Escolar Documentos
Profissional Documentos
Cultura Documentos
2 3 Useful Chemical Building Blocks
Enviado por
griggans100%(1)100% acharam este documento útil (1 voto)
198 visualizações1 páginaYou should be able to: define an element, compound and mixture. Match up symbols and names of elements. Classify materials as elements, compounds or mixtures using information provided.
Descrição original:
Título original
2 3 useful chemical building blocks
Direitos autorais
© Attribution Non-Commercial (BY-NC)
Formatos disponíveis
DOC, PDF, TXT ou leia online no Scribd
Compartilhar este documento
Compartilhar ou incorporar documento
Você considera este documento útil?
Este conteúdo é inapropriado?
Denunciar este documentoYou should be able to: define an element, compound and mixture. Match up symbols and names of elements. Classify materials as elements, compounds or mixtures using information provided.
Direitos autorais:
Attribution Non-Commercial (BY-NC)
Formatos disponíveis
Baixe no formato DOC, PDF, TXT ou leia online no Scribd
100%(1)100% acharam este documento útil (1 voto)
198 visualizações1 página2 3 Useful Chemical Building Blocks
Enviado por
griggansYou should be able to: define an element, compound and mixture. Match up symbols and names of elements. Classify materials as elements, compounds or mixtures using information provided.
Direitos autorais:
Attribution Non-Commercial (BY-NC)
Formatos disponíveis
Baixe no formato DOC, PDF, TXT ou leia online no Scribd
Você está na página 1de 1
2.
3 Useful Chemicals (Building Key Points Key terms
blocks)
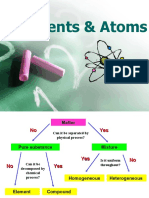
Syllabus Summary (H) = higher only Elements, Compounds and Mixtures Element
You should be able to: • Elements contain only 1 type of atom, compounds contain at least 2 different atoms Product
Compound Balanced
• define an element, compound and mixture; joined together
Mixture Formula
• match up symbols and names of elements • Compounds are very difficult to separate into their elements
Atom Organic
• recall some chemical symbols • Mixtures are two substances that are not chemically combined
Nucleus Inorganic
• classify materials as elements, compounds • Mixtures are easy to separate
Proton
or mixtures using information provided
Neutron
• State that all matter is made up of atoms
Electron
• recall the structure of the atom
Reactant
• recall that all of the atoms that make up an
element have the same number of protons
and electrons in their atoms.
• Match up formulae and names of some
compounds
• Recognise reactants and products in a Element Compound Mixture
symbol equation
• name the some compounds listed given The Atom
• Atoms are made up of protons, neutrons and electrons. Protons and neutrons are
their formulae;
found in the nucleus, electrons orbit the nucleus in shells. The first shell can hold a
• write word and symbol equations to show
max of 2 electrons, the second and third shells hold a max of 8
how atoms are rearranged in the chemical
• Protons are positive and have a mass of 1, neutrons are neutral and have a mass of 1
reactions
and electrons are negative and have a mass of 0
• recall and use the formulae of some
compounds l to write balanced symbol Chemical Symbols and Formulae
equations (H)
• Most element symbols are the first letter of the name e.g. Carbon = C
• state that scientists sort out chemicals into C2H7N
• Some are the first two letters of the name e.g. Cobalt = Co
two groups, those that contain carbon and
those that do not; • A few are the first letter and another letter from the name e.g. Magnesium = Mg
• state that living things all contain the • Some are the first letter or letters of the latin name e.g. Sodium = Natrium = Na
element carbon and non-living usually don’t • The formula of a compound tells us which atoms and how many it contains e.g.
• recall the names of some chemicals that do CO2 = 1 x carbon atom and 2 x oxygen atoms
not contain the element carbon
• recognise inorganic and organic Equations
substances; • An equation can be written in words or symbols
• use the words organic and inorganic
correctly and explain the words origin E.G. Magnesium + Oxygen → Magnesium Oxide
• use chemical formulae to recognise a 2 Mg + O2 → 2MgO (must be balanced)
substance as organic or inorganic
(reactants) → (products)
• recall that there are many useful
substances that contain carbon atoms
Organic compounds
• explain why fossil fuels are a source of
• Organic compounds contain carbon, inorganic compound don’t
organic substances;
• Livings things all contain the element carbon
• state that organic chemicals are very • Many useful substances contain carbon e.g. plastics, petrol, products from oil
important in our lives and most are derived
from crude oil.
Você também pode gostar
- TB Unit3progresscheckfrq 5dc331c19f6919Documento3 páginasTB Unit3progresscheckfrq 5dc331c19f6919api-486324042Ainda não há avaliações
- Midterm 1 BIS2A Section KeyDocumento6 páginasMidterm 1 BIS2A Section KeyKevinZamudioAinda não há avaliações
- Schaum's Easy Outline of Organic Chemistry, Second EditionNo EverandSchaum's Easy Outline of Organic Chemistry, Second EditionNota: 3.5 de 5 estrelas3.5/5 (2)
- Cambridge International AS & A Level Chemistry Practical Workbook Roger Norris, Mike Wooster (2020)Documento225 páginasCambridge International AS & A Level Chemistry Practical Workbook Roger Norris, Mike Wooster (2020)APURVA YADAV100% (6)
- Lab Report Biochemistry-Estimation of Iodine Value of Fats and OilsDocumento6 páginasLab Report Biochemistry-Estimation of Iodine Value of Fats and OilsNida Ridzuan67% (3)
- Laundry - An Housekeeper PerspectiveDocumento24 páginasLaundry - An Housekeeper Perspectivegrgnolis100% (1)
- Percentage of Polymers Used in Packaging: Figure 1 Shows A Kite Flying in The AirDocumento6 páginasPercentage of Polymers Used in Packaging: Figure 1 Shows A Kite Flying in The AirKhadijahAinda não há avaliações
- Chapter 1: Atoms, Molecules and IonsDocumento103 páginasChapter 1: Atoms, Molecules and IonsSyahir HamidonAinda não há avaliações
- GRADE 8 CHEMISTRY Periodic Table Lesson 1Documento20 páginasGRADE 8 CHEMISTRY Periodic Table Lesson 1dodoAinda não há avaliações
- Topic 3 Atomic StructureDocumento36 páginasTopic 3 Atomic StructureKaixin HuangAinda não há avaliações
- BMED 105 Basic Chemistry of Life (NOTES)Documento4 páginasBMED 105 Basic Chemistry of Life (NOTES)Jobelle MalihanAinda não há avaliações
- Essentials Pearson 2016Documento253 páginasEssentials Pearson 2016Devansh SharmaAinda não há avaliações
- Elements Compounds and MixturesDocumento24 páginasElements Compounds and MixturesMegan SarahAinda não há avaliações
- 1 Lec - ChemDocumento6 páginas1 Lec - ChemAllen Rivera ReyesAinda não há avaliações
- CHM 092 CHAPTER 1 - Matter &stoichiometryDocumento128 páginasCHM 092 CHAPTER 1 - Matter &stoichiometryAisyah NadhirahAinda não há avaliações
- Chapter 2 Complete Lecture 4x4Documento17 páginasChapter 2 Complete Lecture 4x4michaelaAinda não há avaliações
- Non Metalic Substances and Covalent BondingDocumento47 páginasNon Metalic Substances and Covalent Bonding42h47n5zvrAinda não há avaliações
- Lecture 3 - Matter, Molecules and Periodic TableDocumento40 páginasLecture 3 - Matter, Molecules and Periodic TableNaff WariAinda não há avaliações
- Study Guides 2.1-2.3Documento8 páginasStudy Guides 2.1-2.3MA. ASUNCION BeroAinda não há avaliações
- Chapter 1 - Chemical FoundationDocumento50 páginasChapter 1 - Chemical Foundation杨致远Ainda não há avaliações
- Engineering Chemistry: Course Code: 211502 Course NameDocumento17 páginasEngineering Chemistry: Course Code: 211502 Course NameSalama NaumanAinda não há avaliações
- Chapter 2 - Material Structure and Interatomic BondingDocumento33 páginasChapter 2 - Material Structure and Interatomic BondingamraqstnaAinda não há avaliações
- Intro Matter 2 PHDocumento27 páginasIntro Matter 2 PHCheick SANOUAinda não há avaliações
- Chapter 2 - Atoms and MoleculesDocumento62 páginasChapter 2 - Atoms and MoleculesRoselyn CastilloAinda não há avaliações
- BioM I Lectures 1 - 3 (SB) 2014Documento69 páginasBioM I Lectures 1 - 3 (SB) 2014nonhle nosiphoAinda não há avaliações
- Chemistry Workbook 1Documento10 páginasChemistry Workbook 1Dean KimAinda não há avaliações
- Atoms, Elements and Molecules & Compounds and MixturesDocumento32 páginasAtoms, Elements and Molecules & Compounds and MixturesTshanna RobertsAinda não há avaliações
- Lecture #9 Introduction To Organic Chemistry Organic ChemistryDocumento10 páginasLecture #9 Introduction To Organic Chemistry Organic ChemistryG8 ODL Mary Angeline M. GalmanAinda não há avaliações
- Atoms and Molecules: Larry Brown Tom HolmeDocumento62 páginasAtoms and Molecules: Larry Brown Tom Holmemuhammad ali shakeelAinda não há avaliações
- CHEMISTRYDocumento3 páginasCHEMISTRYSAN JOSE, KRIZZIA FAYE U.Ainda não há avaliações
- Naming CompoundsDocumento39 páginasNaming CompoundsgallosaaronmatthewAinda não há avaliações
- Org Chem 3Documento11 páginasOrg Chem 3Bertille Marie ArdienteAinda não há avaliações
- Org Chem 5Documento12 páginasOrg Chem 5Bertille Marie ArdienteAinda não há avaliações
- Org Chem 2Documento12 páginasOrg Chem 2Bertille Marie ArdienteAinda não há avaliações
- Elements, Atoms, and The Atomic Theory: Type of AtomDocumento7 páginasElements, Atoms, and The Atomic Theory: Type of AtomSasikumar MadhavanAinda não há avaliações
- GEN CHEM CHEMICAL FORMULA and NAMING OF COMPOUNDSDocumento35 páginasGEN CHEM CHEMICAL FORMULA and NAMING OF COMPOUNDSKC KayeAinda não há avaliações
- Chemistry of LifeDocumento11 páginasChemistry of LifeNadiyah KarriemAinda não há avaliações
- Quarter 1 Module 2 Elements and CompoundsDocumento30 páginasQuarter 1 Module 2 Elements and CompoundsJeline MacallaAinda não há avaliações
- Science 9: The Variety of Carbon CompoundsDocumento10 páginasScience 9: The Variety of Carbon Compoundsrussel castilloAinda não há avaliações
- Principles of BiochemistryDocumento68 páginasPrinciples of Biochemistryblackss copsAinda não há avaliações
- Chapter 1: Fundamentals of Chemistry: Lesson 2: Atoms and MoleculesDocumento9 páginasChapter 1: Fundamentals of Chemistry: Lesson 2: Atoms and MoleculesKristine Cris VenusAinda não há avaliações
- Typeselements CompoundsDocumento27 páginasTypeselements CompoundsEverrome AsicoAinda não há avaliações
- 2.1 The Chemistry of Life EditedDocumento68 páginas2.1 The Chemistry of Life EditedPatricia Jayshree Samuel Jacob100% (1)
- Campbell Lecture Notes Chemistry of LifeDocumento42 páginasCampbell Lecture Notes Chemistry of LifeSophia Andrei VillalunaAinda não há avaliações
- CLASS 10 - ChemistryDocumento38 páginasCLASS 10 - ChemistryKavyansh GuptaAinda não há avaliações
- The Differences Between A Compound and A MixtureDocumento27 páginasThe Differences Between A Compound and A Mixturearies triwidajatiAinda não há avaliações
- Atom, Molecules & Ions (Ref: Dr. Khoa, Dr. Khánh, Dr. Thuận)Documento22 páginasAtom, Molecules & Ions (Ref: Dr. Khoa, Dr. Khánh, Dr. Thuận)HIEP PHAM HOANGAinda não há avaliações
- Basic Chemistry I: Gasal 2011/2012Documento39 páginasBasic Chemistry I: Gasal 2011/2012Mitch EspinasAinda não há avaliações
- Atoms PresentationDocumento10 páginasAtoms PresentationSu AlghAinda não há avaliações
- M1 Lec - HandoutDocumento5 páginasM1 Lec - HandoutAngel Fiona GañaAinda não há avaliações
- Study Questions Introduction To ChemistryDocumento5 páginasStudy Questions Introduction To ChemistryMsAinda não há avaliações
- Quarter1.Week2.Elements and CompoundDocumento47 páginasQuarter1.Week2.Elements and CompoundEvelyn ApostolAinda não há avaliações
- Chemistry Functional Groups LECTUREDocumento34 páginasChemistry Functional Groups LECTUREkassy jayAinda não há avaliações
- Elements and AtomsDocumento28 páginasElements and AtomsJulien Kristi HernandezAinda não há avaliações
- 3.3 Chemical FormulaDocumento18 páginas3.3 Chemical FormulaLIM CHEE BOON MoeAinda não há avaliações
- 2.1 Functional GroupsDocumento4 páginas2.1 Functional GroupsBasti SantiagoAinda não há avaliações
- Chapter 4-CarbonDocumento23 páginasChapter 4-Carbonjanardhan aghavAinda não há avaliações
- ReactivityDocumento42 páginasReactivityDarryl WHDAinda não há avaliações
- Lesson 4 General Chemistry 1Documento16 páginasLesson 4 General Chemistry 1Genevee Ryeleen DelfinAinda não há avaliações
- Chap 03Documento22 páginasChap 03AmandaAinda não há avaliações
- Edexcel AS Chemistry Note 1 Definitions of The TermsDocumento3 páginasEdexcel AS Chemistry Note 1 Definitions of The TermsSajaniAinda não há avaliações
- BASIC CHEMISTRY Lect 1Documento23 páginasBASIC CHEMISTRY Lect 1briosojoshua0Ainda não há avaliações
- Lesson 2 and 3 ChemDocumento3 páginasLesson 2 and 3 ChemJazmine Lei PalomoAinda não há avaliações
- Chemistry of Cell Review: Quick Review Notes Chapter 2No EverandChemistry of Cell Review: Quick Review Notes Chapter 2Ainda não há avaliações
- Heart Gcse Qs OnlyDocumento6 páginasHeart Gcse Qs OnlygriggansAinda não há avaliações
- Exam-QuestionsQUADRATS AND TRANSECTSDocumento8 páginasExam-QuestionsQUADRATS AND TRANSECTSgriggansAinda não há avaliações
- Chemistry c1 Core PracticalsDocumento18 páginasChemistry c1 Core PracticalsgriggansAinda não há avaliações
- Physics p1 Core PracticalsDocumento19 páginasPhysics p1 Core PracticalsgriggansAinda não há avaliações
- Biology b1 Core Practical QuestionsDocumento17 páginasBiology b1 Core Practical Questionsgriggans0% (1)
- Biology FieldworkqsDocumento9 páginasBiology FieldworkqsgriggansAinda não há avaliações
- Balanced ForcesDocumento17 páginasBalanced ForcesgriggansAinda não há avaliações
- Paying 4 ElectricityDocumento1 páginaPaying 4 ElectricitygriggansAinda não há avaliações
- Microorganisms and FoodDocumento4 páginasMicroorganisms and FoodgriggansAinda não há avaliações
- BTEC Science UNIT 5 NervesDocumento4 páginasBTEC Science UNIT 5 NervesgriggansAinda não há avaliações
- BTEC Science UNIT 5 HomeostasisDocumento5 páginasBTEC Science UNIT 5 Homeostasisgriggans100% (4)
- ReactionsandparticlesDocumento4 páginasReactionsandparticlesgriggansAinda não há avaliações
- The Haber ProcessDocumento1 páginaThe Haber ProcessgriggansAinda não há avaliações
- Hormones: Part of Homeostasis and ControlDocumento7 páginasHormones: Part of Homeostasis and ControlgriggansAinda não há avaliações
- As RevisionDocumento4 páginasAs RevisiongriggansAinda não há avaliações
- Extract LeadDocumento2 páginasExtract LeadgriggansAinda não há avaliações
- Bending LightDocumento24 páginasBending Lightgriggans100% (2)
- Metals in AirDocumento1 páginaMetals in AirgriggansAinda não há avaliações
- Revision Class NotesDocumento7 páginasRevision Class NotesgriggansAinda não há avaliações
- GSK AssignmentDocumento4 páginasGSK Assignmentgriggans100% (4)
- SlimeDocumento5 páginasSlimegriggansAinda não há avaliações
- ClassificationDocumento7 páginasClassificationgriggansAinda não há avaliações
- Igneous RocksDocumento4 páginasIgneous RocksgriggansAinda não há avaliações
- How To Reference - Student GuideDocumento1 páginaHow To Reference - Student GuidegriggansAinda não há avaliações
- ReflectionDocumento20 páginasReflectiongriggansAinda não há avaliações
- BTEC First Diploma in Applied Science Student Tracking SheetsDocumento6 páginasBTEC First Diploma in Applied Science Student Tracking Sheetsgriggans100% (6)
- Chromatography StarterDocumento1 páginaChromatography StartergriggansAinda não há avaliações
- Tomatosphere AssignmentDocumento2 páginasTomatosphere AssignmentgriggansAinda não há avaliações
- Task and Mark Sheet 1)Documento6 páginasTask and Mark Sheet 1)griggans100% (1)
- Waves and Radiation Front SheetDocumento3 páginasWaves and Radiation Front Sheetgriggans100% (5)
- Mole Conversions WorksheetDocumento4 páginasMole Conversions Worksheetfatzr100% (1)
- Methods For Chemical Analysis of Steels: Indian StandardDocumento5 páginasMethods For Chemical Analysis of Steels: Indian StandardAryansh Rocky RanaAinda não há avaliações
- Polybase Base PlateDocumento24 páginasPolybase Base PlateNumb JazzAinda não há avaliações
- Determination of Aspirin by Indirect Titration de Jesus, Mirra PDocumento7 páginasDetermination of Aspirin by Indirect Titration de Jesus, Mirra PMirraDeJesusAinda não há avaliações
- Materials: A Study On Tannery Sludge As A Raw Material For Cement MortarDocumento14 páginasMaterials: A Study On Tannery Sludge As A Raw Material For Cement MortarKumar PallavAinda não há avaliações
- Gravimetric Analysis: Gravi - Metric (Weighing - Measure)Documento23 páginasGravimetric Analysis: Gravi - Metric (Weighing - Measure)Ulfa WulandariAinda não há avaliações
- 4 and 5Documento2.908 páginas4 and 5Rina DasAinda não há avaliações
- 3.1 Weathering and SoilsDocumento21 páginas3.1 Weathering and SoilsjjjAinda não há avaliações
- Module 3 Instrumental Methods and NanomaterialsDocumento23 páginasModule 3 Instrumental Methods and Nanomaterialsandru media workAinda não há avaliações
- Intermolecular Forces of AttractionDocumento26 páginasIntermolecular Forces of AttractionEsaïe GreñaAinda não há avaliações
- Replacement of Gas Phase With Liquid HexamineDocumento6 páginasReplacement of Gas Phase With Liquid HexaminePradhita Ramdani HAinda não há avaliações
- Mathematical Modeling of Dissolved Oxygen in Fish Ponds: Full Length Research PaperDocumento14 páginasMathematical Modeling of Dissolved Oxygen in Fish Ponds: Full Length Research PaperBayeAinda não há avaliações
- Chemistry Independent Honors Project ProposalDocumento8 páginasChemistry Independent Honors Project Proposalapi-242175324Ainda não há avaliações
- Manual Analysis Methods For The Brewery Industry Prove 05 2021 Final WebDocumento130 páginasManual Analysis Methods For The Brewery Industry Prove 05 2021 Final WebleidyAinda não há avaliações
- CPD IsoprenoDocumento12 páginasCPD IsoprenoRodrigo Ortiz SotoAinda não há avaliações
- Self Healing Concrete Based On Different Bacteria: A ReviewDocumento7 páginasSelf Healing Concrete Based On Different Bacteria: A ReviewMaria IonescuAinda não há avaliações
- AL Chemistry 1996 Paper 1+2Documento12 páginasAL Chemistry 1996 Paper 1+2api-3734333Ainda não há avaliações
- Eigenmann & VeronelliDocumento8 páginasEigenmann & VeronelliMatteoAinda não há avaliações
- LAB Plug FlowDocumento24 páginasLAB Plug FlowZalina SamsuddinAinda não há avaliações
- 1 - Chapter 1 PDFDocumento82 páginas1 - Chapter 1 PDFMeenakshi AnilAinda não há avaliações
- Demo InflatingaballoonDocumento4 páginasDemo InflatingaballoonjowieAinda não há avaliações
- CE103 GlassDocumento21 páginasCE103 GlassMuhammad AmjadAinda não há avaliações
- Ristig 2022 Ammonia Decomposition in The ProcesDocumento13 páginasRistig 2022 Ammonia Decomposition in The ProcesJosh WangAinda não há avaliações
- Pages From Handbook of Plant and Crop Stress Third Ed 2011 TitluDocumento28 páginasPages From Handbook of Plant and Crop Stress Third Ed 2011 TitluCoțovanu IulianAinda não há avaliações