Escolar Documentos
Profissional Documentos
Cultura Documentos
No.28 and 29
Enviado por
Louie Shaolin Lungao0 notas0% acharam este documento útil (0 voto)
5 visualizações2 páginasm
Título original
no.28-and-29
Direitos autorais
© © All Rights Reserved
Formatos disponíveis
DOCX, PDF, TXT ou leia online no Scribd
Compartilhar este documento
Compartilhar ou incorporar documento
Você considera este documento útil?
Este conteúdo é inapropriado?
Denunciar este documentom
Direitos autorais:
© All Rights Reserved
Formatos disponíveis
Baixe no formato DOCX, PDF, TXT ou leia online no Scribd
0 notas0% acharam este documento útil (0 voto)
5 visualizações2 páginasNo.28 and 29
Enviado por
Louie Shaolin Lungaom
Direitos autorais:
© All Rights Reserved
Formatos disponíveis
Baixe no formato DOCX, PDF, TXT ou leia online no Scribd
Você está na página 1de 2
The data in the table have been obtained on the decomposition of gaseous reactant
A in a constant volume batch reactor at 100 degrees Celsius. The stoichiometry of
the reaction is 2A ------R + S. What size plug flow reactor(in liters) operating at 100
degrees Celsius and 1 atm can treat 100 mol A/hr in a feed consisting of 20% inert
to obtain 95% conversion of A?
t,sec PA, atm
0 1.00
20 0.80
40 0.68
60 0.56
80 0.45
100 0.37
140 0.25
200 0.14
260 0.08
330 0.04
420 0.02
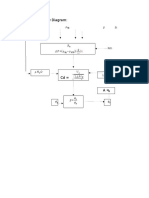
Given: CVBR
100mol/hr
20% inert
100 C 1 atm
Xa=0.9
Required: V(L) of plug flow reactor
Solution:
CVBR
Assume first order reaction
kt = -In(1-XA)
A= 0
Fao= 100(100/80)
Pao=0.8 atm
K=
0.011 0.009 0.009 0.01 0.009 0.009 0.009 0.009 0.009 0.009
2 6 7 9 9 8 7 8 3
K= 0.01/sec
In plug flow reactor
V/Vo= -In(1-Xa)/k
Vo=(FAoRT)/Pao
= (100)(0.08205)(373.15)/0.8
= 3827.12 L/hr
= 1.06 L/sec
V= (1.06)(-In(1-0.95))/0.01
= 317.55L
for plug flow reactor
XA
dX A
v =F ao
0
r A
-ra= kca
=k Ca0 (1-x)
dX A
kcao ( 1x )
XA
v=F ao
0
XA
vo dX A
v=
k 0 (1x)
vo
v= ( ln ( 1x ) )
v
*evaluated from 0 and 0.95
Você também pode gostar
- Chapter 3 (Seborg Et Al.)Documento20 páginasChapter 3 (Seborg Et Al.)Jamel CayabyabAinda não há avaliações
- No 28Documento2 páginasNo 28Louie Shaolin LungaoAinda não há avaliações
- Chapter IV and VDocumento2 páginasChapter IV and VLouie Shaolin LungaoAinda não há avaliações
- Ilocanos: "Kuripot O Matipid?" #Ilocandia: Submitted ToDocumento21 páginasIlocanos: "Kuripot O Matipid?" #Ilocandia: Submitted ToLouie Shaolin LungaoAinda não há avaliações
- IntroductionDocumento3 páginasIntroductionLouie Shaolin LungaoAinda não há avaliações
- The Laplace TransformsDocumento61 páginasThe Laplace TransformsLouie Shaolin Lungao100% (1)
- Information Flow DiagramDocumento1 páginaInformation Flow DiagramLouie Shaolin LungaoAinda não há avaliações
- Chapter IV and V 2Documento2 páginasChapter IV and V 2Louie Shaolin LungaoAinda não há avaliações
- Expt 05 ReportDocumento18 páginasExpt 05 ReportLouie Shaolin LungaoAinda não há avaliações
- VenturimeterDocumento8 páginasVenturimeterAbhishek Kandoi100% (1)
- Interpretation of Results and ObservationsDocumento1 páginaInterpretation of Results and ObservationsLouie Shaolin LungaoAinda não há avaliações
- Chapter 2. Expt IronDocumento1 páginaChapter 2. Expt IronLouie Shaolin LungaoAinda não há avaliações
- Case Study ChernobylDocumento4 páginasCase Study ChernobylLouie Shaolin LungaoAinda não há avaliações
- Expt 05 ReportDocumento18 páginasExpt 05 ReportLouie Shaolin LungaoAinda não há avaliações
- JHVJHDocumento1 páginaJHVJHLouie Shaolin LungaoAinda não há avaliações
- Information Flow DiagramDocumento1 páginaInformation Flow DiagramLouie Shaolin LungaoAinda não há avaliações
- Chapter 4 Expt 4Documento4 páginasChapter 4 Expt 4Louie Shaolin LungaoAinda não há avaliações
- Information Flow DiagramDocumento1 páginaInformation Flow DiagramLouie Shaolin LungaoAinda não há avaliações
- Example PolymerDocumento2 páginasExample PolymerLouie Shaolin LungaoAinda não há avaliações
- Experiment 1 REPORTDocumento18 páginasExperiment 1 REPORTLouie Shaolin LungaoAinda não há avaliações
- No 28Documento2 páginasNo 28Louie Shaolin LungaoAinda não há avaliações
- Results and DiscussionDocumento4 páginasResults and DiscussionLouie Shaolin LungaoAinda não há avaliações
- JHVJHDocumento1 páginaJHVJHLouie Shaolin LungaoAinda não há avaliações
- Coating FormulationDocumento2 páginasCoating FormulationLouie Shaolin LungaoAinda não há avaliações
- NULLDocumento1 páginaNULLLouie Shaolin LungaoAinda não há avaliações
- NULLDocumento1 páginaNULLLouie Shaolin LungaoAinda não há avaliações
- NulllllDocumento1 páginaNulllllLouie Shaolin LungaoAinda não há avaliações
- NulllllaasaDocumento1 páginaNulllllaasaLouie Shaolin LungaoAinda não há avaliações
- NULLDocumento1 páginaNULLLouie Shaolin LungaoAinda não há avaliações
- The Yellow House: A Memoir (2019 National Book Award Winner)No EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Nota: 4 de 5 estrelas4/5 (98)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceNo EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceNota: 4 de 5 estrelas4/5 (895)
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeNo EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeNota: 4 de 5 estrelas4/5 (5794)
- The Little Book of Hygge: Danish Secrets to Happy LivingNo EverandThe Little Book of Hygge: Danish Secrets to Happy LivingNota: 3.5 de 5 estrelas3.5/5 (399)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaNo EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaNota: 4.5 de 5 estrelas4.5/5 (266)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureNo EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureNota: 4.5 de 5 estrelas4.5/5 (474)
- Never Split the Difference: Negotiating As If Your Life Depended On ItNo EverandNever Split the Difference: Negotiating As If Your Life Depended On ItNota: 4.5 de 5 estrelas4.5/5 (838)
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryNo EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryNota: 3.5 de 5 estrelas3.5/5 (231)
- The Emperor of All Maladies: A Biography of CancerNo EverandThe Emperor of All Maladies: A Biography of CancerNota: 4.5 de 5 estrelas4.5/5 (271)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyNo EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyNota: 3.5 de 5 estrelas3.5/5 (2259)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersNo EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersNota: 4.5 de 5 estrelas4.5/5 (344)
- Team of Rivals: The Political Genius of Abraham LincolnNo EverandTeam of Rivals: The Political Genius of Abraham LincolnNota: 4.5 de 5 estrelas4.5/5 (234)
- The Unwinding: An Inner History of the New AmericaNo EverandThe Unwinding: An Inner History of the New AmericaNota: 4 de 5 estrelas4/5 (45)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreNo EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreNota: 4 de 5 estrelas4/5 (1090)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)No EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Nota: 4.5 de 5 estrelas4.5/5 (121)