necessary environmental
conditions are satisfied. This phenomenon is termed seed dormancy.
Seed dormancy may result from coat-imposed dormancy, embryo dormancy, or both. Dormancy
imposed on the embryo by seed coat and other enclosing tissues, such as endosperm, pericarp, or
extrafloral organs, is known as coat-imposed dormancy. The embryos of such seeds will germinate
readily in the presence of water and oxygen once the seed coat and other surrounding tissues have
been either removed or damaged.
Seed dormancy tha is intrinsic to the embryo and is not due to any influence of the seed coat or other
surrounding tissues is called embryo dormancy.
Embryo dormancy is thought to be due to the presence of inhibitors, especially ABA, as well as the
absence of growth promoters, such as GA.
Maintenance of dormancy and the loss of embryo dormancy depends on the relative ratios ABA to GA.
The levels of ABA and GA are regulated by their synthesis and catabolism which are catalyzed by
specific isozymes whose expression is controlled by developmental and environmental factors, such as
after-ripening; chilling; light
Developmental and Physiological Effects of ABA
ABA inhibits GA-induced enzyme production
In addition to the ABA-GA antagonism affecting seed dormancy, ABA inhibits the GA-induced
synthesis of hydrolytic enzymes that are essential for breakdown of storage reserves in germinating
seeds. For example, ABA inhibits the GA-dependent enzyme synthesis by inhibiting the transcription
of -amylase mRNA.
Developmental and Physiological Effects of ABA
ABA promotes root growth and inhibits shoot growth at low water potentials
ABA has different effects on the growth of roots and shoots and the effects are strongly dependent on
the water status of the plant.(Figure 23.11).
The experimental results sugest that endogenous ABA promotes shoot elongation in well-watered plants and
have a much higher effect in promoting root growth during watrer stress by inhibiting ethylene production.
Developmental and Physiological Effects of ABA
ABA closes stomata in response to water stress
ABA regulates ion channels and the plasma membrane ATPase in guard cells
In response to water stress, ABA closes stomata by triggering a transient membrane depolarization due
to influx of positive charge in guard cells (Figure 23.12).
These fleeting changes cause a massive long-term efflux of K
+
and anions from the cell, reducing
guard cell turgor pressure (Figure 23.13).
Developmental and Physiological Effects of ABA
ABA induced changes in the guard cells can inhibit the plasma membrane H
+
-ATPase, thereby
contributing to membrane depolarization (Figure 23.14).
ABA has roles in regulating seed development, seed and bud dormancy, germination,
vegetative growth, senescence, stomatal regulation, as well as stress response.
In the developing seed, the genotype of the embryo and endosperm controls ABA synthesis
that is essential to dormancy induction, whereas the maternal genotype of the seed coat
controls ABA accumulation in med-embyogenesis that suppresses viviparity.
In seed development , ABA promotes the synthesis of storage proteins and lipids, as well as
specific proteins involved in desiccation tolerance.
Seed dormancy and germination are controlled by the ratio ABA to giberelic acid (GA).
In germination seeds, ABA inhibits the GA-induced synthesis of hydrolytic enzymes.
ABAs effects on the growth of roots and shoots depend on the water status of the plant
ABA greatly accelerates the senescence of leaves, thereby increasing ethylene formation
and stimulating abscission.
Developmental and Physiological Effects of ABA
(Summary)
ABA greatly accelerates the senescence of leaves, thereby increasing ethylene formation
and stimulating abscission.
ABA accumulates in dormancy buds, inhibiting their growth; ABA may interact with growth-
promoting hormones such as cytokinins and gibberellins.
In response to water stress, ABA closes stomata by triggering a transient membrane
depolarization due to influx of positive charge in guard cells.These fleeting changes cause a
massive long-term efflux of K
+
and anions from the cell, reducing guard cell turgor pressure.
ABA induced changes in the guard cells can inhibit the plasma membrane H
+
-ATPase,
thereby contributing to membrane depolarization.
Developmental and Physiological Effects of ABA
(Summary)
END
Você também pode gostar
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceNo EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceNota: 4 de 5 estrelas4/5 (895)
- 01Q PDFDocumento1 página01Q PDFFCiênciasAinda não há avaliações
- Never Split the Difference: Negotiating As If Your Life Depended On ItNo EverandNever Split the Difference: Negotiating As If Your Life Depended On ItNota: 4.5 de 5 estrelas4.5/5 (838)
- 02Q PDFDocumento1 página02Q PDFFCiênciasAinda não há avaliações
- The Yellow House: A Memoir (2019 National Book Award Winner)No EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Nota: 4 de 5 estrelas4/5 (98)
- Amber 15Documento883 páginasAmber 15FCiênciasAinda não há avaliações
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeNo EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeNota: 4 de 5 estrelas4/5 (5794)
- 8 - Secondary Metabolites - TERPENOIDS ALKALOIDS PHENOLICS PDFDocumento82 páginas8 - Secondary Metabolites - TERPENOIDS ALKALOIDS PHENOLICS PDFFCiênciasAinda não há avaliações
- 03Q PDFDocumento2 páginas03Q PDFFCiênciasAinda não há avaliações
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaNo EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaNota: 4.5 de 5 estrelas4.5/5 (266)
- 7.02 Recombinant DNA Methods Spring 2005 Exam Study Questions Answer KeyDocumento10 páginas7.02 Recombinant DNA Methods Spring 2005 Exam Study Questions Answer KeyFCiênciasAinda não há avaliações
- The Little Book of Hygge: Danish Secrets to Happy LivingNo EverandThe Little Book of Hygge: Danish Secrets to Happy LivingNota: 3.5 de 5 estrelas3.5/5 (400)
- 1ºteste 2013 2014 Catarina Cunha PDFDocumento4 páginas1ºteste 2013 2014 Catarina Cunha PDFFCiênciasAinda não há avaliações
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureNo EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureNota: 4.5 de 5 estrelas4.5/5 (474)
- Exercício Mutagénese Sítio Dirigida - Questões PDFDocumento25 páginasExercício Mutagénese Sítio Dirigida - Questões PDFFCiênciasAinda não há avaliações
- Review: S. E. Borggreve, R. de Vries and R. P. F. DullaartDocumento19 páginasReview: S. E. Borggreve, R. de Vries and R. P. F. DullaartFCiênciasAinda não há avaliações
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryNo EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryNota: 3.5 de 5 estrelas3.5/5 (231)
- Alcohol - Metab p450 PDFDocumento17 páginasAlcohol - Metab p450 PDFFCiênciasAinda não há avaliações
- Aula071314 PDFDocumento33 páginasAula071314 PDFFCiênciasAinda não há avaliações
- The Emperor of All Maladies: A Biography of CancerNo EverandThe Emperor of All Maladies: A Biography of CancerNota: 4.5 de 5 estrelas4.5/5 (271)
- 08 Recombination PDFDocumento17 páginas08 Recombination PDFFCiênciasAinda não há avaliações
- The Unwinding: An Inner History of the New AmericaNo EverandThe Unwinding: An Inner History of the New AmericaNota: 4 de 5 estrelas4/5 (45)
- LipidogramaDocumento1 páginaLipidogramaFCiênciasAinda não há avaliações
- S1744309110011589 PDFDocumento5 páginasS1744309110011589 PDFFCiênciasAinda não há avaliações
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersNo EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersNota: 4.5 de 5 estrelas4.5/5 (344)
- Aula 6: Bioquimica IIDocumento21 páginasAula 6: Bioquimica IIFCiênciasAinda não há avaliações
- Team of Rivals: The Political Genius of Abraham LincolnNo EverandTeam of Rivals: The Political Genius of Abraham LincolnNota: 4.5 de 5 estrelas4.5/5 (234)
- 08 Recombination PDFDocumento15 páginas08 Recombination PDFFCiênciasAinda não há avaliações
- Remazol Ultra RGB Feb 2010Documento23 páginasRemazol Ultra RGB Feb 2010rauftextile0% (2)
- The Calorific Value of A Fuel Is TheDocumento4 páginasThe Calorific Value of A Fuel Is TheRana PrathapAinda não há avaliações
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreNo EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreNota: 4 de 5 estrelas4/5 (1090)
- Elements Compounds and Mixtures Revision 1Documento6 páginasElements Compounds and Mixtures Revision 1api-310569693Ainda não há avaliações
- Buku Miller's Anesthesia BAB 20 Dan 21Documento88 páginasBuku Miller's Anesthesia BAB 20 Dan 21Rahma TomuAinda não há avaliações
- T105 Trojan Data SheetsDocumento126 páginasT105 Trojan Data SheetsJose Luis PandoAinda não há avaliações
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyNo EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyNota: 3.5 de 5 estrelas3.5/5 (2259)
- Vermicomposting GuideDocumento34 páginasVermicomposting GuideRobin RheaumeAinda não há avaliações
- Jasmuheen - Telepathic CommunicationDocumento6 páginasJasmuheen - Telepathic Communicationmelrakki100% (3)
- Food Chemistry: M.S. Altaki, F.J. Santos, M.T. GalceranDocumento6 páginasFood Chemistry: M.S. Altaki, F.J. Santos, M.T. GalceranAberuAinda não há avaliações
- 2013v12 New Guide PDFDocumento4 páginas2013v12 New Guide PDFlucianolimapgecivAinda não há avaliações
- 5 OzoneDocumento14 páginas5 OzoneParakram KharelAinda não há avaliações
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)No EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Nota: 4.5 de 5 estrelas4.5/5 (121)
- Conservation Equations and Modeling of Chemical and Biochemical ProcessesDocumento650 páginasConservation Equations and Modeling of Chemical and Biochemical ProcessesArman BasmacıoğluAinda não há avaliações
- Boq 153501Documento28 páginasBoq 153501kamakhyaguri pwdAinda não há avaliações
- FumigationDocumento2 páginasFumigationJeje ChouAinda não há avaliações
- Principles of Food Processing and Halal Requirement (WEEK 11)Documento48 páginasPrinciples of Food Processing and Halal Requirement (WEEK 11)aishah1997Ainda não há avaliações
- 3Documento436 páginas3shaziadurraniAinda não há avaliações
- HKR - Expansion Joints CatalogueDocumento80 páginasHKR - Expansion Joints Cataloguemksingh_22Ainda não há avaliações
- Graphene and Semiconductors: International Webinar OnDocumento3 páginasGraphene and Semiconductors: International Webinar Onhong kongAinda não há avaliações
- Chapter 6 Thermodynamics: The First Law: Systems, States, and Energy (Sections 6.1-6.8)Documento12 páginasChapter 6 Thermodynamics: The First Law: Systems, States, and Energy (Sections 6.1-6.8)MostafaRock100% (2)
- Concentration of Solutions DLPDocumento3 páginasConcentration of Solutions DLPLouise Meara Severo70% (10)
- (LIFS1901) (2019) (F) Final Cx5mue7 98268Documento9 páginas(LIFS1901) (2019) (F) Final Cx5mue7 98268yuen lok hinAinda não há avaliações
- Accesorios de Carga CROSBYDocumento52 páginasAccesorios de Carga CROSBYRodolfo AlboresAinda não há avaliações
- Deocument 9172Documento21 páginasDeocument 9172jeffrey.beach2560% (1)
- Laboratory SafetyDocumento2 páginasLaboratory Safetyann wamburaAinda não há avaliações
- SDS PLS2Documento8 páginasSDS PLS2Đinh Quốc LiêmAinda não há avaliações
- AGRI 51 Soil Science NotesDocumento10 páginasAGRI 51 Soil Science Notesgljae479Ainda não há avaliações
- Threshold Limit Values (TLV)Documento7 páginasThreshold Limit Values (TLV)chem_taAinda não há avaliações
- Chemically Enriched UniverseDocumento41 páginasChemically Enriched UniverseMark JamesAinda não há avaliações
- AcarboseDocumento2 páginasAcarboseRaquel BcAinda não há avaliações
- Air PollutionDocumento41 páginasAir PollutionferzanaAinda não há avaliações