Escolar Documentos
Profissional Documentos
Cultura Documentos
Exp 2 Bubble Cap Distillation
Enviado por
Faris HamirDescrição original:
Direitos autorais
Formatos disponíveis
Compartilhar este documento
Compartilhar ou incorporar documento
Você considera este documento útil?
Este conteúdo é inapropriado?
Denunciar este documentoDireitos autorais:
Formatos disponíveis
Exp 2 Bubble Cap Distillation
Enviado por
Faris HamirDireitos autorais:
Formatos disponíveis
Mass Transfer I
CLB 20803
Experiment 2
BUBBLE CAP DISTILLATION PROCESS
1.0
OBJECTIVES
2.0
To operate vapor liquid separation process using a Bubble Cap Distillation
Unit.
To analyze the sample for the Top and Bottom Product by Refractometer to
obtain the Refractive Index in order to determine their respective composition.
To calculate the number of stages by using McCabe Thiele method
OVERVIEW
Distillation is a separation method in which mixture components in a liquid mixture are
separated based on their relative volatilities. The distillation column provides an environment
where the liquid and vapour phases can approach equilibrium within a column. In the packed
column there is high surface area for contact between the vapor and liquid, whereas the plate
column provides distinct stages at which equilibrium can be approached. Separation is achieved
by condensed vapor flowing as a liquid down the column theoretically achieving equilibrium with
the vapor flowing up the column. The distribution of components differs in each phase and results
in the separation. In the case of a binary mixture in batch distillation under total reflux, the vapor
condensing at the top of the distillation column will be rich in one of the components. Liquid
leaving the reboiler at the base of the column rich in the other component. As the system reaches
equilibrium the separation process reaches steady state for that apparatus and set of operating
conditions. This experiment is designed to study the distillation of a binary mixture of Ethanol
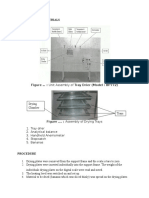
Water in a tray distillation column. Figure 1 is a sketch of a tray with bubble cap and Figure 2
shows the schematic diagram of the apparatus in this experiment.
Figure 1: Bubble cap tray
FG/exp2/July2012
Mass Transfer I
CLB 20803
Figure 2: Bubble Cap distillation column
3.0
Experimental Procedures
3.1
Chemicals and ancillary equipments required:
Chemicals Required.
Ethanol industrial grade.
Deionized-Water
Ancillary Equipments Required:
Two 2000ml Jug and two 25ml beakers.
Refractometer for measurement of sample concentrations.
Calibration Curve of ethanol-water mixtures.
3.2 Calibration curve of ethanol-water mixtures
1. Prepare a 5% of ethanol-water solution in a test tube.
2. Using a refractometer, obtain the refractive index reading for solution.
FG/exp2/July2012
Mass Transfer I
CLB 20803
3. Using increments of 5%, obtain refractive index readings for ethanol-water mixtures
up to 100%.
4. Plot a refractive index vs % of concentration graph.
3.3
General start-up procedures.
1. Prepare 30 litres of 10 % mixture of ethanol in water by mixing appropriate
quantities of the Industrial Grade of ethanol in distilled Water.
2. Ensure valves V1, RCV1, V3, V4, V5, V6, V7, V9 and RCV2 are closed.
3. Ensure valves V2, V8 and V10 are open.
4. Ensure that the Bottom Product sampling valve V5 and Top Product sampling valve
V4 is closed.
5. Slowly turn on the Cooling Water to the Condenser. Make sure water is indeed
flowing through it.
6. Check water flows to the drain from the Cooling Water outlets. Do not start the
experiment until the Cooling Water flow is visible in the outlets to the drain.
7. Set water pressure to 6 L/min.
8. Ensure that the Reboiler Vessel is already charged with the Ethanol Water mixture
through the Charge Port (Refer to the diagram).Fill the reboiler vessel with the
Ethanol-Water mixtures.
9. Check that the liquid level in Reboiler vessel is satisfactory (Refer to instructor /
technician).Otherwise top up with the Ethanol Water mixture through the Charge
Port (Refer to instructor / technician).
10. Ensure that the concentration of feed in the reboiler is correct and the liquid level is
at the vessel equator. (Refer to Lecturer / Technician).
11. The Reboiler Vessel should never be filled with liquid above the maximum level
indicated. The level should never recede below the minimum level while on
operation.
12. Obtain a sample of the ethanol-water mixture from the reboiler through valve V7 and
test its composition with the refractometer.
13. Turn on the main power control switch.
14. Turn on the power switch of the heater.
15. Switch on electrical supply (green heater on button).
16. Set heater controller HC4 to maximum setting (about 230V@ 30A).
17. Close the Reflux Adjustment Valve and valve below Product Cooler.
18. When distillate liquid is seen to flow through R1.A1, reduce the heat input and set
HC.4 to roughly 20A.
19. Allow a period of 15 minutes for the equipment to maintain thermal equilibrium with
surroundings.
20. The unit is now ready to be used for an experiment.
21. Switch on the heaters of the reboiler unit at the control panel and wait until the
mixture is seen boiling and vapor is seen to reach the top of the column
3.4
Experiment: Operation under reflux ratio condition
1. Ensure that the valves for reflux ratio RCV adjustment are opened.
2. Adjust both valve for Reflux Ratio RCV1 and valve below product cooler V2 so that
both readings on R1 and R2 provide a reflux ratio of 1.0 for the operation.
FG/exp2/July2012
Mass Transfer I
CLB 20803
3. Wait for about 15 minutes until the flow rate shown by R1 and the temperature
readings at the top and bottom of the columns are steady. (Refer to instructor /
technician).
4. Collect samples from the Reboiler and the overhead from the Sampling Port for every
5 minutes.
5. Observe the temperature of the reboiler T14.If the temperature is already 90 OC,
reduce the current of the reboiler to between 10A to 15A.
6. Observe the flow rate at R1. Adjust the valve for Reflux Ratio adjustment to ensure
that the Reflux Ratio is maintained at 1.0.
7. The concentration of the samples drawn is measure using the refractive index method
8. Record the reading of temperature for every 5 minutes
9. Readings are recorded in the provided table of results.
10. Then, the experiment is repeated with Reflux ration of 1.5 and 2.0.
3.5
General shut down procedures.
1.
2.
3.
4.
5.
Adjust heater controller HC.4 to minimum setting.
Switch off electrical supply (red heater off button).
Turn off the power switch of the reboiler.
Turn off the Main power control switch.
Do not drain the hot liquid from the Reboiler. If necessary, the liquid within the
system could be drained only when the liquid is already cooled.
6. Allow the cooling water to run for some time.
4.0
ANALYSIS AND DISCUSSION
Discuss all your results. The questions below only serve as a guideline. Your discussion
should not only limit to these questions.
1. Use the table for data collection in Appendix B.
2. Plot Calibration Curve for ethanol-water mixtures (Refractive index vs %
concentration.
3. Referring to calibration curve, estimate the mole fraction ethanol of the Top and
Bottom product samples for each reflux.
4. Plot the Equilibrium Curve for ethanol-water mixture. The data is given in Appendix
A.
5. Calculate the complete overall and component mass balance of the process.
6. By using McCabe Thiele Method, construct the graph to determine the number of
stages that occur in the processes for each reflux.
7. Discuss the results obtained and effect of reflux ratio for ethanol-water mixture
separation
8. Discuss any possible errors in the experiment and state any recommendation to
improve the process.
5.0
REFERENCES
1. Treybal, R.E., Mass Transfer Operations, 3rd ed., Mc-Graw-Hill, 1981
2. McCabe & Smith, Unit Operations of Chemical Engineering, 5th ed, Mc-Graw-Hill,
1993
3. Geankoplis,C. J. Z., Mass Transport Phenomena, 4th Ed., Rine Hart Winston, New
York.
FG/exp2/July2012
Mass Transfer I
CLB 20803
4. Coulson & Richardson, Chemical Engineering. Vol. 2 Pergamon Press, Oxford.
APPENDIX A
Equilibrium Data for Ethanol-Water Mixtures
Mole fraction of ethanol in liquid, x
0.00
0.05
0.10
0.40
0.60
0.80
0.94
0.90
0.94
0.96
0.98
Mole fraction of ethanol in vapor, y
0.00
0.38
0.53
0.75
0.79
0.86
0.94
0.91
0.94
0.96
0.99
FG/exp2/July2012
Mass Transfer I
CLB 20803
APPENDIX B
Table of Results
Reflux ratio
= 1.0
Rotameter reading R1 (L/hr)
= __________________
Rotameter reading R2 (L/hr)
= __________________
Temperature T4 (oC)
= __________________
Temperature T2 (oC)
= __________________
Time, t
(min)
TOP PRODUCT
Refractive index
Mole fraction
(RI)
BOTTOM PRODUCT
Refractive index
Mole fraction
(RI)
0
5
10
15
20
25
30
Reflux ratio
= 1.5
Rotameter reading R1 (L/hr)
= __________________
Rotameter reading R2 (L/hr)
= __________________
Temperature T4 (oC)
= __________________
Temperature T2 (oC)
= __________________
Time, t
(min)
TOP PRODUCT
Refractive index
Mole fraction
(RI)
BOTTOM PRODUCT
Refractive index
Mole fraction
(RI)
0
5
10
15
FG/exp2/July2012
Mass Transfer I
CLB 20803
20
25
30
Reflux ratio
= 2.0
Rotameter reading R1 (L/hr)
= __________________
Rotameter reading R2 (L/hr)
= __________________
Temperature T4 (oC)
= __________________
Temperature T2 (oC)
= __________________
Time, t
(min)
TOP PRODUCT
Refractive index
Mole fraction
(RI)
BOTTOM PRODUCT
Refractive index
Mole fraction
(RI)
0
5
10
15
20
25
30
FG/exp2/July2012
Você também pode gostar
- Exp - 2 Bubble Cap Distillation ColumnDocumento13 páginasExp - 2 Bubble Cap Distillation ColumnAdawiyah Al-jufri100% (1)
- Exp - 4 - Pulsed Column Liquid - Liquid ExtractionDocumento14 páginasExp - 4 - Pulsed Column Liquid - Liquid ExtractionAdawiyah Al-jufri0% (3)
- Experiment 2 - Study of Packed Column DistillationDocumento7 páginasExperiment 2 - Study of Packed Column DistillationAdawiyah Az-zahra100% (1)
- Bubble Cap Distillation ColumnDocumento3 páginasBubble Cap Distillation Columnnhalieza1067Ainda não há avaliações
- Apparatus, Procedure, Recommendation Tray DryerDocumento4 páginasApparatus, Procedure, Recommendation Tray DryerillyzlAinda não há avaliações
- Sieve Plate Distillation ColumnDocumento9 páginasSieve Plate Distillation ColumnAshish VermaAinda não há avaliações
- Lab Report - Distillation of Bubble CapDocumento21 páginasLab Report - Distillation of Bubble Capratish100% (1)
- Lab Report 5Documento12 páginasLab Report 5Norhanisah Zamri Rcsu100% (1)
- Lab Report Aspen Hysis UiTMDocumento12 páginasLab Report Aspen Hysis UiTMAhmad SiddiqAinda não há avaliações
- Experiment 1B - Tubular ReactorDocumento14 páginasExperiment 1B - Tubular ReactorNajmul Puda PappadamAinda não há avaliações
- CSTRDocumento20 páginasCSTRSharing Caring100% (1)
- CKB 20104 Reaction Engineering UniKL MICET Experiment 1a: The Batch Saponification of Ethyl Acetate Full Lab ReportDocumento11 páginasCKB 20104 Reaction Engineering UniKL MICET Experiment 1a: The Batch Saponification of Ethyl Acetate Full Lab ReportSiti Hajar Mohamed82% (11)
- 123 Reynolds ApparatusDocumento5 páginas123 Reynolds ApparatusKonem SolutionsAinda não há avaliações
- Isothermal Batch ReactorDocumento10 páginasIsothermal Batch ReactorSaswiny Ritchie0% (2)
- Lab Report On Fitting LossDocumento13 páginasLab Report On Fitting LossJyiou Yimushi100% (1)
- Lab10 CompleteDocumento22 páginasLab10 CompleteMastura Ahmad Termizi100% (1)
- Absorption in Packed Bed Lab ManualDocumento5 páginasAbsorption in Packed Bed Lab ManualAshish Verma100% (1)
- Batch Distillation LabDocumento18 páginasBatch Distillation LabBarryAinda não há avaliações
- Exp 3-Vapor-Liquid Equilibrium UnitDocumento18 páginasExp 3-Vapor-Liquid Equilibrium UnitKhairulAzwanizam100% (2)
- Continuous Stirred Tank Reactor: CHEN-410 Unit Operation LabDocumento34 páginasContinuous Stirred Tank Reactor: CHEN-410 Unit Operation LabMohamad Abou DaherAinda não há avaliações
- PDFDocumento88 páginasPDFMuralidharanAinda não há avaliações
- Climbing FilmDocumento34 páginasClimbing FilmTunji Aminu100% (1)
- Investigation of Liquid-Solid and Gas-Solid Fluidized BedDocumento18 páginasInvestigation of Liquid-Solid and Gas-Solid Fluidized Bedmahbub1332100% (1)
- Experiment 2 - Forced Draft Cooling TowerDocumento14 páginasExperiment 2 - Forced Draft Cooling TowerSonia YuAinda não há avaliações
- Exp 4 Batch Evaporative Crystallization PDFDocumento9 páginasExp 4 Batch Evaporative Crystallization PDFmirza farhanAinda não há avaliações
- Lab Report CSTR RTDDocumento13 páginasLab Report CSTR RTDNurul IzzahAinda não há avaliações
- VI Sem Mass Transfer Lab ManualDocumento53 páginasVI Sem Mass Transfer Lab Manualoctoviancletus80% (10)
- Lab Report 4 (Physical Chemistry)Documento10 páginasLab Report 4 (Physical Chemistry)Hanif YusofAinda não há avaliações
- PK - FKK.PPM - Manual Makmal Che565: Chemical Engineering Laboratory IiiDocumento21 páginasPK - FKK.PPM - Manual Makmal Che565: Chemical Engineering Laboratory Iiibedirtupak92% (12)
- LAB LLE FULL REPORT ZkinDocumento26 páginasLAB LLE FULL REPORT ZkinAmir Al-AimanAinda não há avaliações
- Cre 1 IntroductionDocumento4 páginasCre 1 IntroductionEvangeline LauAinda não há avaliações
- Lab Report Gas AbsorptionDocumento14 páginasLab Report Gas AbsorptionM Asrar SidonAinda não há avaliações
- CKB 20104 Reaction Engineering UniKL MICET Experiment 2a Effect of RTD On The Reaction in CSTR Full Lab ReportDocumento29 páginasCKB 20104 Reaction Engineering UniKL MICET Experiment 2a Effect of RTD On The Reaction in CSTR Full Lab ReportSiti Hajar Mohamed100% (6)
- Level Control (Our Report)Documento31 páginasLevel Control (Our Report)Brishti Anwar100% (1)
- Climbing Film EvaporatorDocumento8 páginasClimbing Film Evaporatorsaz140% (1)
- Experiment 7 - Batch ReactorDocumento5 páginasExperiment 7 - Batch Reactormythili83% (6)
- Isothermal Semi-Batch Reactor PPT RJC SirDocumento16 páginasIsothermal Semi-Batch Reactor PPT RJC Sirsdjdsf100% (1)
- Exp - S1A - Solid in Air DiffusionDocumento7 páginasExp - S1A - Solid in Air DiffusionAnuj SrivastavaAinda não há avaliações
- Vapour Liquid Equilibrium ExpDocumento5 páginasVapour Liquid Equilibrium ExpAakash Sharma100% (1)
- CLB11003 - Exp 4Documento6 páginasCLB11003 - Exp 4Nur DiyanahAinda não há avaliações
- Gas AbsorptionDocumento24 páginasGas AbsorptionShalini Krishnan100% (1)
- Calibration of Peristaltic Pumps - Lab 1Documento12 páginasCalibration of Peristaltic Pumps - Lab 1mmccomas08100% (2)
- Batch Reactor Exp.Documento21 páginasBatch Reactor Exp.Laila Al-shafieAinda não há avaliações
- Osbourne ReynoldDocumento13 páginasOsbourne ReynoldN Afiqah Razak0% (1)
- Lab 10-Batch ReactorDocumento22 páginasLab 10-Batch Reactorniraj_bairagiAinda não há avaliações
- Vle UnitDocumento26 páginasVle UnitAhmad Ifwat50% (2)
- Separation Processes Lab ReportDocumento15 páginasSeparation Processes Lab ReportArslanQureshi0% (1)
- Lab ManualDocumento24 páginasLab ManualAasia FarrukhAinda não há avaliações
- Manual For Wetted Wall ColumnDocumento8 páginasManual For Wetted Wall ColumnBalRam DhimanAinda não há avaliações
- Energy BalanceDocumento16 páginasEnergy BalancewizlanAinda não há avaliações
- Lab Report (Distillation)Documento10 páginasLab Report (Distillation)najihahAinda não há avaliações
- Exp 1 Packed Column DistillationDocumento12 páginasExp 1 Packed Column DistillationLuqman WasirAinda não há avaliações
- Continuous Distillation UnitDocumento3 páginasContinuous Distillation UnitHusna Hafiza Bt. R.AzamiAinda não há avaliações
- Report Distillation ColumnDocumento20 páginasReport Distillation ColumnAzam Najmi33% (3)
- Batch Reactive DistillationDocumento7 páginasBatch Reactive DistillationChalmer BelaroAinda não há avaliações
- Packed Column Distillation ProcessDocumento8 páginasPacked Column Distillation ProcessKhairin RenAinda não há avaliações
- Binary Distillation in A Bubble Cap Tray ColumnDocumento3 páginasBinary Distillation in A Bubble Cap Tray ColumnShelly Theresse Catian NovoAinda não há avaliações
- EXP 2B-Packed Column Distillation-Jan 2015Documento5 páginasEXP 2B-Packed Column Distillation-Jan 2015Siti KhalidaAinda não há avaliações
- Fractional Distillation Experiment - ChEDocumento6 páginasFractional Distillation Experiment - ChEmutencoAinda não há avaliações
- Process Steam Systems: A Practical Guide for Operators, Maintainers, and DesignersNo EverandProcess Steam Systems: A Practical Guide for Operators, Maintainers, and DesignersAinda não há avaliações
- Hydrogen Design 2520of 2520equipmentsDocumento37 páginasHydrogen Design 2520of 2520equipmentsjavilapiedraAinda não há avaliações
- Assignment 1 (10%) Instructions:: 1. in This Assignment, It Is Allowed To Work in Groups of at Most Five (5) Members. OneDocumento2 páginasAssignment 1 (10%) Instructions:: 1. in This Assignment, It Is Allowed To Work in Groups of at Most Five (5) Members. OneFaris HamirAinda não há avaliações
- Experiment 4-Oil Water SeparationDocumento10 páginasExperiment 4-Oil Water SeparationFaris Hamir67% (3)
- Obj. Activities 2010 2011 2012 2013 Sem 1 Sem 2 Sem 3 Sem 4 Sem 5 Sem 6Documento1 páginaObj. Activities 2010 2011 2012 2013 Sem 1 Sem 2 Sem 3 Sem 4 Sem 5 Sem 6Faris HamirAinda não há avaliações
- Propulsion of Sea ShipDocumento15 páginasPropulsion of Sea ShipFaris Hamir100% (1)
- Tutorial Exp 4Documento4 páginasTutorial Exp 4Faris Hamir100% (4)
- Global WarmDocumento13 páginasGlobal WarmFaris HamirAinda não há avaliações
- The Realization of Global WarmingDocumento5 páginasThe Realization of Global WarmingFaris HamirAinda não há avaliações
- Exp 4 Centrifugal CompressorDocumento11 páginasExp 4 Centrifugal CompressorFaris HamirAinda não há avaliações
- The Realization of Global WarmingDocumento5 páginasThe Realization of Global WarmingFaris HamirAinda não há avaliações
- Nose Cone & Fin OptimizationDocumento48 páginasNose Cone & Fin Optimizationnhung_33Ainda não há avaliações
- Vessel CalculationDocumento106 páginasVessel CalculationBhadresh PatelAinda não há avaliações
- SM Assignments (2020-21)Documento11 páginasSM Assignments (2020-21)4035VAID P KULKARNI SEMECHAAinda não há avaliações
- Chap 2Documento47 páginasChap 2Amanu WorkuAinda não há avaliações
- Unit 9.2 - Temp and Heat - Giancoli ProblemsDocumento2 páginasUnit 9.2 - Temp and Heat - Giancoli ProblemslucasAinda não há avaliações
- Design For Duct-1: Straight Duct Friction Loss (How To Decide " Mmaq/M", "Pa/M")Documento3 páginasDesign For Duct-1: Straight Duct Friction Loss (How To Decide " Mmaq/M", "Pa/M")Azrinshah Abu BakarAinda não há avaliações
- CHAPTER 3 2 Micro Process in A Fluid Motion of Particle in A FluidDocumento21 páginasCHAPTER 3 2 Micro Process in A Fluid Motion of Particle in A FluidmymamforeverAinda não há avaliações
- Basic Principles of Food Process EngineeringDocumento12 páginasBasic Principles of Food Process EngineeringShalehin IslamAinda não há avaliações
- Quantum Mechanical Carnot Engine: Journal of Physics A General Physics July 2000Documento11 páginasQuantum Mechanical Carnot Engine: Journal of Physics A General Physics July 2000Kin MattAinda não há avaliações
- M8 - Vapor Absorption RefrDocumento29 páginasM8 - Vapor Absorption RefrAbraham KhaleedAinda não há avaliações
- Entropy: Energy, Entropy and Exergy Concepts and Their Roles in Thermal EngineeringDocumento34 páginasEntropy: Energy, Entropy and Exergy Concepts and Their Roles in Thermal EngineeringHisham TariqAinda não há avaliações
- Combustion - Part 1 - WatermarkedDocumento30 páginasCombustion - Part 1 - Watermarked84105851Ainda não há avaliações
- Epanet ManualDocumento11 páginasEpanet ManualWilson UlcoAinda não há avaliações
- Environmental Fluid Mechanics: Benoit Cushman-RoisinDocumento10 páginasEnvironmental Fluid Mechanics: Benoit Cushman-Roisinpawn1000kAinda não há avaliações
- Equation of State of Ideal Gases With Cobra 4Documento10 páginasEquation of State of Ideal Gases With Cobra 4DanielLugoAinda não há avaliações
- Lecture 10 - Design of Concrete BeamsDocumento29 páginasLecture 10 - Design of Concrete BeamsKhaled AlregebAinda não há avaliações
- Motalab Anand CyclingDocumento10 páginasMotalab Anand CyclingAminul HoqueAinda não há avaliações
- CVD NotesDocumento5 páginasCVD NotesAyesha TariqAinda não há avaliações
- Introduction and Properties of Pure SubstancesDocumento63 páginasIntroduction and Properties of Pure SubstancesTushyAinda não há avaliações
- Review of Wind TunnelsDocumento15 páginasReview of Wind TunnelsFahad AliAinda não há avaliações
- V-Cone Flowmeter Solves Compressor Control Problems For The Oil & Gas IndustryDocumento2 páginasV-Cone Flowmeter Solves Compressor Control Problems For The Oil & Gas IndustryL. LAAinda não há avaliações
- Design of BeamsDocumento14 páginasDesign of BeamsNoor MohdAinda não há avaliações
- Experiment No. 9: Chemical EquilibriumDocumento25 páginasExperiment No. 9: Chemical Equilibriumtwinkledreampoppies100% (22)
- Lecture 5 Smoke Control SystemsDocumento108 páginasLecture 5 Smoke Control SystemsMiu For LeungAinda não há avaliações
- ME 205-Mechanics of Materials TestDocumento2 páginasME 205-Mechanics of Materials TestDaniel Deng KuolAinda não há avaliações
- Solved Problems in Heat TransferDocumento16 páginasSolved Problems in Heat TransferAngelica Joyce Benito100% (3)
- THAIOIL - DOUBLE SHEAR CLEAT ANGLE - BOLTED - 005 - Rev ADocumento11 páginasTHAIOIL - DOUBLE SHEAR CLEAT ANGLE - BOLTED - 005 - Rev ACivil StructureAinda não há avaliações
- Wang, L. - Fluid-structure-interaction-modelling-of-horizontalaxis-wind-turbine-blades-based-on-CFD-and-FEAArticle - 2016Documento15 páginasWang, L. - Fluid-structure-interaction-modelling-of-horizontalaxis-wind-turbine-blades-based-on-CFD-and-FEAArticle - 2016Mayra ZezattiAinda não há avaliações
- (Welty, Rorrer, Foster, 6th Edition International Student Version 14.7)Documento5 páginas(Welty, Rorrer, Foster, 6th Edition International Student Version 14.7)Khuram MaqsoodAinda não há avaliações
- Answer PHYSIC STPM Trial Sem 1 2013Documento6 páginasAnswer PHYSIC STPM Trial Sem 1 2013Zuraini ArshadAinda não há avaliações