Escolar Documentos
Profissional Documentos
Cultura Documentos
Atq
Enviado por
Elah Palaganas0 notas0% acharam este documento útil (0 voto)
618 visualizações3 páginasThis document provides answers to questions about carbohydrate chemistry. It defines the osazones produced from glucose, fructose, and maltose. It ranks sugars' reactivity with Benedict's reagent from most to least reactive. It explains the differences between Benedict's and Barfoed's tests for reducing sugars. It states that Seliwanoff's test cannot differentiate sucrose from fructose. It shows the hydrolysis products of sucrose and starch as glucose and fructose or glucose, respectively. Finally, it describes molecularly how starch traps iodine through helical amylose structures, reducing iodine poisoning.
Descrição original:
chem 31
Título original
atq
Direitos autorais
© © All Rights Reserved
Formatos disponíveis
DOCX, PDF, TXT ou leia online no Scribd
Compartilhar este documento
Compartilhar ou incorporar documento
Você considera este documento útil?
Este conteúdo é inapropriado?
Denunciar este documentoThis document provides answers to questions about carbohydrate chemistry. It defines the osazones produced from glucose, fructose, and maltose. It ranks sugars' reactivity with Benedict's reagent from most to least reactive. It explains the differences between Benedict's and Barfoed's tests for reducing sugars. It states that Seliwanoff's test cannot differentiate sucrose from fructose. It shows the hydrolysis products of sucrose and starch as glucose and fructose or glucose, respectively. Finally, it describes molecularly how starch traps iodine through helical amylose structures, reducing iodine poisoning.
Direitos autorais:
© All Rights Reserved
Formatos disponíveis
Baixe no formato DOCX, PDF, TXT ou leia online no Scribd
0 notas0% acharam este documento útil (0 voto)
618 visualizações3 páginasAtq
Enviado por
Elah PalaganasThis document provides answers to questions about carbohydrate chemistry. It defines the osazones produced from glucose, fructose, and maltose. It ranks sugars' reactivity with Benedict's reagent from most to least reactive. It explains the differences between Benedict's and Barfoed's tests for reducing sugars. It states that Seliwanoff's test cannot differentiate sucrose from fructose. It shows the hydrolysis products of sucrose and starch as glucose and fructose or glucose, respectively. Finally, it describes molecularly how starch traps iodine through helical amylose structures, reducing iodine poisoning.
Direitos autorais:
© All Rights Reserved
Formatos disponíveis
Baixe no formato DOCX, PDF, TXT ou leia online no Scribd
Você está na página 1de 3
QUESTIONS:
1. Draw the structures of the osazones produced in the experiment.
2. Theoretically, arrange the following sugars according to their reactivity
with the Benedicts reagent.
Glucose, Fructose, Maltose, Sucrose, Starch
3. What is the difference between Benedicts Test and Barfoeds test?
4. Can the Seliwanoffs test be used to differentiate sucrose from fructose?
Why?
5. Draw the hydrolysis products of sucrose and starch.
6. Give a molecular explanation for the role of starch as an antidote for the
iodine poisoning.
ANSWERS TO QUESTIONS:
1. A. Glucose produces glucosazone
B. Fructose produces fructosazone
C. Maltose produces maltosazone
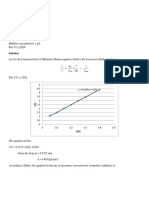
2. Glucose > Fructose > Maltose > Starch > Sucrose
3. Benedicts test is used to determine the presence of reducing sugars,
including both monosaccharides and disaccharides, lactose and maltose.
Barfoeds test, on the other hand, is also used in detecting the presence of
reducing sugars but specifically for monosaccharides. Barfoeds reagent is
generally mild, having a low pH, so reducing monosaccharides react quickly
in Barfoeds reagent but disaccharides react very slowly or not at all. Simply
put, compared to Benedicts test, Barfoeds test is the one to be used to
distinguish between a monosaccharide and disaccharide.
4. No. Seliwanoffs test is used to differentiate an aldohexose from a
ketohexose. Fructose is a ketohexose. Sucrose on the other hand is a
disaccharide made up of fructose and glucose. Since both molecules contain
the ketone functionalities, it would be hard to differentiate the two
compounds using the Seliwanoffs test.
5. A. Hydrolysis products of sucrose: Glucose and Fructose
Glucose
Fructose
B. Hydrolysis product of starch: Glucose
Glucose
6. Starch is composed of a branched amylopectin and an unbranched, linear
amylose that consists of only -1,4 linked glucoses. The linear amylase, when
reacted with iodine, forms a blue complex (amylose-iodine) wherein the
amylose forms a helical structure. The cavity formed by the starch helices
acts as a cage/space that captures and sequesters the iodine. Thus, the
amylose in starch removes the presence of excess iodine reducing its toxicity.
Você também pode gostar
- Don't Trust MeDocumento2 páginasDon't Trust Memark pascuaAinda não há avaliações
- David FR16Documento7 páginasDavid FR16FrettyDavidAinda não há avaliações
- Postalb 16-Synthesis of Sudan 1Documento2 páginasPostalb 16-Synthesis of Sudan 1edcalumpitAinda não há avaliações
- Chem31.1 ATQ12 Santos PDFDocumento3 páginasChem31.1 ATQ12 Santos PDFClaire SantosAinda não há avaliações
- Chem 31.1 - FR2 CastroDocumento4 páginasChem 31.1 - FR2 CastroMatthew Rei De LeonAinda não há avaliações
- Postlab 3Documento6 páginasPostlab 3Ice cream dudeAinda não há avaliações
- Aliphatic and Aromatic HydrocarbonsDocumento1 páginaAliphatic and Aromatic HydrocarbonsJulian Bato0% (3)
- Chem 31.1 Exp 10Documento2 páginasChem 31.1 Exp 10qwertyuasiop100% (1)
- Relative Rates of Electrophilic Aromatic SubstitutionDocumento2 páginasRelative Rates of Electrophilic Aromatic SubstitutionAnonymous GO6JVW9Wud100% (4)
- Chem 31.1 ATQ Experiment 1Documento4 páginasChem 31.1 ATQ Experiment 1Ying YangAinda não há avaliações
- Quantitative Analysis of Acetylsalicylic Acid in Aspirin Tablet by Back-TitrationDocumento2 páginasQuantitative Analysis of Acetylsalicylic Acid in Aspirin Tablet by Back-TitrationZyrle Nikko UchidaAinda não há avaliações
- Chem 31.1 Lab ReviewerDocumento3 páginasChem 31.1 Lab ReviewerJamie Joice NocheAinda não há avaliações
- Postlab8 9Documento3 páginasPostlab8 9Niño Sandro Jocson MercadoAinda não há avaliações
- Nucleophlic Acyl Substitution - The Synthesis of Ethyl ButanoateDocumento5 páginasNucleophlic Acyl Substitution - The Synthesis of Ethyl ButanoateRuther Cabral67% (3)
- Nucleophilic Acyl Substitution Chem 31Documento5 páginasNucleophilic Acyl Substitution Chem 31Frances Abegail QuezonAinda não há avaliações
- Fr3 Synthesis of 1 Phenylazo 2 NaphtholDocumento4 páginasFr3 Synthesis of 1 Phenylazo 2 NaphtholRon Andrei Soriano0% (1)
- Chem 31.1 Exp 2 3 4Documento2 páginasChem 31.1 Exp 2 3 4qwertyuasiop100% (1)
- Atq E4Documento3 páginasAtq E4BuiHopeAinda não há avaliações
- Last FR Chem 31Documento4 páginasLast FR Chem 31James Soriano100% (1)
- Chem 31.1 Expt 8 Lab ReportDocumento30 páginasChem 31.1 Expt 8 Lab ReportJohn Christian LopezAinda não há avaliações
- Exp 7 ATQDocumento3 páginasExp 7 ATQDean Dela CruzAinda não há avaliações
- Experiment 4 ATQ Chem 31.1Documento2 páginasExperiment 4 ATQ Chem 31.1Elah Palaganas100% (1)
- Exp 16-FR 3 SudanDocumento14 páginasExp 16-FR 3 Sudanherbie_wanAinda não há avaliações
- Nucleophilic Acyl SubstitutionDocumento4 páginasNucleophilic Acyl SubstitutionseryuyuyuAinda não há avaliações
- Preparation and Purification of An Alkyl Halide FRDocumento6 páginasPreparation and Purification of An Alkyl Halide FRCamille GrefaldiaAinda não há avaliações
- Formal Report: Nucleophilic Acyl Subtitution: The Synthesis of EthersDocumento3 páginasFormal Report: Nucleophilic Acyl Subtitution: The Synthesis of EthersJuris Marie G. GarciaAinda não há avaliações
- Experiment 9 Relative Rates of Electrophilic Aromatic Substitution QuestionsDocumento1 páginaExperiment 9 Relative Rates of Electrophilic Aromatic Substitution QuestionsElah PalaganasAinda não há avaliações
- Hinsberg's Method For Characterizing Primary, Secondary and Tertiary AminesDocumento2 páginasHinsberg's Method For Characterizing Primary, Secondary and Tertiary AminesGaby de GuzmanAinda não há avaliações
- Department of Chemical Engineering, University of The Philippines, Diliman, Quezon CityDocumento2 páginasDepartment of Chemical Engineering, University of The Philippines, Diliman, Quezon CityElaine Nicole CanebaAinda não há avaliações
- Synthesis of 1-Phenylazo-2-NaphtholDocumento4 páginasSynthesis of 1-Phenylazo-2-NaphtholEdric RaguindinAinda não há avaliações
- Solubility of Organic Compounds: Answers To QuestionsDocumento3 páginasSolubility of Organic Compounds: Answers To QuestionsMatthew Rei De LeonAinda não há avaliações
- Aldehydes and Ketones: Answers To QuestionsDocumento3 páginasAldehydes and Ketones: Answers To Questionsmark pascuaAinda não há avaliações
- Department of Mining, Metallurgical and Materials Engineering, University of The Philippines, Diliman, Quezon CityDocumento4 páginasDepartment of Mining, Metallurgical and Materials Engineering, University of The Philippines, Diliman, Quezon CityRochelle ManchamAinda não há avaliações
- Exp 13 - FR 2 Nucleuphilic Acyl Substitution, The Synthesis of EstersDocumento12 páginasExp 13 - FR 2 Nucleuphilic Acyl Substitution, The Synthesis of Estersherbie_wanAinda não há avaliações
- ATQ-1 Solubility of Organic CompoundsDocumento2 páginasATQ-1 Solubility of Organic CompoundsAnne Raever BenavidezAinda não há avaliações
- Solubility Behavior of Organic CompoundsDocumento2 páginasSolubility Behavior of Organic CompoundsIlac CapangpanganAinda não há avaliações
- Chem 31.1 Expt 2Documento1 páginaChem 31.1 Expt 2Kelvin LabarezAinda não há avaliações
- Aldehydes and KetonesDocumento1 páginaAldehydes and KetonesThea Mae Dino0% (1)
- Experiment 9 Relative Rates of Electrophilic Aromatic Substitution QuestionsDocumento2 páginasExperiment 9 Relative Rates of Electrophilic Aromatic Substitution QuestionsElah PalaganasAinda não há avaliações
- Expt. #14 - ATQDocumento1 páginaExpt. #14 - ATQClarice Mae Dacasin100% (1)
- Chem 31 AtqE1Documento3 páginasChem 31 AtqE1Anonymous GO6JVW9WudAinda não há avaliações
- Formal Report Chem 31.1Documento6 páginasFormal Report Chem 31.1Elah PalaganasAinda não há avaliações
- ATQ 11 Chem 31.1Documento2 páginasATQ 11 Chem 31.1BethAinda não há avaliações
- Synthesis of 1-Phenylazo 'Sudan 1'Documento5 páginasSynthesis of 1-Phenylazo 'Sudan 1'seryuyuyu100% (2)
- Chem 31 PROCEDURES (Practicals)Documento9 páginasChem 31 PROCEDURES (Practicals)FMDCAinda não há avaliações
- Experiment 13 Results and Discussion Report: Determination of Total Ion Concentration Using Ion Exchange ChromatographyDocumento3 páginasExperiment 13 Results and Discussion Report: Determination of Total Ion Concentration Using Ion Exchange ChromatographyNathalie Dagmang100% (3)
- Chem 31.1 FR1 SantosDocumento5 páginasChem 31.1 FR1 SantosClaire SantosAinda não há avaliações
- ATQ4Documento2 páginasATQ4Julius Alfredo ViloriaAinda não há avaliações
- Preparation and Purification of An Alkyl HalideDocumento4 páginasPreparation and Purification of An Alkyl HalideDaphne MercadoAinda não há avaliações
- Experiment 1 - ATQDocumento2 páginasExperiment 1 - ATQAndrea Nicole RocafortAinda não há avaliações
- Experiment 9: Relative Rates of Electrophilic Aromatic Substitution Chem 31.1 Post-LabDocumento3 páginasExperiment 9: Relative Rates of Electrophilic Aromatic Substitution Chem 31.1 Post-LabJoone Xyron CreenciaAinda não há avaliações
- Preparation and Purification of An Alkyl HalideDocumento8 páginasPreparation and Purification of An Alkyl HalideNoOneGotThisUsernameYetAinda não há avaliações
- Atq 11Documento4 páginasAtq 11AspWrites100% (1)
- Nucleophilic Acyl Substitution: The Synthesis of EstersDocumento5 páginasNucleophilic Acyl Substitution: The Synthesis of EstersNashDanielSilava100% (2)
- Midterm Exam Reviewer (Mas Malala Talaga Ang Real Exam)Documento3 páginasMidterm Exam Reviewer (Mas Malala Talaga Ang Real Exam)Ying YangAinda não há avaliações
- Carboxylic Acids and Acid DerivativesDocumento2 páginasCarboxylic Acids and Acid DerivativesJuan De FocaAinda não há avaliações
- UP CHEM 31 Carbohydrates Organic ChemistryDocumento3 páginasUP CHEM 31 Carbohydrates Organic ChemistryZsara CampanoAinda não há avaliações
- Qualitative Analysis of Carbohydrates ExperimentDocumento12 páginasQualitative Analysis of Carbohydrates Experimentasdf653652547Ainda não há avaliações
- CarbsDocumento10 páginasCarbszendaveapostolacidoAinda não há avaliações
- CHM 215 Biochemistry Laboratory: Biruni University Istanbul 2020Documento18 páginasCHM 215 Biochemistry Laboratory: Biruni University Istanbul 2020ŞEVVAL SARGINAinda não há avaliações
- Honors Chemistry - Course OutlineDocumento2 páginasHonors Chemistry - Course OutlineElah PalaganasAinda não há avaliações
- Palaganas Hw2 Che 197Documento5 páginasPalaganas Hw2 Che 197Elah Palaganas100% (1)
- Motion Notes: Speed, Velocity, and Acceleration!Documento18 páginasMotion Notes: Speed, Velocity, and Acceleration!Elah PalaganasAinda não há avaliações
- Lesson 1Documento77 páginasLesson 1Elah PalaganasAinda não há avaliações
- Newtonian Revolution: Mechanics Is The Branch of Physics That Focuses On The MotionDocumento12 páginasNewtonian Revolution: Mechanics Is The Branch of Physics That Focuses On The MotionElah PalaganasAinda não há avaliações
- Created by Unlicensed Version: OCTOBER 9, 2018Documento1 páginaCreated by Unlicensed Version: OCTOBER 9, 2018Elah PalaganasAinda não há avaliações
- Line Sizing Vapor LiquidDocumento25 páginasLine Sizing Vapor LiquidElah PalaganasAinda não há avaliações
- Trends & The Periodic TableDocumento34 páginasTrends & The Periodic TableElah PalaganasAinda não há avaliações
- Three Quatrains: 2 PoemDocumento1 páginaThree Quatrains: 2 PoemElah PalaganasAinda não há avaliações
- Zoology - Genetics-Botany - EcologyDocumento1 páginaZoology - Genetics-Botany - EcologyElah PalaganasAinda não há avaliações
- 1st LE - Seta Item AnalyzedDocumento10 páginas1st LE - Seta Item AnalyzedElah PalaganasAinda não há avaliações
- Effect of Different Parameters To % Oil RemainingDocumento3 páginasEffect of Different Parameters To % Oil RemainingElah PalaganasAinda não há avaliações
- ChE 133 WKL-FXY - Final StandingDocumento35 páginasChE 133 WKL-FXY - Final StandingElah PalaganasAinda não há avaliações
- Cost Saving Idea: Do Changeover From PKO To PO in CLSBE Units Only When The PO Demand IsDocumento2 páginasCost Saving Idea: Do Changeover From PKO To PO in CLSBE Units Only When The PO Demand IsElah PalaganasAinda não há avaliações
- Project Proposal PalaganasDocumento1 páginaProject Proposal PalaganasElah PalaganasAinda não há avaliações
- Non Verbal CommunicationDocumento40 páginasNon Verbal CommunicationElah PalaganasAinda não há avaliações
- Current Issues in The Palm Oil Industry Related To Environmental Sustainability: 1. 2. 3. 4. 5Documento1 páginaCurrent Issues in The Palm Oil Industry Related To Environmental Sustainability: 1. 2. 3. 4. 5Elah PalaganasAinda não há avaliações
- Physics 71 EquationsDocumento3 páginasPhysics 71 EquationsElah PalaganasAinda não há avaliações
- "Electric Circuit": Project in ScienceDocumento2 páginas"Electric Circuit": Project in ScienceElah PalaganasAinda não há avaliações
- HSEDocumento7 páginasHSEElah PalaganasAinda não há avaliações
- Profile Profile: Writing Style Educational BackgroundDocumento5 páginasProfile Profile: Writing Style Educational BackgroundElah PalaganasAinda não há avaliações
- GRADE 5 - SOCIAL STUDIES - NotesDocumento34 páginasGRADE 5 - SOCIAL STUDIES - NotesJaveria FatimaAinda não há avaliações
- Ijsrmjournal, Journal Manager, 5 IjsrmDocumento10 páginasIjsrmjournal, Journal Manager, 5 IjsrmOshiniAinda não há avaliações
- Cancer Cytopathology - 2023 - Rossi - Second Edition of The Milan System For Reporting Salivary Gland CytopathologyDocumento12 páginasCancer Cytopathology - 2023 - Rossi - Second Edition of The Milan System For Reporting Salivary Gland Cytopathologyskraja7534Ainda não há avaliações
- Case Study Drinking Up The PaycheckDocumento1 páginaCase Study Drinking Up The Paychecksmart.soldier110Ainda não há avaliações
- Mental Disorders 1Documento15 páginasMental Disorders 1api-509374276Ainda não há avaliações
- Biochemic Medicines For Twelve Tissue Remedies by Masood HomeopathyDocumento4 páginasBiochemic Medicines For Twelve Tissue Remedies by Masood Homeopathyadeelshahzadqureshi8086100% (1)
- Alcoholism in The Animal AbuseDocumento3 páginasAlcoholism in The Animal Abusejulian stiven rodriguez tamayoAinda não há avaliações
- Brochure - Alere Determine TB LAM AgDocumento6 páginasBrochure - Alere Determine TB LAM AgHandoko HalimAinda não há avaliações
- Hair Product Formation - Project ProposalDocumento4 páginasHair Product Formation - Project ProposalLee ViramAinda não há avaliações
- Catalan vs. Basa (Digest)Documento2 páginasCatalan vs. Basa (Digest)Tini GuanioAinda não há avaliações
- Informative Speech TemplateDocumento6 páginasInformative Speech Templateapi-267083617Ainda não há avaliações
- B1+ Editable Final TestDocumento6 páginasB1+ Editable Final TestTrifan DavidAinda não há avaliações
- Unit 1 Chap 1 and 2Documento7 páginasUnit 1 Chap 1 and 2Ariane Grace OcampoAinda não há avaliações
- Executive Order 1Documento2 páginasExecutive Order 1Cy ValenzuelaAinda não há avaliações
- Thesis Statement On Pit BullsDocumento6 páginasThesis Statement On Pit BullsNeedHelpWithPaperSingapore100% (2)
- Post-Traumatic Stress DisorderDocumento1 páginaPost-Traumatic Stress DisorderVictor FreebsdAinda não há avaliações
- Jurnal Stikes Awal Bros PanamDocumento6 páginasJurnal Stikes Awal Bros PanamNaili NurAinda não há avaliações
- IPAQ - AUTOMATIC REPORT - English Advanced Version - Self-Admin Short - Di Blasio & Izzicupo Et AlDocumento20 páginasIPAQ - AUTOMATIC REPORT - English Advanced Version - Self-Admin Short - Di Blasio & Izzicupo Et AlAhmad Naufal AminAinda não há avaliações
- NFS Journal: Navomy George, Annica A.M. Andersson, Roger Andersson, Afaf Kamal-EldinDocumento6 páginasNFS Journal: Navomy George, Annica A.M. Andersson, Roger Andersson, Afaf Kamal-EldinMuhammad AdiAinda não há avaliações
- Faktor-Faktor Yang Berhubungan Dengan Kejadian Anemia Pada Ibu HamilDocumento10 páginasFaktor-Faktor Yang Berhubungan Dengan Kejadian Anemia Pada Ibu Hamilsri rahmatikaAinda não há avaliações
- Caviar-Extract Paraben-Free TDS PDFDocumento2 páginasCaviar-Extract Paraben-Free TDS PDFamalinaAinda não há avaliações
- Topic 1: Before You Begin Evaluate and Prioritise The Needs of A Person With Complex Support IssuesDocumento20 páginasTopic 1: Before You Begin Evaluate and Prioritise The Needs of A Person With Complex Support Issuesdefa rey100% (1)
- Nasm FlashcardsDocumento172 páginasNasm FlashcardsHilde Camp100% (3)
- Lab Man Prelims To FinalsDocumento42 páginasLab Man Prelims To FinalsMica CapistranoAinda não há avaliações
- De Thi Thu THPT QG 2021 Mon Anh Hong Linh Lan 1Documento5 páginasDe Thi Thu THPT QG 2021 Mon Anh Hong Linh Lan 1nhat baoAinda não há avaliações
- Provide Advanced Nursing Care (HLT NUR4 01222) : For Regular Nursing (Level Iv - TVET) Students AAMBC, January 2022Documento56 páginasProvide Advanced Nursing Care (HLT NUR4 01222) : For Regular Nursing (Level Iv - TVET) Students AAMBC, January 2022Abel100% (1)
- Module 10: Challenging Maladaptive Thoughts and Beliefs: ObjectivesDocumento7 páginasModule 10: Challenging Maladaptive Thoughts and Beliefs: ObjectivesMaria BagourdiAinda não há avaliações
- Most Common Chronic Pain ConditionsDocumento7 páginasMost Common Chronic Pain ConditionsNicolas RitoAinda não há avaliações
- Outcomes 1Documento449 páginasOutcomes 1Sam SubediAinda não há avaliações
- Medical TechnologyDocumento14 páginasMedical Technologydprincebuchikoy100% (1)