Escolar Documentos
Profissional Documentos
Cultura Documentos
Cloning Cauliflower
Enviado por
brijmohansingh401124Descrição original:
Direitos autorais
Formatos disponíveis
Compartilhar este documento
Compartilhar ou incorporar documento
Você considera este documento útil?
Este conteúdo é inapropriado?
Denunciar este documentoDireitos autorais:
Formatos disponíveis
Cloning Cauliflower
Enviado por
brijmohansingh401124Direitos autorais:
Formatos disponíveis
Caulifower
Teachers and technicians notes
Cloning
National Centre for Biotechnology Education
1.1
2 www.ncbe.reading.ac.uk Cloning caulifower
Plant tissue culture is widely used in commercial
horticulture and scientifc research. It involves growing
individual plant cells or tissues in aseptic conditions.
In theory, almost any part of a plant can be used as
the source of cells (the explant) from which tissue is
cultured. In practice, however, young, actively-dividing
(meristematic) cells give the best results. When placed
on a sterile growth medium containing the right blend
of vitamins, minerals and plant hormones (plant growth
regulators), plus sucrose as a source of energy, these cells
can be triggered into growing and dividing. Under the
infuence of light and various plant growth substances,
they can produce shoots and roots, then will eventually
develop into small plants.
Plant tissue culture methods were frst devised by
scientists in the 1950s and are now used routinely to study
biochemistry, cell biology and genetics.
Tissue culture is applied in a commercial context too
micropropagation techniques enable the production
of large numbers of disease-free, genetically-identical
plants (clones) for sale. Increasingly, the technique is also
used to rescue critically-endangered species, by growing
many plants from a small amount of starting material so
that they can be re-introduced into habitats from which
they have almost become extinct.
Conservation Biotechnology at the Royal Botanic
Gardens, Kew, specialises in saving plant species on the
verge of extinction. Tissue culture forms a vital part of
their strategy. For instance, Cylindrocline lorencei is a small
tree which was native to Mauritius. It became extinct in
the wild in 1990, when the last-known specimen died.
Although some seeds had been collected from the tree,
these could not be germinated. Using tissue culture
methods, however, scientists at Brest Botanic Gardens (in
Brittany) managed to culture plants from the embryos in
the seeds. Back-up cultures were sent to Kew for further
micropropagation. Fifteen rooted plants were eventually
sent from Kew to Mauritius for potential re-introduction.
Introduction
In the brief period after the introduction of the GCSE
examination in England and Wales, but before the National
Curriculum came fully into force, biotechnology education
fourished. Practical experience of plant tissue culture was
called for in many biology and in some science syllabuses
notably in the frst incarnation of the Suffolk Science
GCSE course.
A landmark publication triggered this interest: Plant
tissue culture by Tony Storr was published by the ASE
in 1985 (see Further information, page 8). The plant most
frequently used for such work in schools and colleges
was the caulifower. Carrots were sometimes tried, but it
was diffcult to isolate the actively-dividing meristematic
tissue from the tap root. Caulifower, in contrast, proved
ideal for school use because it was readily-available, easy
to isolate the meristematic tissue from (the tips of the
white curd) and robust enough to withstand the rigours of
being handled by students. Caulifower tissue also grows
rapidly: growth can be seen after 10 days and plantlets are
ready for transplantation into soil within 12 weeks. There
was one persistent problem, however: maintaining aseptic
conditions.
With conventional plant tissue culture methods, you
have to surface sterilise the isolated pieces of plant tissue
(explants) in bleach solution. This must then be removed
by several successive washes with sterile water. Not only
is this a lot to prepare, but students rarely achieve success.
Too often, they see no growth at all (the plants are killed by
the bleach) or black fungus grows over everything (due to
incomplete surface sterilisation or poor aseptic technique).
Several solutions have been proposed. Antimicrobial
additives such as silver nitrate, antibiotics and even an
extract of rowan berries (Sorbus sp.) have been added to
plant growth media with varying degrees of success. None
of these methods have overcome the need for autoclaving
the growth media and rinsing water however, and all have
added signifcant expense.
The method used in this kit is different. It was devised by
the Conservation Biotechnology team at the Royal Botanic
Gardens, Kew. It was developed for use in situations where
laboratory facilities are limited, but it is also excellent
for use in schools. The materials are easy to prepare, the
procedure is straightforward and it gives good results. No
autoclaving of media or rinsing water is required the
growth medium simply needs to be microwaved. The
aseptic techniques are safe and reliable no Bunsen
burners are required and neither does disinfectant have
to be rinsed from the plant tissue.
Educational context
3 www.ncbe.reading.ac.uk Cloning caulifower
Straight from the kit
a copy of the Students guide [1]
a 7 mL bijou bottle with cap
a pair of plastic forceps [2]
Prepared in advance by
the teacher or technician
(see pages 45 of this guide for instructions)
Virkon
solution, for sterilising the work surface, made
by dissolving each tablet in 500 mL of tap water.
80 mL of Milton
solution, made using half a 4 g Milton
tablet in each 80 mL of water, for sterilising the forceps
and the caulifower tissue [3]
a vial containing ~10 mL of growth medium
Equipment and materials
Not in the kit: supplied by you
a permanent marker pen
a scalpel
a non-sterile Petri dish lid or base, cutting board or tile
a small (e.g., 100 mL) beaker for waste
a small foret from a caulifower
paper towels
sunny windowsill or lights to illuminate the plant
cultures
1. Supporting PowerPoint and Keynote presentations are available on
the NCBEs web site: www.ncbe.reading.ac.uk/ptc
2. Metal will be damaged by prolonged immersion in Milton
solution.
3. Note that only the tablet form of Milton
is suitable. The liquid form
of the product is another chemical entirely.
Notes
Each student or working group needs
4 www.ncbe.reading.ac.uk Cloning caulifower
For 500 mL of growth medium you will need
Equipment
Eye protection
Heat-proof gloves
Microwave oven
Balance
250 mL measuring cylinder
1 mL syringe for the kinetin stock solution
10 mL syringe for the Milton
solution
1 L beaker (or plastic jug), to ft inside microwave
Glass stirring rod
Materials
6 g Murashige and Skoog (MS) agar powder
10 g (4 sachets) granulated cane sugar [1]
1.25 mL kinetin solution (containing 0.001 g of kinetin
per millilitre of 70% ethanol) [2]
Milton
stock solution (that is, one 4 g Milton
tablet
dissolved in 100 mL of distilled or deinonised water) [3]
Sterile vials with caps [4]
Clingflm
Virkon
disinfectant solution or 70% ethanol and paper
towels for wiping down the bench surface
Notes
1. Cane sugar is purer than that produced from sugar beet, and is
therefore preferable here.
2. If you wish, eventually, to grow the plants in soil (see page 6),
add 0.65 mL of kinetin solution to half of the growth medium at
Step 6, and do not add any kinetin to the other half. If you wish,
you can colour-code the different media by adding 0.5 mL of
food colouring to each 250 mL batch of medium.
3. You must use Milton
tablets, not Milton
liquid, which is a totally
different chemical.
4. The vials that are supplied in the kit are manufactured under
sterile conditions and should be handled carefully (as if they
were Petri dishes) to ensure that they remain sterile.
Preparing the growth medium
How to prepare the growth medium
1. Measure 480 mL of distilled water into a litre beaker.
2. Add the sugar and stir until dissolved.
3. Add the MS agar powder, stirring constantly until
it is evenly dispersed in the liquid (the agar will not
dissolve fully until the mixture is heated).
4. Cover the beaker with clingflm, pierced several times
to allow steam to escape.
5. Heat the beaker in a microwave oven on high power
for 90 seconds, stir, re-cover and heat for another
2minutes (these times may need to be varied according
to the power rating of the oven. The times given are
for a 500 mL volume in an 800 W microwave oven).
IMPORTANT: You must watch the liquid constantly
as it is heating to ensure that it does not boil over.
6. When the agar has dissolved completely, and the
solution has boiled, take the beaker from the oven
and stir 1.25 mL of kinetin solution into the liquid
[but see note 2]. CAUTION: Any solution heated in a
microwave oven may become superheated and boil
vigorously and suddenly when moved or touched.
Take great care when handling the beaker and
ensure that you wear heat-proof gloves.
7. Allow the liquid to cool to about 50 C (that is, still liquid
but cool enough to reduce chlorine release in the next
step). It will take about 30 minutes for the agar to cool
to 50 C. The agar will begin to set at about 42 C.
8. In a well-ventilated area or a fume cupboard, add 20 mL
of the Milton
stock solution to the beaker of heated
and cooled growth medium [remember, if you are
preparing half of the medium without kinetin, you
will need to add 10 mL of the Milton
solution to each
250 mL volume of medium]. Stir well, taking care not
to breathe in any chlorine fumes.
9. Dispose of the remaining Milton
stock solution. N.B.
You must dilute the waste Milton
solution with
plenty of water as you dispose of it it is highly
toxic to aquatic organisms.
10. Immediately dispense the medium into the vials as
follows:
wipe down the work surface with Virkon
solution.
remove the lids from the vials and place them on the
sterilised work surface, inner side downwards.
dispense the medium into the vials (~10 mL per vial)
leave to cool with the lids on top of the vials but
slightly ajar to allow steam to escape (see picture).
when the medium has cooled and set (after about 60
minutes), press the caps down to seal the vials.
Storage
Once cool, the vials of growth medium can be stored for
about six weeks in a fridge at 35 C until required.
~ 10 mL mark
Note
Leave the lids slightly
ajar while the growth
medium is setting.
5 www.ncbe.reading.ac.uk Cloning caulifower
Virkon
Virkon
is a wide-spectrum surface disinfectant which
is suitable for use in school laboratories. It kills bacteria,
fungi and viruses.
Each pink tablet should be dissolved in 500 mL of tap
water before use. Wear a lab coat, suitable protective gloves
and tightly-ftting safety goggles when making up the
solution. Avoid splashes of concentrated solution by adding
the tablet to the water (NOT the water to the tablet).
Avoid raising an airborne dust: you should NOT grind the
tablets up to encourage them to dissolve (they will dissolve
quickly without being powdered).
Once made up, the solution should be used within 5 to 7
days (the pink colour fades when the solution is no longer
active). Virkon
solution at 1% concentration does not irritate
the skin and eyes, therefore eye protection and gloves
are not essential. Unlike chlorine-based disinfectants,
Virkon
does not have a toxic vapour and therefore extra
ventilation is not required.
Milton
Milton
is widely used for disinfecting babies bottles etc.
Its main active ingredient is sodium dichloroisocyanurate
(SDICN, also known as triclosene sodium). When used at the
dilution suggested on the packet, it is very safe to handle.
In the current practical activity, however, it is being used
at several times the recommended concentration.
Because of this, it is essential that eye protection is worn
by students undertaking this task. Splashes in the eye
should be rinsed with water and medical attention should
be sought immediately. Splashes on the skin or work
surfaces should be washed with water. Concentrated Milton
solution can bleach fabrics, so lab coats should be worn to
prevent accidental damage to clothing.
Safety
Microwave ovens
Because it contains a disinfectant, the growth medium
needs only to be microwaved and does not need to be
autoclaved.
There are three potential hazards when heating agar
media in a microwave oven. Firstly, the medium must
be placed in an open container so that any gas can escape
(heating media in a sealed container risks a dangerous
explosion).
Secondly, there is a risk that the viscous medium will
bubble over the top of the container. It should therefore
be watched constantly and the power should be stopped
(usually by opening the door of the microwave oven) at the
frst sign of overheating.
Thirdly, as with any liquid heated in a microwave
oven, there is a danger that the agar medium may become
superheated and froth up unexpectedly when the container
is taken from the oven. For this reason, heat-proof gloves
must be worn when handling the beakers of hot liquid.
Scalpels
Care should be taken when using scalpels.
Contaminated cultures
In the unlikely event of any plant tissue cultures becoming
contaminated with microorganisms, the vial(s) should not
be opened, but instead be placed in an autoclave bag and
sterilised by autoclaving.
WEAR EYE PROTECTION
WEAR A LAB COAT
As mentioned on page 4, teachers or technicians should
ensure that the Milton
stock solution is added to the
growth medium in a well-ventilated area (ideally in a
fume cupboard), as chlorine gas can be released when the
disinfect is added to hot, molten agar.
For educational reasons, you may wish to label the
Milton
solution given to students with the appropriate GHS
hazard symbols (the Exclamation mark and Environment
pictograms as shown in the Students guide).
Note
Autoclaving is necessary
only if the cultures are
contaminated.
6
Put on a lab coat.
Wear eye protection (safety glasses or goggles)
throughout the procedure.
Wash your hands with antibacterial soap.
Wipe down the work surface using disinfectant
solution and paper towels.
Set out the work area with everything you need:
plant growth medium containing kinetin, vitamins
and minerals; Milton
disinfectant solution; a piece
of caulifower curd; a scalpel; a surface for cutting on,
such as a tile or Petri dish; a pair of plastic forceps; a
waste container; a bijou bottle; and a marker pen.
Cut some small pieces about 5 mm x 5 mm x 5 mm
from the outer part of the caulifower curd. These will
be the explants from which caulifowers will be cloned.
Pour approximately 5 mL of Milton
disinfectant into
the bijou bottle.
Use the forceps to place the explants in the bijou
bottle of disinfectant.
Tightly cap the bijou bottle.
Place the forceps in the beaker of Milton
disinfectant.
Gently shake the bijou bottle so that the explants are
fully coated in disinfectant solution.
After 5 minutes, gently shake the explants again.
After 10 minutes, gently shake the explants again.
After 15 minutes, gently shake the explants again.
Take the cap off the bijou bottle and, holding the
bottle against the side of the waste container so that
the caulifower explants are not lost, carefully pour off
the disinfectant.
Use the now sterile forceps to place the explants on
the growth medium. Do this quickly, and do not place
the lid of the vial on the bench, to reduce the risk of
microbial contamination.
Gently push the explants slightly into the surface of the
growth medium so that they are held there.
Snap the lid fully onto the vial.
Label the vial with your initials and the date.
Place the vial in a warm, well-lit place.
The procedure in words
This can be used in conjunction with the Students guide
It can be used as a sequencing exercise, where students are given the sentences
below in a random order, and have to place them in the correct sequence with the
aid of the pictures in the Students guide.
www.ncbe.reading.ac.uk Cloning caulifower
7
Cloning caulifower
1. Describe how the steps used in this procedure prevent contamination.
2. Explain the purpose of the following ingredients in the plant growth
medium: sucrose; kinetin; Murashige and Skoog medium; agar; Milton
.
3. What are the advantages of using this method to cultivate new plants?
4. What are the disadvantages of using this method to cultivate new plants?
www.ncbe.reading.ac.uk Cloning caulifower
8
National Centre for Biotechnology Education, University of Reading, 2 Earley Gate, Reading RG6 6AU. United Kingdom
Tel: + 44 (0) 118 9873743. Fax: + 44 (0) 118 9750140. eMail: NCBE@reading.ac.uk Web: www.ncbe.reading.ac.uk
Copyright Dean Madden, 2014
Acknowledgements
The plant tissue culture technique used in this kit is based on
one developed by Conservation Biotechnology at the Royal
Botanic Gardens, Kew. The method is used by Kew scientists
in their conservation programmes.
Dan Jenkins at Science and Plants for Schools (SAPS) gave
advice and encouragement in the development of this kit.
This booklet and the associated Students guide were
written and illustrated by Dean Madden at the NCBE.
General reading
Plants from test tubes. An introduction to micropropagation by
Lydiane Kyte et al (2013) Timber Press. 4th Edtn. ISBN: 978
1604692068. An accessible, authoritative and comprehensive
guide to plant tissue culture, suitable for domestic and
educational practitioners.
Plant cell culture by H. A. Collin and S. Edwards (1998) Garland
Science. ISBN: 978 1872748474. This book is intended for
university students or researchers taking up plant tissue culture
for the frst time. It describes how to set up and run a tissue
culture facility, and includes an overview of many basic tissue
culture techniques.
Growing orchids from seed by Philip Seaton and Margaret
Ramsay (2005) Royal Botanic Gardens, Kew. ISBN: 978 184
246091 7. This book, written for the amateur enthusiast, describes
the cultivation of orchids by conventional tissue culture methods.
Plant tissue culture by Tony Storr (1985) Association for
Science Education, Hatfeld. ISBN: 0 86357 031 3. This
publication describes several educational protocols that are
suitable for schools and colleges, using conventional tissue
culture methods. It has been out of print for many years,
but a facsimile can be downloaded from the NCBEs website:
www.ncbe.reading.ac.uk/ptc
Further information
Web sites
Science and Plants for Schools (SAPS)
The SAPS website has numerous useful resources for
teaching plant science, including educational materials on
plant tissue culture: www.saps.org.uk
Plant TC Cases Wiki
This collection of open source practical protocols is aimed
at schools and amateur growers. The site includes a Microsoft
Excel
Workbook to convert molar quantities of plant growth
substances to masses to assist in the preparation of plant
growth media: www.planttccases.org
Royal Botanic Gardens, Kew
Conservation Biotechnology at Kew uses tissue culture
methods to save critically-endangered plants for
reintroduction into the wild:
www.kew.org/science/micropropagation.html
Writhlington School Orchid project
Pupils at Writhlington school use tissue culture techniques
in an ambitious and successful orchid cultivation project.
http://wsbeorchids.org.uk
Growing the plants on
After a few weeks, the caulifower callus tissue will have
developed small shoots and leaves, but no proper roots.
By transferring the tissue onto a new vial of MS growth
medium that contains no kinetin, however, the formation
of roots will be encouraged.
Once the small caulifower plantlets have produced
roots, they can be transplanted. The agar should be washed
off the roots, then the plantlets can be planted in fne seed
compost and grown into full-sized plants.
The leaves of tissue-cultured plants grown in humid
conditions lack a fully-developed waxy cuticle; their
stomata also respond too slowly to prevent desiccation of
the plant. It is therefore essential to keep the plants in a
humid environment at frst, by securing a clear plastic bag
over the plant pot.
After 10 days or so, the plantlets will have acclimatised
and can be grown as normal.
Timing (at 26 C)
Weeks State of plants
12 Curd enlarges and produces chlorophyll
23 Leaves produced
3 Shoots produced
36 Move tissue to kinetin-free medium to encourage roots
612 Rooted plantlets ready for transfer to soil
Você também pode gostar
- HydroponicsDocumento45 páginasHydroponicsRavindra Kumara100% (15)
- Chapter 1: The Scope of Food BiotechnologyDocumento43 páginasChapter 1: The Scope of Food BiotechnologyGG MMAinda não há avaliações
- Plate Count Agar (Standard Methods Agar) : Intended UseDocumento3 páginasPlate Count Agar (Standard Methods Agar) : Intended UsenuraenirianAinda não há avaliações
- Constructing The IncubatorDocumento4 páginasConstructing The IncubatorVeterinary Online InstituteAinda não há avaliações
- Siemens MV Gas Insulated Switch GearDocumento14 páginasSiemens MV Gas Insulated Switch GearSajesh Thykoodan T VAinda não há avaliações
- Space Analysis in Orthodontic: University of GlasgowDocumento16 páginasSpace Analysis in Orthodontic: University of GlasgowNizam Muhamad100% (1)
- Plant Tissue Culture: Biology-100 Section: 7 DR - Jebunnahar KhandakarDocumento17 páginasPlant Tissue Culture: Biology-100 Section: 7 DR - Jebunnahar Khandakarijaj imran100% (2)
- ItilDocumento11 páginasItilNarendar P100% (2)
- Zygomatic Complex FracturesDocumento128 páginasZygomatic Complex FracturesTarun KashyapAinda não há avaliações
- Lecture 03. Carbohydrate Metabolism. Common Pathway, Regulation and PathologyDocumento80 páginasLecture 03. Carbohydrate Metabolism. Common Pathway, Regulation and PathologyВіталій Михайлович НечипорукAinda não há avaliações
- Nicenstripy Gardening Risk AssessmentDocumento38 páginasNicenstripy Gardening Risk AssessmentVirta Nisa100% (1)
- Banana Tissue CultureDocumento20 páginasBanana Tissue Culturesupratimc61267Ainda não há avaliações
- Plant Tissue CultureDocumento37 páginasPlant Tissue Culturebits_who_am_iAinda não há avaliações
- 1 Fungi in BiotechnologyDocumento13 páginas1 Fungi in BiotechnologyThimani WijepalaAinda não há avaliações
- Microbial Growth PhaseDocumento2 páginasMicrobial Growth PhaseMahathir Mohmed100% (5)
- Chapter 6 - MicropropagatioonDocumento104 páginasChapter 6 - MicropropagatioonDương Hà Trúc Tâm100% (1)
- Psilocybin Mushrooms: A step-by-step guide to growing your own magic fungi at homeNo EverandPsilocybin Mushrooms: A step-by-step guide to growing your own magic fungi at homeAinda não há avaliações
- Panchakanya 1Documento9 páginasPanchakanya 1sriramana100% (1)
- FermenterDocumento19 páginasFermenterIshtiaque HossainAinda não há avaliações
- Dilution Control Systems Quat Sanitizer Test Strips 421200: Use To Test Multi-Clean MC-10 Sanitizer SolutionDocumento2 páginasDilution Control Systems Quat Sanitizer Test Strips 421200: Use To Test Multi-Clean MC-10 Sanitizer Solutionmanugeorge100% (1)
- Experiment 2Documento3 páginasExperiment 2nabilah0% (1)
- Screening of Industrially Imp MoDocumento7 páginasScreening of Industrially Imp MoramuAinda não há avaliações
- Capsule Staining Notes SimpleDocumento2 páginasCapsule Staining Notes Simplesairam diagnostics kosgiAinda não há avaliações
- Presence of Moisture The Growth of Microorganism (Mucor)Documento6 páginasPresence of Moisture The Growth of Microorganism (Mucor)nurliyana_samsudin100% (1)
- Microbiology 214 Skills Test Notes PDFDocumento21 páginasMicrobiology 214 Skills Test Notes PDFTayla Nel100% (1)
- Bacteria CountingDocumento30 páginasBacteria Countingnavidhah 22Ainda não há avaliações
- Mushroom CultivationDocumento9 páginasMushroom CultivationShalmali ChatterjeeAinda não há avaliações
- Methods of Protoplast FusionDocumento4 páginasMethods of Protoplast FusionMelissa A. BernardoAinda não há avaliações
- Streak PlatesDocumento0 páginaStreak PlatesJenry Reyes TrispaAinda não há avaliações
- How To Prepare Stock SolutionDocumento6 páginasHow To Prepare Stock SolutionaleenaAinda não há avaliações
- Purification Bacteria CultureDocumento5 páginasPurification Bacteria CultureWan Nabil0% (1)
- AQC-222, E-Practical Manual On Fish Nutrition and Feed Technology (Dr. M.L. Ojha, 08-02-2021)Documento33 páginasAQC-222, E-Practical Manual On Fish Nutrition and Feed Technology (Dr. M.L. Ojha, 08-02-2021)Amit SharmaAinda não há avaliações
- JetQuick Plasmid Miniprep - GenomedDocumento4 páginasJetQuick Plasmid Miniprep - GenomedCarlos de PazAinda não há avaliações
- PHD Thesis The Production of Bacteriocins From Lactic Acid Bacteria 2011Documento2 páginasPHD Thesis The Production of Bacteriocins From Lactic Acid Bacteria 2011Myrto-Panagiota ZacharofAinda não há avaliações
- Sterilization of MediumDocumento18 páginasSterilization of MediumGuleRia ShiVani100% (1)
- In Vitro Regeneration and Somatic Embryogenesis and Their Antimicrobial Studies in Selected Ornamental PlantsDocumento20 páginasIn Vitro Regeneration and Somatic Embryogenesis and Their Antimicrobial Studies in Selected Ornamental Plantsbakru248326Ainda não há avaliações
- Kirby Bauer Disc DiffusionDocumento9 páginasKirby Bauer Disc DiffusionSubhradeep GhoshAinda não há avaliações
- Suspension CultureDocumento12 páginasSuspension Culturemendal sciAinda não há avaliações
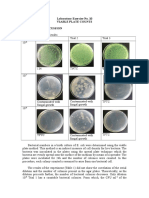
- Laboratory Exercise No. 10 Viable Plate Counts Results and DiscussionDocumento3 páginasLaboratory Exercise No. 10 Viable Plate Counts Results and Discussionvanessa olga100% (1)
- Biology 11 FederalDocumento426 páginasBiology 11 FederalAroona AkhaniAinda não há avaliações
- FDA - CFSAN BAM - Aerobic Plate Count PDFDocumento11 páginasFDA - CFSAN BAM - Aerobic Plate Count PDFsmatalab100% (1)
- Making High-Quality Cassava Flour: For More Information ContactDocumento4 páginasMaking High-Quality Cassava Flour: For More Information Contactvinay44Ainda não há avaliações
- HPFB Mfhpb-18 Sept 2020 enDocumento6 páginasHPFB Mfhpb-18 Sept 2020 enJamie BautistaAinda não há avaliações
- Food Analysis Lab ManualDocumento17 páginasFood Analysis Lab ManualChing YeeAinda não há avaliações
- SOP For TCPDocumento45 páginasSOP For TCPVarun Thakur50% (2)
- Vermicompost Harvesting - Amol HandoreDocumento46 páginasVermicompost Harvesting - Amol HandoreamolAinda não há avaliações
- Antibiotic FermentationDocumento12 páginasAntibiotic FermentationEli Leopoldo Geneston0% (1)
- ColiformDocumento1 páginaColiformIshtiaque IshtiAinda não há avaliações
- Lecture4 - Monolayer Cell CulturesDocumento34 páginasLecture4 - Monolayer Cell Culturesacswain100% (1)
- Sunflower 2Documento20 páginasSunflower 2Aastha DubeyAinda não há avaliações
- Standard Operating ProceduresDocumento3 páginasStandard Operating ProceduresPrasanna BabuAinda não há avaliações
- 206 Lab Ex - 6 - Bacterial CulturesDocumento10 páginas206 Lab Ex - 6 - Bacterial CulturesVia SongcalAinda não há avaliações
- Plant Tissue Culture Methodology: Steps and Role of HormonesDocumento51 páginasPlant Tissue Culture Methodology: Steps and Role of HormonesPURSUIT OF GROWTHAinda não há avaliações
- BIO461 Practical 2Documento7 páginasBIO461 Practical 2NURUL AIHAN AHMAD HILMIAinda não há avaliações
- ENVIRONMENT MANAGEMENT Waste Minimization StrategiesDocumento14 páginasENVIRONMENT MANAGEMENT Waste Minimization StrategiesAsmanieza IbrahimAinda não há avaliações
- Banana Tissue Culture TechniquesDocumento16 páginasBanana Tissue Culture Techniquestejaskamble45Ainda não há avaliações
- Microwave Pressure Cooker User Guide & RecipesDocumento46 páginasMicrowave Pressure Cooker User Guide & RecipesZomBillz HallAinda não há avaliações
- Determination of Zein ValueDocumento2 páginasDetermination of Zein ValuechintanictAinda não há avaliações
- Temporary Immersion Systems in Plant MicropropagationDocumento17 páginasTemporary Immersion Systems in Plant MicropropagationaguilaquecaeAinda não há avaliações
- Embryo Rescue TechniquesDocumento2 páginasEmbryo Rescue TechniquesUtuh KalambuaiAinda não há avaliações
- Sericogenic Resources - Muga & EriDocumento11 páginasSericogenic Resources - Muga & EriSamim SultanaAinda não há avaliações
- Micro Propagation of Banana Musa SP CV Agnishwar by in Vitro Shoot Tip CultureDocumento6 páginasMicro Propagation of Banana Musa SP CV Agnishwar by in Vitro Shoot Tip Cultureadididi33Ainda não há avaliações
- Determin of Aero Anaerobic Spore FormersDocumento6 páginasDetermin of Aero Anaerobic Spore FormersbhaleshAinda não há avaliações
- Practical 3 Aseptic TechniqueDocumento10 páginasPractical 3 Aseptic TechniqueEzety Termizi100% (1)
- Microorganism Preservation Methods GuideDocumento4 páginasMicroorganism Preservation Methods GuidePoojaswini ReddyAinda não há avaliações
- Plant Tissue Culture MediaDocumento34 páginasPlant Tissue Culture MediavijendAinda não há avaliações
- 36447-SAPS - Cauliflower Cloning - Student WorksheetDocumento2 páginas36447-SAPS - Cauliflower Cloning - Student Worksheetahdatu uliAinda não há avaliações
- Why There Is Something Rather Than Nothing ExplainedDocumento9 páginasWhy There Is Something Rather Than Nothing ExplainedDaniel JakobianAinda não há avaliações
- Kufri Himalini PDFDocumento6 páginasKufri Himalini PDFbrijmohansingh401124Ainda não há avaliações
- V S Naipaul PDFDocumento50 páginasV S Naipaul PDFbrijmohansingh401124Ainda não há avaliações
- Ghashiram Kotwal PDFDocumento77 páginasGhashiram Kotwal PDFbrijmohansingh401124100% (2)
- Blackberry Micropropagation PDFDocumento9 páginasBlackberry Micropropagation PDFbrijmohansingh4011240% (1)
- Now Grow Your Own Forest in Your Backyard PDFDocumento3 páginasNow Grow Your Own Forest in Your Backyard PDFbrijmohansingh401124Ainda não há avaliações
- Mass Multiplication in Hydroponic System PDFDocumento3 páginasMass Multiplication in Hydroponic System PDFbrijmohansingh401124Ainda não há avaliações
- Faiz Fehmi PDFDocumento4 páginasFaiz Fehmi PDFbrijmohansingh401124Ainda não há avaliações
- Ghashiram Kotwal PDFDocumento77 páginasGhashiram Kotwal PDFbrijmohansingh401124100% (2)
- 'Operation Bluestar' The Untold StoryDocumento42 páginas'Operation Bluestar' The Untold StorySikh TextsAinda não há avaliações
- Raising Native ForestsDocumento1 páginaRaising Native Forestsbrijmohansingh401124Ainda não há avaliações
- BrassTacks 014 Dance of The DevilDocumento102 páginasBrassTacks 014 Dance of The DevilabbasisaAinda não há avaliações
- From Farm To ForestDocumento15 páginasFrom Farm To ForestsillymindsAinda não há avaliações
- Now Grow Your Own Forest in Your Backyard PDFDocumento3 páginasNow Grow Your Own Forest in Your Backyard PDFbrijmohansingh401124Ainda não há avaliações
- Cloning PDFDocumento11 páginasCloning PDFbrijmohansingh401124Ainda não há avaliações
- 5 Tips To Improve Your Willpower PDFDocumento5 páginas5 Tips To Improve Your Willpower PDFbrijmohansingh401124Ainda não há avaliações
- Developing An in Vitro Method of Propagating Strelitziaceae PDFDocumento6 páginasDeveloping An in Vitro Method of Propagating Strelitziaceae PDFbrijmohansingh401124Ainda não há avaliações
- Culture Shock PDFDocumento6 páginasCulture Shock PDFbrijmohansingh401124Ainda não há avaliações
- Advantages of Clonal Propagation PDFDocumento14 páginasAdvantages of Clonal Propagation PDFbrijmohansingh401124Ainda não há avaliações
- Hindu Terrorism by ReshmaDocumento13 páginasHindu Terrorism by Reshmabrijmohansingh401124Ainda não há avaliações
- Tissue Culture of Coloured Common Callalily (Zantedeschla) - Abstract - Europe PubMed Central PDFDocumento2 páginasTissue Culture of Coloured Common Callalily (Zantedeschla) - Abstract - Europe PubMed Central PDFbrijmohansingh401124Ainda não há avaliações
- 1999 Kargil Conflict PDFDocumento5 páginas1999 Kargil Conflict PDFbrijmohansingh401124Ainda não há avaliações
- From Micropropagation To MicroponicsDocumento5 páginasFrom Micropropagation To Microponicsbrijmohansingh401124Ainda não há avaliações
- Warm Climate Production of Calla Lily PDFDocumento5 páginasWarm Climate Production of Calla Lily PDFbrijmohansingh401124Ainda não há avaliações
- Chola Dynasty PDFDocumento6 páginasChola Dynasty PDFbrijmohansingh401124100% (1)
- Cost Effective Protocol For Cymbidium Aloifolium Using Internode CultureDocumento8 páginasCost Effective Protocol For Cymbidium Aloifolium Using Internode Culturebrijmohansingh401124Ainda não há avaliações
- Micropropagationristolochia ManshuriensisDocumento36 páginasMicropropagationristolochia Manshuriensisbrijmohansingh401124Ainda não há avaliações
- Auditor General Insurance Regulation Dec 2011Documento23 páginasAuditor General Insurance Regulation Dec 2011Omar Ha-RedeyeAinda não há avaliações
- Owners Manual Water Mist PDFDocumento6 páginasOwners Manual Water Mist PDFZeeAinda não há avaliações
- Nitric OxideDocumento20 páginasNitric OxideGanesh V GaonkarAinda não há avaliações
- 2020-11 HBG Digital EditionDocumento116 páginas2020-11 HBG Digital EditionHawaii Beverage GuideAinda não há avaliações
- Gebauer 2012Documento26 páginasGebauer 2012Seán GallagherAinda não há avaliações
- Critical Criminal Justice IssuesDocumento132 páginasCritical Criminal Justice IssuesAnnamarella Amurao CardinezAinda não há avaliações
- Synthesis, Experimental and Theoretical Characterizations of A NewDocumento7 páginasSynthesis, Experimental and Theoretical Characterizations of A NewWail MadridAinda não há avaliações
- Lesson Plan 7 Tabata TrainingDocumento4 páginasLesson Plan 7 Tabata Trainingapi-392909015100% (1)
- Đề cương ôn tập tiếng anh 9Documento28 páginasĐề cương ôn tập tiếng anh 9Nguyễn HoaAinda não há avaliações
- Vturn-NP16 NP20Documento12 páginasVturn-NP16 NP20José Adalberto Caraballo Lorenzo0% (1)
- Bio-Tank Guidelines for Indian RailwayDocumento51 páginasBio-Tank Guidelines for Indian Railwayravi100% (2)
- M-LVDT: Microminiature Displacement SensorDocumento2 páginasM-LVDT: Microminiature Displacement Sensormahdi mohammadiAinda não há avaliações
- 2-D Motion Based Real Time Wireless Interaction System For Disabled PatientsDocumento5 páginas2-D Motion Based Real Time Wireless Interaction System For Disabled PatientsSantalum AlbumAinda não há avaliações
- Exercise 4 Summary - KEY PDFDocumento3 páginasExercise 4 Summary - KEY PDFFrida Olea100% (1)
- PDS in Paschim MidnaporeDocumento12 páginasPDS in Paschim Midnaporesupriyo9277Ainda não há avaliações
- Pet - WikipediaDocumento12 páginasPet - Wikipediabdalcin5512Ainda não há avaliações
- Marketing-Aspect 4Documento10 páginasMarketing-Aspect 4Leiren RingorAinda não há avaliações
- G10 Bio CellsDocumento6 páginasG10 Bio CellsswacaneAinda não há avaliações
- 07 Chapter2Documento16 páginas07 Chapter2Jigar JaniAinda não há avaliações
- 2.1. Pharmacological Therapeutics. 2.2. Basic Cardiac Life Support (BCLS) and Advanced Cardiac Life Support (ACLS) in Neonates and ChildDocumento3 páginas2.1. Pharmacological Therapeutics. 2.2. Basic Cardiac Life Support (BCLS) and Advanced Cardiac Life Support (ACLS) in Neonates and Childclint xavier odangoAinda não há avaliações
- Adolescent Development & Competency in Juvenile JusticeDocumento16 páginasAdolescent Development & Competency in Juvenile JusticeJudith KAinda não há avaliações
- TDS Versimax HD4 15W40Documento1 páginaTDS Versimax HD4 15W40Amaraa DAinda não há avaliações
- Intake Sheet SampleDocumento1 páginaIntake Sheet SampleRochelleAinda não há avaliações
- Clinical Case Report No 2Documento11 páginasClinical Case Report No 2ملک محمد صابرشہزاد50% (2)
- 05 AcknowledgementDocumento2 páginas05 AcknowledgementNishant KushwahaAinda não há avaliações