Escolar Documentos
Profissional Documentos
Cultura Documentos
Roux 2013
Enviado por
andrew herringDescrição original:
Direitos autorais
Formatos disponíveis
Compartilhar este documento
Compartilhar ou incorporar documento
Você considera este documento útil?
Este conteúdo é inapropriado?
Denunciar este documentoDireitos autorais:
Formatos disponíveis
Roux 2013
Enviado por
andrew herringDireitos autorais:
Formatos disponíveis
PAIN 154 (2013) 14421448
www.elsevier.com/locate/pain
Buprenorphine/naloxone as a promising therapeutic option for opioid
abusing patients with chronic pain: Reduction of pain, opioid
withdrawal symptoms, and abuse liability of oral oxycodone
Perrine Roux a,b,c,d,, Maria A. Sullivan a, Julien Cohen b,c, Lionel Fugon b,c, Jermaine D. Jones a,
Suzanne K. Vosburg a, Ziva D. Cooper a, Jeanne M. Manubay a, Shanthi Mogali a, Sandra D. Comer a
a
Division on Substance Abuse, New York State Psychiatric Institute, Department of Psychiatry, and College of Physicians and Surgeons, Columbia University, 1051 Riverside Dr,
Unit 120, New York, NY 10032, USA
b
INSERM U912 (SESSTIM), Marseille, France
c
Universit Aix Marseille, IRD, UMR-S912, Marseille, France
d
ORS PACA, Observatoire Rgional de la Sant Provence Alpes Cte dAzur, Marseille, France
Sponsorships or competing interests that may be relevant to content are disclosed at the end of this article.
a r t i c l e
i n f o
Article history:
Received 20 November 2012
Received in revised form 12 April 2013
Accepted 1 May 2013
Keywords:
Buprenorphine
Chronic pain
Opioid dependence
Oxycodone
Reinforcing effect
a b s t r a c t
Few studies have examined abuse of prescription opioids among individuals with chronic pain under
buprenorphine/naloxone (Bup/Nx) maintenance. The current 7-week inpatient study assessed oral oxycodone self-administration by patients with chronic pain who had a history of opioid abuse. Participants
(n = 25) were transitioned from their preadmission prescribed opioid to Bup/Nx. All of the participants
were tested under each of the sublingual Bup/Nx maintenance doses (2/0.5, 8/2 or 16/4 mg) in random
order. During each maintenance period, participants could self-administer oxycodone orally (0, 10, 20,
40 or 60 mg prescription opioids) or receive money during laboratory sessions. Drug choice (percentage)
was the primary dependent variable. Subjective ratings of clinical pain and withdrawal symptoms also
were measured. Mann-Whitney tests compared percentage of drug choice for each active oxycodone dose
to placebo. Logistic regression analyses identied correlates of oxycodone preference, dened as 60% or
greater choice of oxycodone compared to money. Pain was signicantly reduced while participants were
maintained on Bup/Nx compared to preadmission ratings. No differences in percentage drug choice were
observed between the active oxycodone doses and placebo under each Bup/Nx maintenance dose. However, factors associated with oxycodone preference were lower Bup/Nx maintenance dose, more withdrawal symptoms and more pain. These data suggest that Bup/Nx was effective in reducing pain and
supplemental oxycodone use. Importantly, adequate management of pain and withdrawal symptoms
by Bup/Nx may reduce oxycodone preference in this population.
Published by Elsevier B.V. on behalf of International Association for the Study of Pain.
1. Introduction
In the United States, the problem of nonmedical use of prescription opioids has emerged as a major public health issue [1]. Other
countries, such as Australia, New Zealand [12] and Canada [33],
also are concerned about the phenomenon of prescription opioids
abuse. In the United States, oxycodone and hydrocodone are
among the most commonly prescribed or regularly used opioids,
as well as the most commonly diverted prescription opioids analgesics [5,25]. These data indicate that prescription opioids abuse
has steadily increased among heroin and recreational polydrug
users since 2000 [5]. An additional concern related to the increased
Corresponding author at: INSERM U912 (SESSTIM), Marseille, France. Tel.: +33 4
96102876.
E-mail address: perrine.roux@inserm.fr (P. Roux).
use of prescription opioids is opioid overdose, which increased in
the United States from the mid-1990s to the present time [10,11]
and recently became a leading cause of accidental death in the
United States [4]. Thus, the risks of prescription opioids abuse
and overdose make physicians reluctant to prescribe prescription
opioids in general, and access to adequate pain management in
drug users in particular is becoming increasingly difcult [20,39].
In those patients who are prescribed prescription opioids for pain
relief, misuse may occur in pain patients with no history of opioid
abuse who become dependent on the medications for their reinforcing properties, whether good drug effects or relief of anxiety
or mood symptoms, or misuse may occur in drug-seeking individuals with preexisting opioid abuse histories. Thus, balancing the
need for effective pain relief and reducing the risks of opioid abuse
and overdose remains a challenge for public health policy [7].
0304-3959/$36.00 Published by Elsevier B.V. on behalf of International Association for the Study of Pain.
http://dx.doi.org/10.1016/j.pain.2013.05.004
P. Roux et al. / PAIN 154 (2013) 14421448
In this context of easy access to opioid analgesics for the general
population, opioid overdoses due to diversion [8] and difculties in
accessing treatment for pain and opioid dependence, expanding
access to a less risky treatment for pain and opioid dependence
is a logical and plausible public health response.
For decades, buprenorphine, a partial l-opioid agonist, has been
used to treat acute and chronic pain [16]. More recently, a sublingual formulation of buprenorphine has been used to treat opioid
dependence [38] in both substance abuse treatment clinics and
in primary care settings [18]. Although sublingual buprenorphine
is not approved by the U.S. Food and Drug Administration for treating pain, some have suggested its use in the management of concurrent pain and opioid dependence [23]. However, few studies
have systematically examined whether buprenorphine maintenance alters prescription opioids abuse in this population. Thus,
assessment of the effectiveness of buprenorphine in treating pain
and also reducing the abuse liability of oxycodone would be relevant to the public health problem of prescription opioids abuse.
In the current study, patients with chronic, nonmalignant pain
who were abusing their prescription opioids medications were
maintained on different doses of sublingual buprenorphine/naloxone (2/0.5, 8/2 and 16/4 mg per day in divided doses) and given the
opportunity to self-administer oxycodone in a laboratory setting.
2. Methods
2.1. Participants
All of the participants were under the care of a physician for
mild to moderate chronic, nonmalignant pain. They were also required to meet DSM-IV criteria for opioid dependence, but were
not seeking treatment for their opioid dependence. Potential participants were excluded from the study if they had a current major
axis I psychopathology other than opioid dependence (ie, schizophrenia or bipolar disorder) or met DSM-IV criteria for dependence
on drugs other than opioids, nicotine or caffeine, or had a primary
diagnosis of neuropathic pain, malignant pain, headache or chronic
lower back pain with failed surgeries. Current buprenorphine
maintenance and history of failed treatment with buprenorphine
maintenance for pain also were exclusionary.
2.2. Data collection
After completing an initial telephone interview, eligible participants came into the laboratory to provide consent to receive additional screening, which included completing detailed medical
history and drug use questionnaires, interviews with a psychologist
and psychiatrist, and a medical evaluation conducted by a physician.
Prescription opioids use was ascertained in multiple ways (selfreport, verication with prescribing physician and/or presentation
of prescription opioids bottles) and was converted to number of morphine equivalents used per day. We also collected sociodemographic
data such as age, gender, ethnicity, education and employment. Urine
drug toxicologies (assessed by urine quick tests) also were performed
several times during screening to test for opioids, benzoylecgonine
(cocaine metabolite), benzodiazepines, cannabinoids and amphetamines. During laboratory sessions, subjective responses were measured and reinforcing effects of oxycodone were assessed.
1443
naloxone combination (Bup/Nx). During the rst week after admission, participants were withdrawn from their previous opioid analgesic regimen and stabilized on 1 of 3 doses of Bup/Nx (2/0.5, 8/2
or 16/4 mg/d). The total daily Bup/Nx dose was administered q.i.d.
in equally divided doses throughout the day (Fig. 1). Participants
were treated for emergent withdrawal symptoms with various
supplemental medications until withdrawal symptoms dissipated
based on self-report and observer ratings. Patients were maintained on each Bup/Nx dose for approximately 2 weeks: 1 week
of stabilization followed by 1 week of laboratory testing. Each participant received all 3 Bup/Nx doses in random order under double-blind conditions.
2.4. Reinforcing effects
During each maintenance period, participants could self-administer oral oxycodone (0, 10, 20, 40 or 60 mg) during separate laboratory sessions. Each laboratory day consisted of 2 types of
sessions, a sample session during which participants were provided with one of the possible doses of drug (oxycodone) and
US$20, and a self-administration (choice) session that occurred a
few hours later on the same day. During the sample session, the
subjective, physiological and analgesic effects of oxycodone were
measured. During the choice session, participants could selfadminister the dose of oxycodone that was provided during the
sample session or receive money. Participants then completed a
self-administration task to receive portions of the dose of drug or
money they sampled (0100% in increments of 10%). For each
10% increment of drug or money, participants were required to
complete an increasing number of nger presses on a computer
mouse (50, 100, 200, 400, 800, 1200, 1600, 2000, 2400, 2800).
Immediately after the self-administration task, money and/or the
total amount of drug earned during the task was administered.
The sample session began at approximately 11 AM, and the choice
session began at approximately 3 PM (Fig. 1).
2.5. Subjective effects
All of the questionnaires used in this study have been described
previously [17]. For the present analysis, opioid withdrawal symptoms and clinical pain were measured after administration of the
rst daily Bup/Nx maintenance dose. Subjective symptoms of opioid withdrawal were assessed with the Subjective Opioid Withdrawal Scale (SOWS; range 064 [9]). Clinical pain assessment
was made with the 15-item Short-Form McGill Pain Questionnaire
[26] based on their general pain condition while maintained on
Bup/Nx. Clinical pain was also assessed with the McGill Pain Questionnaire (MPQ) at the rst screening visit before initiation of Bup/
Nx maintenance.
2.3. Study design
This study was described in detail elsewhere in a report of preliminary results [17]. Individuals who met the eligibility criteria
were admitted to an inpatient research unit for 7 weeks and transitioned from their baseline prescription opioids to buprenorphine/
Fig. 1. Time points of a laboratory session day during the second week of
maintenance. Oxy, oxycodone; Bup, buprenorphine; Nx, naloxone.
1444
P. Roux et al. / PAIN 154 (2013) 14421448
2.6. Statistical analyses
For the present analysis, we used only a subset of the dependent
variables collected during the study because of our focused interest
in the impact of Bup/Nx maintenance dose, pain level and withdrawal symptoms on oxycodone self-administration. The time
point used in our analyses was 1 h after the rst Bup/Nx dose
administration in order to capture peak Bup/Nx effects on pain
and opioid withdrawal symptoms in the absence of oxycodone
effects.
In addition, as we selected only individuals who completed the
7-week inpatient laboratory period, we compared participants
who had complete data for the outcomes of interest to those
who did not in order to conrm the absence of a selection bias.
The rst analysis consisted of assessing the effectiveness of Bup/
Nx on pain perception comparing the MPQ score at baseline before
Bup/Nx initiation and at each laboratory session under the different Bup/Nx maintenance doses. We performed a bivariate analysis
between MPQ score at baseline and MPQ score under each Bup/Nx
maintenance dose using a generalized estimating equations (GEE)
model to take into account the intraindividual correlation for pain
measurements under Bup/Nx maintenance. In addition, we used a
linear regression model based on GEE to assess the association between MPQ pain score and Bup/Nx maintenance doses.
Next, we conrmed the results of our previous study [17] comparing the reinforcing effect of oxycodone among the different dosing conditions (Bup/Nx and oxycodone) by repeated-measures
ANOVA with a signicance level of alpha at 0.05. A Mann-Whitney
test was performed to compare the difference between percent
drug choice for placebo and each active oxycodone dose condition:
placebo vs 10, 20, 40 and 60 mg.
To investigate the risk of oxycodone self-administration, we
generated a variable called oxycodone preference, dened as
self-administration of 60% or more of the dose of oxycodone during
the choice sessions. Then we used a logit model based on GEE [40]
to identify variables associated with oxycodone preference. This
approach was used in order to study factors associated with oxycodone preference during laboratory sessions, while taking into account the correlation between repeated measures on the same
individuals [2]. We used an unstructured covariance matrix to
model the correlation between the repeated measures. Variables
with P values of <.20 in the bivariate analysis were considered eligible for the nal model, which was built using a backward procedure based on the log-likelihood ratio test.
In addition, we assessed the association between the 2 variables
that have pain dimensions, the pain score and the withdrawal
score measured respectively with the MPQ and the SOWS, and also
their possible interaction with oxycodone self-administration.
Then we performed 2 separate multivariate models to identify factors associated with oxycodone preference using rst MPQ score
and then SOWS score as explanatory variables.
All of the analyses were performed by SPSS software, version
18.0 (SPSS, Chicago, IL, USA) and Stata version 10.1 (StataCorp, College Station, TX, USA) for Windows software packages.
3. Results
During the enrollment phase of the study, 191 individuals were
assessed for eligibility and 140 were excluded because they did not
meet the inclusion criteria [(n = 78; medical exclusion (n = 21),
psychiatric exclusion (n = 10), not opioid abusing/dependent
(n = 29), other substance dependence (n = 13), or other (n = 5)] or
lost to follow-up (n = 62). Of the 51 who were randomized to the
trial, 8 discontinued before the inpatient phase [medical drop
(n = 2), not interested in treatment (n = 5) and lost to follow-up
(n = 1)]. Among the 43 participants who entered the inpatient
study, 12 dropped before the end of the study (2 for medical reasons, 2 for noncompliance, 7 for self-withdrawal and 1 for another
reason). Of the 31 participants who completed the 7-week inpatient laboratory period, 6 participants completed a pilot phase during which a different oxycodone dose range was tested, so we only
used data from the remaining 25 participants for our analyses. The
participants who were selected for the analyses were similar to the
others who were excluded regarding all the sociodemographic
variables. Data from a total of 25 participants were used in the
analyses, accounting for 375 observations. Of the 25 participants
selected for the analyses, the median (interquartile range [IQR])
age was 48 (4354) years, and 9 (36%) were women. Nine (36%)
of the participants were African American, 8 (32%) were Hispanic
and 8 (32%) were white. Reasons for study discontinuation among
the 18 participants who did not complete the 7-week study were:
psychiatric issues (n = 2), intolerance to environment (n = 2), intolerance to Bup/Nx (n = 2 for nausea and n = 1 for heavy sedation),
behavioral issues (n = 5), seeking treatment (n = 1) and lost to follow-up (n = 5).
The median (IQR) duration of prescription opioids use was 5
(28) years. Prescription opioids that were used during screening
included mainly oxycodone, hydrocodone, hydromorphone and
tramadol. The median (IQR) number of pills used per day was 7
(410), corresponding to 60 (38144) mg of morphine equivalents
(Table 1). At the screening visit, 9 (36%) of the participants had urine samples positive for cocaine. Only one participant reported
using heroin during the previous month. Among the 21 participants who completed the McGill Pain Questionnaire at baseline,
the median (IQR) pain score at the screening visit was 38
(3050). The median (IQR) MPQ score under Bup/Nx maintenance
for the 25 participants accounting for 375 time points was 21
(1531). MPQ score signicantly decreased between baseline and
laboratory sessions (coefcient 15.88, 95% CI 18.67 to 13.10,
P < .001), suggesting that Bup/Nx had a positive impact on pain
score relative to prestudy pain ratings. Moreover, compared to
2 mg Bup/Nx, 8 mg Bup/Nx signicantly decreased MPQ pain score
(OR 1.68, 95% CI 3.15 to 0.21; P = .03) and even more under
the 16 mg Bup/Nx dose condition (OR 2.74, 95% CI 4.21 to
1.27); P < .001).
Consistent with our previous ndings [17], oxycodone was not
self-administered above placebo levels at any dose tested; nor did
it vary as a function of Bup/Nx maintenance dose condition (Fig. 2).
Mann-Whitney tests revealed no signicant differences (P > .05)
between oxycodone placebo and active oxycodone doses under
the 3 Bup/Nx doses (2/0.5, 8/2 and 16/4 mg). Although oxycodone
produced minimal reinforcing effects overall, during 41 (11%) of
the sessions, participants chose more than 60% of the available
dose of oxycodone, accounting for 9 (36%) participants. Withdrawal symptoms were reported in 307 (83%) sessions with a median (IQR) score of 4 (19) out of a maximum possible score of 64.
The results of the bivariate analysis are presented in Table 1. We
excluded from the analyses the variables found in fewer than 10%
of the participants, such as self-reported heroin use (1 participant),
self-reported cannabis use (1 participant), positive cannabis toxicology (2 participants) and positive methadone toxicology (2 participants). The variables eligible for the nal model are indicated
in Table 1. Some baseline variables were found to be associated
with oxycodone preference, such as being white, reporting use of
a higher number of prescription opioids pills per day, using more
morphine equivalents per day and having a urine sample positive
for benzodiazepines at the baseline interview. Focusing on the laboratory session dependent variables, the results revealed that the
highest Bup/Nx maintenance dose (16/4 mg) was associated with
less oxycodone preference. Furthermore, higher pain scores using
the MPQ were associated with greater oxycodone preference.
1445
P. Roux et al. / PAIN 154 (2013) 14421448
Table 1
Factors associated with oxycodone self-administration in a sample of opioid-dependent pain participants under buprenorphine maintenance during laboratory drug choice
sessions.a
Characteristic
Age, y
Gender
Female
Male
Years of educationb
Ethnicity
African American
White
Hispanic
Percentage of participants employed
Prescription opioids pills/d c,d
Morphine equivalents, mg/d c
Duration of prescription opioids use, y
Benzodiazepine-positive urine
Methadone-positive urine e
Cannabis-positive urine c,e
Cocaine-positive urine c
Self-reported heroin use c,e
Self-reported cocaine use c
Self-reported alcohol use c,f
Buprenorphine/naloxone dose
2/0.5 mg (ref)
8/2 mg
16/4 mg
Oxycodone dose (prescription opioids)
0 mg (ref)
10 mg
20 mg
40 mg
60 mg
MPQ score g,h
SOWS score g,h
Value
No. of participants
48 (4354)
135 (36)
240 (64)
13 (1214)
9
16
135 (36)
120 (32)
120 (32)
75 (20)
7 (410)
60 (38144)
5 (28)
60 (16)
30 (8)
30 (10)
69 (14)
15 (4)
45 (12)
0 (04)
9
8
8
5
4
2
2
9
1
3
Bivariate analysis
OR (95% CI)
P value
1.02 (0.911.15)
.71
.54
1
1.84 (0.2613.08)
0.81 (0.551.19)
.29
1
4.76
0.36
0.28
1.09
1.23
1.03
4.76
0.74
1.57
0.83
.08
.49
.42
.04
.08
.76
.13
.77
.14
.84
.08
.65
.31
(0.8327.2)
(0.026.72)
(0.016.46)
(1.001.19)
(0.971.55)
(0.861.23)
(0.6335.9)
(0.0414.78)
(0.2211.14)
(0.571.20)
125 (33)
125 (33)
125 (33)
25
25
25
1
0.79 (0.471.30)
0.38 (0.190.77)
.35
.007
75 (20)
75 (20)
75 (20)
75 (20)
75 (20)
21 (1531)
4 (19)
25
25
25
25
25
1
1.32
1.36
1.00
1.54
1.30
1.89
.46
.42
1.00
.26
.003
.003
(0.632.81)
(0.642.89)
(0.462.19)
(0.733.23)
(1.091.55)
(1.242.86)
SA, self-administration of more than 60% of the total dose of oxycodone (drug break point >1200); OR, odds ratio; CI, condence interval; MPQ, McGill Pain Questionnaire;
SOWS, Subjective Opioid Withdrawal Scale.
a
Logistic regression model based on generalized estimating equations: bivariate analysis (n = 25 participants, 375 observations). Data are expressed as no. of sessions (%) or
median (interquartile range).
b
For each grade year increase.
c
At the consent visit before entering the study (baseline visit).
d
For 100 point-increase.
e
Not included in the nal model; number of subjects <10%.
f
Number of drinks per month.
g
One hour after the rst daily dose of buprenorphine/naloxone.
h
For each 10-unit increase.
In addition, higher total SOWS scores were associated with oxycodone preference.
The results presented in Table 2 show 2 different models after
multiple adjustments, one including a withdrawal symptoms variable and the other a pain variable. Both models in Table 2 demonstrate that 2 variables remained associated with oxycodone
preference, Bup/Nx dose and withdrawal/pain variable. When
participants were maintained on the highest dose of Bup/Nx
(16/4 mg), they had a 2-fold lower risk of preferring oxycodone.
Furthermore, those who reported more withdrawal symptoms
(OR 1.96, 95% CI 1.293.00, P = .002) and those who had higher
pain scores (OR 1.59, 95% CI 1.152.20, P = .005) were more likely
to self-administer oxycodone. Interestingly, the number of morphine equivalents used per day at baseline did not remain signicantly associated with the outcome. No signicant interaction was
found between MPQ pain score and SOWS score regarding oxycodone preference (P = .44). However, we found a signicant association between MPQ pain score and SOWS score (OR 0.85, 95% CI
0.631.07, P < .001).
4. Discussion
Overall, the data indicate that Bup/Nx-maintained opioiddependent patients with pain do not robustly self-administer
oxycodone, as described in our previous article [17]. This result is
consistent with the conclusions drawn in a major article on buprenorphine pharmacology that demonstrated that the effects of
morphine, a full l-receptor selective opioid agonist similar to
oxycodone [30], were signicantly attenuated when provided to
participants during the time of chronic buprenorphine administration [14].
In addition, Bup/Nx maintenance demonstrated a positive impact on pain in our participants, with greater analgesic effects under maintenance on higher doses of Bup/Nx, especially with
16/4 mg. These ndings are of great interest because they demonstrate the potential utility of Bup/Nx for treating pain in populations who also abuse prescription opioids. Indeed, buprenorphine
is known as an effective opioid maintenance treatment for opioid
dependence, and studies have previously demonstrated that buprenorphine is effective in relieving pain in nondependent and opioid-dependent individuals under certain conditions. For example,
a recent randomized study in adults with acute pain demonstrated
that sublingual buprenorphine is as effective at reducing pain as
intravenous morphine [13]. In a sample of opioid-naive patients
with chronic back pain, another randomized trial demonstrated
the efcacy of transdermal buprenorphine in reducing pain [34].
Another article reviewed the treatment options for opioid dependence and pain and mentioned the possibility of using the partial
1446
P. Roux et al. / PAIN 154 (2013) 14421448
Mann-Whitney tests:
- placebo versus 10mg Oxy : Bup/Nx 2/0.5 mg p=0.54 ; Bup/Nx 8/2 mg p=0.71;
Bup/Nx 16/4 mg p=0.29)
- placebo versus 20mg Oxy : Bup/Nx 2/0.5 mg p=0.29 ; Bup/Nx 8/2 mg p=0.74;
Bup/Nx 16/4 mg p=0.14)
- placebo versus 40mg Oxy : Bup/Nx 2/0.5 mg p=0.59 ; Bup/Nx 8/2 mg p=0.97;
Bup/Nx 16/4 mg p=0.47)
- placebo versus 60mg Oxy : Bup/Nx 2/0.5 mg p=0.18 ; Bup/Nx 8/2 mg p=0.36;
Bup/Nx 16/4 mg p=0.37)
Fig. 2. Percentage of the sampled oxycodone dose chosen under each Bup/Nx maintenance dose. Mann-Whitney tests: placebo vs 10 mg Oxy: Bup/Nx 2/0.5 mg, P = .54; Bup/
Nx 8/2 mg, P = .71; Bup/Nx 16/4 mg, P = .29; placebo vs 20 mg Oxy: Bup/Nx 2/0.5 mg, P = .29; Bup/Nx 8/2 mg, P = .74; Bup/Nx 16/4 mg, P = .14; placebo vs 40 mg Oxy: Bup/Nx
2/0.5 mg, P = .59; Bup/Nx 8/2 mg, P = .97; Bup/Nx 16/4 mg, P = .47; placebo vs 60 mg Oxy: Bup/Nx 2/0.5 mg, P = .18; Bup/Nx 8/2 mg, P = .36; Bup/Nx 16/4 mg, P = .37. Oxy,
oxycodone; Bup, buprenorphine; Nx, naloxone.
Table 2
Factors associated with oxycodone self-administration.a
Model
Model 1: Withdrawal symptoms (SOWS score)
Model 2: Pain score (MPQ score)
Dose and score
Multivariate analysis
Buprenorphine/naloxone dose
2/0.5 mg (ref)
8/2 mg
16/4 mg
SOWS score b,c
Buprenorphine/naloxone dose
2/0.5 mg (ref)
8/2 mg
16/4 mg
MPQ score b,c
aOR (95%CI)
P value
1
0.84 (0.501.42)
0.41 (0.200.82)
1.96 (1.293.00)
.002
1
0.75 (0.421.33)
0.42 (0.200.90)
1.59 (1.152.20)
.47
.03
.005
aOR, adjusted odds ratio; CI, condence interval; SOWS, Subjective Opioid Withdrawal Scale; MPQ, McGill Pain Questionnaire.
a
Multivariate logistic regression model based on generalized estimating equations. Multivariate analysis of 25 subjects, 369 observations; only sessions with complete
data for all variables were included in the nal model.
b
One hour after the rst daily dose of buprenorphine/naloxone.
c
For each 10-unit increase.
l-agonist buprenorphine in the management of concurrent pain
and opioid addiction [23]. In addition, a recent randomized study
demonstrated that in patients with chronic pain, there is a similar
analgesic and tolerability prole when patients are converted from
hydrocodone/acetaminophen (Vicodin) to transdermal buprenorphine [31]. Finally, a retrospective study reported positive results
in pain reduction with Bup/Nx to treat co-occurring chronic noncancer pain and opioid dependence in a primary care setting
[28]. Data from our study suggest that among patients with cooccurring pain and opioid abuse, higher doses of buprenorphine
are more effective than lower doses for simultaneously controlling
both pain and opioid abuse.
To present our ndings in a more public health approach, we
performed further analyses regarding oxycodone preference.
Although our ndings suggest that buprenorphine may be effective
in reducing the reinforcing effects of oxycodone among patients
with chronic pain who abuse their prescription opioids, an
in-depth analysis of the correlates of preference for oxycodone under Bup/Nx maintenance seemed to be important to identify some
potential factors associated with the abuse liability of oxycodone
in this population. After adjusting the model aimed at identifying
factors associated with oxycodone preference, no variable related
to participant baseline characteristics remained associated with
oxycodone preference. It is interesting to note that participants
who reported using a higher number of morphine equivalents before entering the study were not more likely to self-administer
oxycodone. This result suggests that Bup/Nx is effective in reducing
the abuse liability of oxycodone regardless of level of prescription
opioids use. However, caution is required because our participants
were selected according to specic inclusion criteria that could
have selected less heavy prescription opioids users.
It is known that specic subpopulations might be at risk of
abusing prescription opioids such as oxycodone [29]. Our bivariate
analysis demonstrated that benzodiazepine users, cannabis users
P. Roux et al. / PAIN 154 (2013) 14421448
and those who endorsed using heroin at baseline were more likely
to self-administer oxycodone. These ndings highlight the importance of identifying patients at higher risk of opioid misuse or
addiction as recommended by the Canadian guideline for safe
and effective use of opioids for chronic pain [19], as well as circumstances under which opioid misuse is more likely. Interestingly, the
variables that remained associated with the oxycodone selfadministration were pain score, withdrawal symptoms, and Bup/
Nx dose. The results suggested that pain assessment remains a
key component to adapt the pharmacological treatment. In this
study, higher pain scores and higher withdrawal symptoms scores
were associated with more oxycodone self-administration. It has
been previously demonstrated that nonmedical use of prescription
opioids was associated with bodily pain in a population of injecting
drug users [21]. Moreover, withdrawal symptoms have been found
to be a predictor of nonmedical use of opioids during opioid maintenance treatment [32]. Our results highlight the difculty in differentiating pain from withdrawal and from a chronic pain
condition. For this reason, several studies have noted the importance of including opioid withdrawal assessments in the management of pain, especially in an opioid-dependent population
[35,37].
In addition, our ndings demonstrated that buprenorphine
maintenance dose could play a role in oxycodone preference. It is
known that suboptimal doses of buprenorphine may have negative
consequences on treatment efcacy [24]. Because dose adjustment
depends on multiple pharmacokinetic and pharmacodynamic
parameters, characterized by interindividual differences, it is crucial to measure patient perceptions regarding the effectiveness of
buprenorphine in alleviating withdrawal symptoms and pain.
One strategy for treating chronic pain is to administer opioid doses
in an escalating manner. Naliboff and colleagues used this method
and demonstrated some improvements in self-reported acute relief
from pain without an increase in supplemental opioid use compared to a stable dose strategy [27]. Previous studies have found
that the analgesic effects of buprenorphine could be improved by
increasing the dose or by prescribing daily divided doses [22]
and that doses of 32 mg or higher could be safe and effective
[15]. Indeed, the therapeutic choice of using a partial l-agonist
medication is known to reduce the risk of overdoses compared to
other full agonist analgesics [3]. A previous study found that buprenorphine causes limited respiratory depression with a ceiling effect at higher dose [6].
Although use of a within-subjects, repeated-measures design in
participants housed on an inpatient research unit is strong experimentally, some limitations also must be acknowledged. First, the
participants we enrolled had to fulll several inclusion and exclusion criteria, which may have reduced the external validity of the
results. Indeed, our sample excluded the most severely opioid
dependent individuals and those with certain types of pain (eg,
those with neuropathic pain, multiple failed back surgeries, as well
as those with a previous lack of analgesic response to buprenorphine). Thus, further studies are needed in opioid dependent individuals with other types of pain and a more complex addictive
prole. Moreover, the highest dose we tested (16 mg) may be considered as inadequate for some patients and may need to be increased to manage both pain and opioid dependence. Also,
because oxycodone is commonly abused by the intranasal or intravenous routes, future studies should test higher doses of Bup/Nx in
combination with opioid agonists administered by nonoral routes.
Finally, the context of laboratory sessions is different from the ecological context of the participants lives. Therefore, a clinical trial
conducted on an outpatient basis and/or an epidemiological study
would be appropriate to conrm the present results [36].
Nevertheless, our data suggest that buprenorphine could be an
effective medication for those patients who need to be treated for
1447
chronic pain and opioid abuse/dependence. Further studies should
be implemented to determine the prole of pain patients most
likely to abuse prescription opioids.
Conict of interest
The authors declare that over the past 3 years SDC, MAS, SKV,
ZDC, JMM, SM and JDJ have received compensation (in the form
of partial salary support) from investigator-initiated studies supported by Reckitt-Benckiser Pharmaceuticals, Schering-Plough Corporation, Johnson & Johnson Pharmaceutical Research &
Development, Endo Pharmaceuticals, and MediciNova. In addition,
SDC has served as a consultant to the following companies: Analgesic Solutions, BioDelivery Sciences, Cephalon, Grunenthal,
Inexxion, King, Neuromed, Pzer, Salix and Shire. PR, JC and LF
have no conicts to report.
Acknowledgments
We gratefully acknowledge the support of the National Institute
on Drug Abuse in the form of research grants to MAS (R01
DA20448), SDC (R01 DA16759), and HDK (P50 DA09236). In addition, we thank Robert Vorel, MD, Ben Bryan, MD, Elias Dakwar, MD,
David Mysels, MD, Janet Murray, RN, and Claudia Tindall, RN, for
their medical assistance, as well as Elisa Payne BA, Debra Wolkenfeld, BA, Phillip Saccone, BA, and Joseph Lazar, BA, for their technical assistance.
References
[1] Becker WC, Sullivan LE, Tetrault JM, Desai RA, Fiellin DA. Non-medical use,
abuse and dependence on prescription opioids among US adults: psychiatric,
medical and substance use correlates. Drug Alcohol Depend 2008;94:3847.
[2] Carlin JB, Wolfe R, Coffey C, Patton GC. Analysis of binary outcomes in
longitudinal studies using weighted estimating equations and discrete-time
survival methods: prevalence and incidence of smoking in an adolescent
cohort. Stat Med 1999;18:265579.
[3] Carrieri MP, Amass L, Lucas GM, Vlahov D, Wodak A, Woody GE.
Buprenorphine use: the international experience. Clin Infect Dis
2006;43:S197215.
[4] Control Centers for Disease and Prevention (CDC). CDC grand rounds:
prescription drug overdosesa US epidemic. MMWR Morb Mortal Wkly Rep
2012;61:103.
[5] Cicero TJ, Inciardi JA, Munoz A. Trends in abuse of Oxycontin and other opioid
analgesics in the United States, 20022004. J Pain 2005;6:66272.
[6] Dahan A. Opioid-induced respiratory effects: new data on buprenorphine.
Palliat Med 2006;20:s38.
[7] Gilson AM. The concept of addiction in law and regulatory policy related to
pain management: a critical review. Clin J Pain 2010;26:707.
[8] Gilson AM, Kreis PG. The burden of the nonmedical use of prescription opioid
analgesics. Pain Med 2009;10:S89S100.
[9] Gossop M. The development of a Short Opiate Withdrawal Scale (SOWS).
Addict Behav 1990;15:48790.
[10] Green TC, Grau LE, Carver HW, Kinzly M, Heimer R. Epidemiologic trends and
geographic patterns of fatal opioid intoxications in Connecticut, USA, 1997
2007. Drug Alcohol Depend 2011;115:2218.
[11] Hall AJ, Logan JE, Toblin RL, Kaplan JA, Kraner JC, Bixler D, Crosby AE, Paulozzi
LJ. Patterns of abuse among unintentional pharmaceutical overdose fatalities.
JAMA 2008;300:261320.
[12] Huxtable CA, Roberts LJ, Somogyi AA, MacIntyre PE. Acute pain management in
opioid-tolerant patients: a growing challenge. Anaesth Intensive Care
2011;39:80423.
[13] Jalili M, Fathi M, Moradi-Lakeh M, Zehtabchi S. Sublingual buprenorphine in
acute pain management: a double-blind randomized clinical trial. Ann Emerg
Med 2011;59:27680.
[14] Jasinski DR, Pevnick JS, Grifth JD. Human pharmacology and abuse potential
of the analgesic buprenorphine: a potential agent for treating narcotic
addiction. Arch Gen Psychiatry 1978;35:50116.
[15] Johnson RE, Chutuape MA, Strain EC, Walsh SL, Stitzer ML, Bigelow GE. A
comparison of levomethadyl acetate, buprenorphine, and methadone for
opioid dependence. N Engl J Med 2000;343:12907.
[16] Johnson RE, Fudala PJ, Payne R. Buprenorphine: considerations for pain
management. J Pain Symptom Manage 2005;29:297326.
[17] Jones JD, Sullivan MA, Manubay J, Vosburg SK, Comer SD. The subjective,
reinforcing, and analgesic effects of oxycodone in patients with chronic, nonmalignant pain who are maintained on sublingual buprenorphine/naloxone.
Neuropsychopharmacology 2011;36:41122.
1448
P. Roux et al. / PAIN 154 (2013) 14421448
[18] Kahan M, Srivastava A, Ordean A, Cirone S. Buprenorphine: new treatment of
opioid addiction in primary care. Can Fam Physician 2011;57:2819.
[19] Kahan M, Wilson L, Mailis-Gagnon A, Srivastava A. Canadian guideline for safe
and effective use of opioids for chronic noncancer pain: clinical summary for
family physicians. Part 2: special populations. Can Fam Physician
2011;57:126976. e41928.
[20] Keller CE, Ashraoun L, Neumann AM, Van Klein J, Fox CH, Blondell RD.
Practices, perceptions, and concerns of primary care physicians about opioid
dependence associated with the treatment of chronic pain. Subst Abus
2012;33:10313.
[21] Khosla N, Juon HS, Kirk GD, Astemborski J, Mehta SH. Correlates of nonmedical prescription drug use among a cohort of injection drug users in
Baltimore City. Addict Behav 2011;36:12827.
[22] Kogel B, Christoph T, Strassburger W, Friderichs E. Interaction of mu-opioid
receptor agonists and antagonists with the analgesic effect of buprenorphine
in mice. Eur J Pain 2005;9:599611.
[23] Ling W, Mooney L, Hillhouse M. Prescription opioid abuse, pain and addiction:
clinical issues and implications. Drug Alcohol Rev 2011;30:3005.
[24] Ling W, Wesson DR, Charuvastra C, Klett CJ. A controlled trial comparing
buprenorphine and methadone maintenance in opioid dependence. Arch Gen
Psychiatry 1996;53:4017.
[25] Manchikanti L, Fellows B, Ailinani H, Pampati V. Therapeutic use, abuse, and
nonmedical use of opioids: a ten-year perspective. Pain Physician
2010;13:40135.
[26] Melzack R. The short-form McGill Pain Questionnaire. PAIN 1987;30:1917.
[27] Naliboff BD, Wu SM, Schieffer B, Bolus R, Pham Q, Baria A, Aragaki D, Van Vort
W, Davis F, Shekelle P. A randomized trial of 2 prescription strategies for
opioid treatment of chronic nonmalignant pain. J Pain 2011;12:28896.
[28] Pade PA, Cardon KE, Hoffman RM, Geppert CM. Prescription opioid abuse,
chronic pain, and primary care: a co-occurring disorders clinic in the chronic
disease model. J Subst Abuse Treat 2012;43:44650.
[29] Pergolizzi Jr JV, Gharibo C, Passik S, Labhsetwar S, Taylor Jr R, Pergolizzi JS,
Muller-Schwefe G. Dynamic risk factors in the misuse of opioid analgesics. J
Psychosom Res 2012;72:44351.
[30] Poyhia R, Vainio A, Kalso E. A review of oxycodones clinical pharmacokinetics
and pharmacodynamics. J Pain Symptom Manage 1993;8:637.
[31] Ripa SR, McCarberg BH, Munera C, Wen W, Landau CJ. A randomized, 14-day,
double-blind study evaluating conversion from hydrocodone/acetaminophen
(Vicodin) to buprenorphine transdermal system 10 mug/h or 20 mug/h in
patients
with
osteoarthritis
pain.
Expert
Opin
Pharmacother
2012;13:122941.
[32] Roux P, Carrieri PM, Cohen J, Ravaux I, Spire B, Gossop M, Comer SD. Nonmedical use of opioids among HIV-infected opioid dependent individuals on
opioid maintenance treatment: the need for a more comprehensive approach.
Harm Reduct J 2011;8:31.
[33] Silversides A. Use of strong opioids in Canada rose sharply between 2005 and
2010. BMJ 2011;343:d5441.
[34] Steiner DJ, Sitar S, Wen W, Sawyerr G, Munera C, Ripa SR, Landau C. Efcacy
and safety of the seven-day buprenorphine transdermal system in opioidnaive patients with moderate to severe chronic low back pain: an enriched,
randomized, double-blind, placebo-controlled study. J Pain Symptom Manage
2011;42:90317.
[35] Townsend CO, Kerkvliet JL, Bruce BK, Rome JD, Hooten WM, Luedtke CA,
Hodgson JE. A longitudinal study of the efcacy of a comprehensive pain
rehabilitation program with opioid withdrawal: comparison of treatment
outcomes based on opioid use status at admission. PAIN 2008;140:17789.
[36] Turk DC, OConnor AB, Dworkin RH, Chaudhry A, Katz NP, Adams EH,
Brownstein JS, Comer SD, Dart R, Dasgupta N, Denisco RA, Klein M,
Leiderman DB, Lubran R, Rappaport BA, Zacny JP, Ahdieh H, Burke LB, Cowan
P, Jacobs P, Malamut R, Markman J, Michna E, Palmer P, Peirce-Sandner S,
Potter JS, Raja SN, Rauschkolb C, Roland CL, Webster LR, Weiss RD, Wolf K.
Research design considerations for clinical studies of abuse-deterrent opioid
analgesics: IMMPACT recommendations. PAIN 2012;153:19972008.
[37] Vowles KE, Ashworth J. Is opioid withdrawal necessary within comprehensive
pain rehabilitation programs? PAIN 2011;152:194850.
[38] Wesson DR, Smith DE. Buprenorphine in the treatment of opiate dependence. J
Psychoactive Drugs 2010;42:16175.
[39] Wolfert MZ, Gilson AM, Dahl JL, Cleary JF. Opioid analgesics for pain control:
Wisconsin physicians knowledge, beliefs, attitudes, and prescribing practices.
Pain Med 2010;11:42534.
[40] Zeger SL, Liang KY. Longitudinal data analysis for discrete and continuous
outcomes. Biometrics 1986;42:12130.
Você também pode gostar
- PerioperativeDocumento15 páginasPerioperativeJEFFERSON MUÑOZAinda não há avaliações
- Handbook of Methadone Prescribing and Buprenorphine TherapyNo EverandHandbook of Methadone Prescribing and Buprenorphine TherapyAinda não há avaliações
- Jurnalnantipsychotic PDFDocumento9 páginasJurnalnantipsychotic PDFamiaslimAinda não há avaliações
- Bupe and Acute PainDocumento14 páginasBupe and Acute PainHollowbinAinda não há avaliações
- Medical Journal of Australia - 2020 - Hopkins - Educating Junior Doctors and Pharmacists To Reduce Discharge Prescribing ofDocumento7 páginasMedical Journal of Australia - 2020 - Hopkins - Educating Junior Doctors and Pharmacists To Reduce Discharge Prescribing ofQurat ul ainAinda não há avaliações
- National Use of Prescription Medications For Insomnia: NHANES 1999-2010Documento10 páginasNational Use of Prescription Medications For Insomnia: NHANES 1999-2010Agustina ZairidaAinda não há avaliações
- Opioid Use Disorder and TreatmentDocumento5 páginasOpioid Use Disorder and TreatmentloloasbAinda não há avaliações
- Buvidal PDF 1158123740101Documento7 páginasBuvidal PDF 1158123740101riffs_ahoyAinda não há avaliações
- Guía de OpioidesDocumento13 páginasGuía de OpioidesJAIME MUÑOZAinda não há avaliações
- Peirano 2016Documento6 páginasPeirano 2016coloAinda não há avaliações
- Use of Methadone For Opioid Weaning in Children: Prescribing Practices and TrendsDocumento8 páginasUse of Methadone For Opioid Weaning in Children: Prescribing Practices and TrendsJessica VillanuevaAinda não há avaliações
- Opioid Epidemic in The United StatesDocumento30 páginasOpioid Epidemic in The United StatesAlkadir del AzizAinda não há avaliações
- NIH Public Access: Buprenorphine-Nalxone Therapy in Pain ManagementDocumento25 páginasNIH Public Access: Buprenorphine-Nalxone Therapy in Pain ManagementyoiiiAinda não há avaliações
- Article Shunkina ChukhrayDocumento10 páginasArticle Shunkina ChukhrayСофія ШунькінаAinda não há avaliações
- Music Intervensi PainDocumento5 páginasMusic Intervensi PainfitrikumalaAinda não há avaliações
- Perception and Practice Among Emergency Medicine Health Care Providers Regarding Discharging Patients After Opioid AdministrationDocumento15 páginasPerception and Practice Among Emergency Medicine Health Care Providers Regarding Discharging Patients After Opioid AdministrationSUPHAWICH KHOIKAEWAinda não há avaliações
- Medications To Treat Opioid Use Disorder Research ReportDocumento34 páginasMedications To Treat Opioid Use Disorder Research ReportBakthika KumarageAinda não há avaliações
- Actigraphic Measurements in Opioid Detoxification With Methadone or BuprenorphineDocumento8 páginasActigraphic Measurements in Opioid Detoxification With Methadone or BuprenorphineSaiber PanAinda não há avaliações
- Short-Acting Opioid Drug Class ReviewDocumento44 páginasShort-Acting Opioid Drug Class ReviewlabendetAinda não há avaliações
- Drug Utilization Patterns Using Who Core Prescribing Indicators in Different Out Patient Departments at Secondary Care Hospital, KarimnagarDocumento9 páginasDrug Utilization Patterns Using Who Core Prescribing Indicators in Different Out Patient Departments at Secondary Care Hospital, KarimnagarIJAR JOURNALAinda não há avaliações
- Carousel Restricted AccessDocumento12 páginasCarousel Restricted AccessJess BaerAinda não há avaliações
- Chronic Pain Patient and Anaesthesia: Review ArticleDocumento8 páginasChronic Pain Patient and Anaesthesia: Review ArticlewawanAinda não há avaliações
- Research Article: Risk Assessment of Opioid Misuse in Italian Patients With Chronic Noncancer PainDocumento10 páginasResearch Article: Risk Assessment of Opioid Misuse in Italian Patients With Chronic Noncancer PaintestenewAinda não há avaliações
- Effects of Ascending Buprenorphine Doses On Measur - 2019 - Journal of SubstanceDocumento7 páginasEffects of Ascending Buprenorphine Doses On Measur - 2019 - Journal of SubstanceARACELY RAFAELA CORNEJO MARTINEZAinda não há avaliações
- Joy R. Ross Et Al - Clinical Pharmacology and Pharmacotherapy of Opioid Switching in Cancer PatientsDocumento10 páginasJoy R. Ross Et Al - Clinical Pharmacology and Pharmacotherapy of Opioid Switching in Cancer PatientsBic0000Ainda não há avaliações
- Association of Levels of Opioid Use With Pain and Activity Interference Among Patients Initiating Chronic Opioid Therapy A Longitudinal Study PDFDocumento22 páginasAssociation of Levels of Opioid Use With Pain and Activity Interference Among Patients Initiating Chronic Opioid Therapy A Longitudinal Study PDFReztariestyadiAinda não há avaliações
- Buprenorphine Treatment For Young AdultsDocumento2 páginasBuprenorphine Treatment For Young AdultsGAinda não há avaliações
- Opioids and Chronic Neuropathic Pain: Kathleen M. Foley, M.DDocumento3 páginasOpioids and Chronic Neuropathic Pain: Kathleen M. Foley, M.DFionita BeelkisAinda não há avaliações
- Anesthesia Books 2019 Bonica's-2001-3000Documento1.000 páginasAnesthesia Books 2019 Bonica's-2001-3000rosangelaAinda não há avaliações
- Anesthesia Books 2019 Bonica's-2001-3000Documento845 páginasAnesthesia Books 2019 Bonica's-2001-3000rosangelaAinda não há avaliações
- B. DepressionDocumento7 páginasB. Depressionチャン フィラAinda não há avaliações
- Opioid-Induced Hyperalgesia in Patients After Surgery: A Systematic Review and A Meta-AnalysisDocumento14 páginasOpioid-Induced Hyperalgesia in Patients After Surgery: A Systematic Review and A Meta-AnalysisNatalia SantacruzAinda não há avaliações
- E1252 FullDocumento7 páginasE1252 FullwawanAinda não há avaliações
- Relapse in Outpatient Treatment For Marijuana Dependence: Brent A. Moore PH.D., Alan J. Budney, PH.DDocumento5 páginasRelapse in Outpatient Treatment For Marijuana Dependence: Brent A. Moore PH.D., Alan J. Budney, PH.DAntonio Tejero PocielloAinda não há avaliações
- Managing Opioid Withdrawal With BuprenorphineDocumento7 páginasManaging Opioid Withdrawal With BuprenorphineAgata KurniawatyAinda não há avaliações
- Turo J. Nurmikko Et Al - Sativex Successfully Treats Neuropathic Pain Characterised by Allodynia: A Randomised, Double-Blind, Placebo-Controlled Clinical TrialDocumento11 páginasTuro J. Nurmikko Et Al - Sativex Successfully Treats Neuropathic Pain Characterised by Allodynia: A Randomised, Double-Blind, Placebo-Controlled Clinical TrialPoloGreenAinda não há avaliações
- New Opioids: Ournal of Linical NcologyDocumento6 páginasNew Opioids: Ournal of Linical Ncologyjuan tAinda não há avaliações
- Prot Sap 000Documento20 páginasProt Sap 000QenquAinda não há avaliações
- Pain Acute Pain OpioidTolerantDocumento20 páginasPain Acute Pain OpioidTolerantRoy LizalAinda não há avaliações
- JPM 10 00027Documento9 páginasJPM 10 00027Kepompong KupukupuAinda não há avaliações
- Exploringthe Opioid Prescribing Practices of General Practitioners (GPS) in West and South Essex, UkDocumento13 páginasExploringthe Opioid Prescribing Practices of General Practitioners (GPS) in West and South Essex, UkIJAR JOURNALAinda não há avaliações
- Pharmacoepidemiology, Pharmacoeconomics,PharmacovigilanceNo EverandPharmacoepidemiology, Pharmacoeconomics,PharmacovigilanceNota: 3 de 5 estrelas3/5 (1)
- Painreports 2 E589Documento7 páginasPainreports 2 E589goliath.chad2000Ainda não há avaliações
- Guide Lines LTOTDocumento8 páginasGuide Lines LTOTSteve GreenAinda não há avaliações
- Prescription Painkillers: History, Pharmacology, and TreatmentNo EverandPrescription Painkillers: History, Pharmacology, and TreatmentAinda não há avaliações
- Cooper 2020Documento18 páginasCooper 2020Lohann MedeirosAinda não há avaliações
- Vitis Vini Fera Ratna UtamiDocumento21 páginasVitis Vini Fera Ratna UtamiTyo Aham JrAinda não há avaliações
- Perioperative Opioids, The Opioid Crisis, and The AnesthesiologistDocumento15 páginasPerioperative Opioids, The Opioid Crisis, and The AnesthesiologistlimantofloreenAinda não há avaliações
- Opioid Dependence Rationale For and Efficacy of ExDocumento6 páginasOpioid Dependence Rationale For and Efficacy of ExÁlvaro Raúl Conto MorenoAinda não há avaliações
- Per Op Multimodal AnalgesiaDocumento16 páginasPer Op Multimodal AnalgesiaSrinivas PingaliAinda não há avaliações
- 2reconsidering Opioid Therapy Mar 2013Documento6 páginas2reconsidering Opioid Therapy Mar 2013Anonymous 4txA8N8etAinda não há avaliações
- Counseling Plus Buprenorphine-Naloxone Maintenance Therapy For Opioid DependenceDocumento10 páginasCounseling Plus Buprenorphine-Naloxone Maintenance Therapy For Opioid DependenceIlham WahyuAinda não há avaliações
- Pain Management JournalDocumento7 páginasPain Management JournalPengurus Pusat HipgabiAinda não há avaliações
- Research: Cite This Article As: BMJ, Doi:10.1136/bmj.38881.382755.2f (Published 6 July 2006)Documento6 páginasResearch: Cite This Article As: BMJ, Doi:10.1136/bmj.38881.382755.2f (Published 6 July 2006)dr.cintaAinda não há avaliações
- Subjective Effectiveness of Ibogaine Treatment For Problematic Opioid Consumption: Short-And Long-Term Outcomes and Current Psychological FunctioningDocumento9 páginasSubjective Effectiveness of Ibogaine Treatment For Problematic Opioid Consumption: Short-And Long-Term Outcomes and Current Psychological FunctioningB. ZapoteAinda não há avaliações
- Acute Pain Management in Patients With Opioid ToleranceDocumento6 páginasAcute Pain Management in Patients With Opioid ToleranceHendrikus Surya Adhi PutraAinda não há avaliações
- Jurnal ApendiktomyDocumento6 páginasJurnal ApendiktomySiti HosimahAinda não há avaliações
- PDF TestDocumento15 páginasPDF TestJess BaerAinda não há avaliações
- Thesis Rough DraftDocumento42 páginasThesis Rough Draftapi-457509028100% (1)
- 2opioid and Alcohol - EditedDocumento4 páginas2opioid and Alcohol - EditedAugustus KiokoAinda não há avaliações
- K Opiods For Pain BupDocumento2 páginasK Opiods For Pain Bupandrew herringAinda não há avaliações
- Original Article: Serratus Plane Block: A Novel Ultrasound-Guided Thoracic Wall Nerve BlockDocumento7 páginasOriginal Article: Serratus Plane Block: A Novel Ultrasound-Guided Thoracic Wall Nerve Blockandrew herringAinda não há avaliações
- Highland Guidelines - LDK 2013-2-15Documento3 páginasHighland Guidelines - LDK 2013-2-15andrew herringAinda não há avaliações
- Buprenorphine 2016-7-18Documento1 páginaBuprenorphine 2016-7-18andrew herringAinda não há avaliações
- Rib Fracture SerratusDocumento3 páginasRib Fracture Serratusandrew herring100% (1)
- Intravenous Lidocaine To Treat Postoperative Pain : Lidocaína Intravenosa No Tratamento Da Dor Pós-OperatóriaDocumento6 páginasIntravenous Lidocaine To Treat Postoperative Pain : Lidocaína Intravenosa No Tratamento Da Dor Pós-Operatóriaandrew herringAinda não há avaliações
- Rib Fracture W: PecsDocumento8 páginasRib Fracture W: Pecsandrew herring100% (1)
- Ultrasound-Guided Block of TheDocumento6 páginasUltrasound-Guided Block of Theandrew herringAinda não há avaliações
- Herring ED Intractable Abdominal pain-N+VDocumento1 páginaHerring ED Intractable Abdominal pain-N+Vandrew herringAinda não há avaliações
- 3.2.16.highland Emergency Medicine Journal Club Methods.2Documento2 páginas3.2.16.highland Emergency Medicine Journal Club Methods.2andrew herringAinda não há avaliações
- Ketamine As Rescue Treatment FDocumento8 páginasKetamine As Rescue Treatment Fandrew herringAinda não há avaliações
- Dex For Dental Pain & SwellingDocumento7 páginasDex For Dental Pain & Swellingandrew herring100% (1)
- Nej MR A 1511480Documento9 páginasNej MR A 1511480Tohari Masidi AminAinda não há avaliações
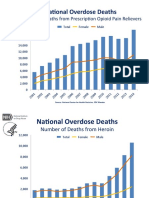
- OPR - Heroin DeathsDocumento2 páginasOPR - Heroin Deathsandrew herringAinda não há avaliações
- CVS Action Plan v2Documento1 páginaCVS Action Plan v2andrew herringAinda não há avaliações
- Ultrasound Guided Interventional Procedures In.13Documento14 páginasUltrasound Guided Interventional Procedures In.13andrew herring100% (1)
- Ultrasound Guided Interventional Procedures In.4Documento13 páginasUltrasound Guided Interventional Procedures In.4andrew herringAinda não há avaliações
- T Wycross 1984Documento6 páginasT Wycross 1984Ha HaAinda não há avaliações
- Opioid Pharmacology and Pharmaco Genetics DraftDocumento18 páginasOpioid Pharmacology and Pharmaco Genetics DraftPrudhvi Sai NidadavoluAinda não há avaliações
- ###Tutorial Work 217 Questions and Answers Mcqs PDFDocumento32 páginas###Tutorial Work 217 Questions and Answers Mcqs PDFDave DMAinda não há avaliações
- PethidineDocumento9 páginasPethidineAnonymous VZtcCECtAinda não há avaliações
- Psychoactive DrugsDocumento14 páginasPsychoactive DrugspoopooAinda não há avaliações
- Chapter 4 Dangerous DrugsDocumento14 páginasChapter 4 Dangerous DrugsMary Joyce Anne ColloAinda não há avaliações
- Pharmacology Online 2011Documento11 páginasPharmacology Online 2011evanyllaAinda não há avaliações
- Drugpowerpoint Without Date 2Documento35 páginasDrugpowerpoint Without Date 2THE TRIBEAinda não há avaliações
- Intensive & Critical Care NursingDocumento10 páginasIntensive & Critical Care NursingDwi Utari JAinda não há avaliações
- ... My Reflection On Action (Leadershinp and Management)Documento15 páginas... My Reflection On Action (Leadershinp and Management)Yasmeen zulfiqarAinda não há avaliações
- List of Narcotic DrugsDocumento24 páginasList of Narcotic DrugshmtlionAinda não há avaliações
- Therapeutics: A Benjamin Srivastava, John J Mariani, Frances R LevinDocumento11 páginasTherapeutics: A Benjamin Srivastava, John J Mariani, Frances R LevinPsiquiatria 2021-2Ainda não há avaliações
- A Rapid, High-Yield Conversion of Codeine To MorphineDocumento2 páginasA Rapid, High-Yield Conversion of Codeine To Morphinegeovani250% (2)
- Opioid AnalgesicsDocumento41 páginasOpioid AnalgesicsSayan NagAinda não há avaliações
- Somali Essential Medicines List 2019Documento36 páginasSomali Essential Medicines List 2019Usama MohaAinda não há avaliações
- Forensic Science International: Frank Musshoff, Ulrike M. Stamer, Burkhard MadeaDocumento10 páginasForensic Science International: Frank Musshoff, Ulrike M. Stamer, Burkhard MadeaFakhry FathaniyAinda não há avaliações
- Opium Poisoning: Pooja Yadav 19M6805 Roll - No:14Documento20 páginasOpium Poisoning: Pooja Yadav 19M6805 Roll - No:14Pooja YadavAinda não há avaliações
- Cough RemediesDocumento14 páginasCough RemediesLejla SisicAinda não há avaliações
- High Alert MedicationsDocumento2 páginasHigh Alert Medicationsshajahanputhusseri100% (2)
- Morphine (Astramorph)Documento1 páginaMorphine (Astramorph)Adrianne BazoAinda não há avaliações
- Acute Post-Op Pain ManagementDocumento4 páginasAcute Post-Op Pain Management1234chocoAinda não há avaliações
- Narcotic Analgesics - Notes - SY B. PharmaDocumento12 páginasNarcotic Analgesics - Notes - SY B. PharmaSneha charakAinda não há avaliações
- Drugs ORDocumento17 páginasDrugs ORCatherine ZamoraAinda não há avaliações
- Compilation of Forensic Science and Toxicology (Agadtantra Vyavaharayrved Evm Vidhivaidyak)Documento84 páginasCompilation of Forensic Science and Toxicology (Agadtantra Vyavaharayrved Evm Vidhivaidyak)Jagdish Bhandge100% (1)
- Dangerous DrugsDocumento48 páginasDangerous DrugsNigel HopeAinda não há avaliações
- Inj Damage From Krokodil in Eurasia PDFDocumento10 páginasInj Damage From Krokodil in Eurasia PDFSvetlozar NikolovAinda não há avaliações
- Savage Worlds - For King and CountryDocumento29 páginasSavage Worlds - For King and CountryShannon Russell (necron99)Ainda não há avaliações
- Ketamine (INN) : Update Review ReportDocumento46 páginasKetamine (INN) : Update Review ReportJeremia AnkesaAinda não há avaliações
- Definition of TermsDocumento8 páginasDefinition of TermsJoshua D None-NoneAinda não há avaliações
- Persiapan To 4 Utbk EngDocumento5 páginasPersiapan To 4 Utbk EngNavia SyafaAinda não há avaliações