Escolar Documentos
Profissional Documentos
Cultura Documentos
Heat Energy and Thermodynamics
Enviado por
nkurujoeDireitos autorais
Formatos disponíveis
Compartilhar este documento
Compartilhar ou incorporar documento
Você considera este documento útil?
Este conteúdo é inapropriado?
Denunciar este documentoDireitos autorais:
Formatos disponíveis
Heat Energy and Thermodynamics
Enviado por
nkurujoeDireitos autorais:
Formatos disponíveis
HEAT AND THERMODYNAMICS
HEAT ENERGY
SPECIFIC HEAT CAPACITY AND HEAT CAPACITY ( S.H.C and H.C)
The amount of heat (H) needed to change the temperature of the body depends on;
The material of the body
The mass of the body (m)
The temperature change,
= ,where c is the specific heat capacity
Terms used
1. Heat capacity (C) : this is the amount of heat required to change the temperature of a body by
1K
2. Specific heat capacity (c) is the amount of heat required to change the temperature of 1kg mass
of a substance by 1K
Heat capacity = mass x specific heat capacity
C = mc
1. 100g of water at 1000C is mixed with 100g of water at room temperature (250C). what is the
final temperature of the mixture
2. A copper cooking vessel contains 50g of water. how much energy is required to boil water in the
vessel if the mass of the vessel is 0.5kg [assume initial temperature of water 00C , s.h.c of
water=4200Jkg-1K-1 and that if copper is 400Jkg-1K-1]
Specific Heat Capacity Table
Specific Heat Capacity
Substance
at 25oC in J/goC
H2 gas 14.267
He gas 5.300
H2O(l) 4.184
lithium 3.56
ethyl alcohol 2.460
ethylene glycol 2.200
o
ice @ 0 C 2.010
o
steam @ 100 C 2.010
vegetable oil 2.000
copy right reserved by Bukenya U (+250-785783786) Page 1
sodium 1.23
air 1.020
magnesium 1.020
aluminum 0.900
Concrete 0.880
glass 0.840
potassium 0.75
sulphur 0.73
calcium 0.650
iron 0.444
nickel 0.440
zinc 0.39
copper 0.385
brass 0.380
sand 0.290
silver 0.240
tin 0.21
lead 0.160
mercury 0.14
gold 0.129
Measurement of specific heat capacity
Method 1: method of mixture
This method can be used to determine specific heat capacity of either solid or liquid
A: solid
In this method, the specific heat capacity of a solid will be determined using the following steps;
Weigh the calorimeter (mc)
Pour water in the calorimeter and re-weigh (m)
Mass of water mw = m-mc
The calorimeter is insulated to prevent heat losses
The temperature of water and calorimeter is noted 1
The metal solid of known mass is heated using a steam jacket to ensure that all the parts are at
the same temperature
copy right reserved by Bukenya U (+250-785783786) Page 2
The temperature of the solid is raised up to 2
The solid is transferred fast into the calorimeter
The mixture is stirred until a uniform steady temperature is
attained 3
Assumptions: There is no heat lost to the surrounding
Heat lost by solid = heat gained by water + heat gained by
calorimeter
(2 1 ) = (3 1 ) + (3 1 )
( + )(3 1 )
Thus; =
(2 3 )
B: liquids
For liquids, a solid of known specific heat capacity is dropped in a liquid whose specific heat capacity is
not known. Using the method above, the specific heat capacity of the liquid will be given by;
(2 3 ) (3 1 )
=
(3 1 )
Example
A piece of copper of mass 100g is heated to 1000C and then transferred to a well lagged copper can of
mass 50g containing 200g of water at 100C .Neglecting heat loss, calculate the final steady temperature
of water after it has been well stirred.
[Take the specific heat capacity of copper and water to be 400Jkg-1K-1 and 4200Jkg-1K-1 respectively]
Solution
NB. Heat losses to the surrounding must be minimized
copy right reserved by Bukenya U (+250-785783786) Page 3
1. Conduction losses are minimized by lagging the calorimeter with any material that can trap air
e.g cotton wool and the calorimeter is placed on an insulating wood
2. Convection losses are reduced by covering the calorimeter with card board. it should have two
holes , one for thermometer and the other for stirrer
3. Radiation losses are not minimized in the experiment
Cooling correction ()
Because of heat losses to the surrounding, the observed final temperature 3 .of the mixture is less
than what it should have been if there were no heat losses. The small value of temperature(), to be
added to the experimentally observed maximum temperature of the mixture to compensate for the
heat loss to the surrounding is called a cooling correction
Consider the curve below
3 is experimental maximum temperature
is cooling correction
3 is the corrected maximum temperature
of the mixture
1 is the initial temperature
Corrected maximum temperature 3 = 3 +
The accurate value of specific heat capacity is got by;
(2 1 ) = (3 1 ) + (3 1 )
Obtaining a cooling correction for a poor conductor of heat and a good conductor
If the hot solid is a poor conductor of heat, the heat transfer to the calorimeter and liquid (water) will be
very slow and some time will elapse before the mixture reaches its maximum steady temperature
During this period, a given amount of heat is lost to the surrounding even if the calorimeter is lagged. A
cooling correction would be very necessary
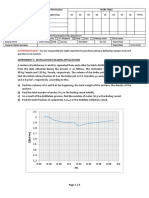
To obtain the cooling correction, the temperature of the mixture is recorded at regular intervals of time
i.e every 30seconds or 1min. starting just before the hot solid is dropped into the calorimeter 1 .and
ending when the temperature has fallen by about 10C below the observed maximum temperature 2
A graph of temperature of the mixture against time is plotted starting the temperature axis at 1 and the
time axis at 0. As shown below
copy right reserved by Bukenya U (+250-785783786) Page 4
AB is drawn through the top of the curve parallel
to the temperature axis and CD further along the
curve. The areas A1 and A2 are estimated by
counting the squares on the graph paper
1
The cooling correction is given by; =
2
Example;
In the experiment to determine s.h.c of rubber by the method of mixture, the following data were
obtained
Mass of rubber = 27.05g
Mass of calorimeter = 44.70g
Mass of water in calorimeter = 70.00g
Initial temperature of rubber = 96.50C
The temperature of the mixture was read and recorded every minute and the following results were
obtained
Time (min) 0.0 1.0 2.0 3.0 4.0 5.0 6.0 7.0 8.0 9.0 10.0
Temp. of mixture (0C) 30.5 34.5 35.5 36.0 36.5 36.3 36.2 36.0 35.9 35.6 35.4
i. Plot a graph of temperature against time
ii. Determine the cooling correction
iii. Calculate the s.h.c. of rubber (sh.c of water and calorimeter are 4200Jkg-1K-1 respectively )
Examples
1. A heating coil is placed in a thermos flask containing 0.06kg of water for 600s. The temperature
of water rose by 250C during this time. The water is replaced by 0.04kg of another liquid and the
same temperature rise occurs in 180s. calculate the s.h.c of the liquid given the s.h.c of water to
be 4200Jkg-1K-1 and state any assumption made
2. When a block of a metal of mass 0.11kg and s.h.c 400Jkg-1K-1 is heated to 1000C and quickly
transferred to a calorimeter containing 0.2kg of a liquid at 100C, the resulting temperature is
180C. On repeating the experiment with 0.4kg of the liquid in the same container and at the
same initial temperature of 100C. The resulting temperature is 14.50C. calculate ;
a. The specific heat capacity of the liquid
b. The thermal capacity of the container
Method 2: Electrical method
copy right reserved by Bukenya U (+250-785783786) Page 5
A: solid
Two holes of reasonable size are drilled into the metal solid (one for heater and the other for
thermometer)
The metal is weighed to give a mass (m)
The solid is lagged to avoid heat losses to the surrounding and the setup of the apparatus is as
shown bellow
The initial temperature of the metal is noted 1
Switch k is closed and a stop clock is started
When the temperature rises to a reasonable
value 2 after time t, the switch is switched off. assume
no heat is lost to the surrounding
Work done by the heater = heat gained by the block
= (2 1 )
The specific heat capacity of the solid metal is given by; =
(2 1 )
NB. Some oil is poured in the holes of the thermometer and coil to ensure thermo conductivity. The
current (I) is read from the ammeter (A) and the p.d from thee voltmeter (V)
B: For a liquid
Calorimeter is weighed (mc)
Its filled with liquid and re-weighed (m) . mass of the liquid mL = m mc
The coil connected to the current system is connected as shown below;
The initial temperature 1 .is noted
A stop clock is started and switch k closed
When the temperature rises to 2 .it is stirred to
ensure uniform temperature and switch (k) is opened
Assume no heat lost to the surrounding,
Heat absorbed by liquid + calorimeter = work done by heater
(2 1 ) + (2 1 ) =
(2 1 )
The specific heat capacity of the liquid is given by; =
(2 1 )
Example
copy right reserved by Bukenya U (+250-785783786) Page 6
A current of 2.5A passing through a heating coil immersed in 180g of paraffin (s.h.c 2000Jkg-1K-1)
contained in 100g calorimeter (s.h.c 400Jkg-1K-1) rises the temperature from 50C below room
temperature to 50C above room temperature in 100seconds . What is the reading of the voltmeter
connected across the heating coil
B) Continuous flow method for determining s.h.c of a liquid
The apparatus below is used in the method
Where;
k is a constant head liquid tank
u is a tube and R is a spiral resistance
heating coil
G is a glass jacket evacuated to reduce loss
of heat by convection
T is thermometer
If the apparatus has been running for some time, it settles eventually to a steady state in which the heat
supplied is carried away by the liquid and is used to upset the heat losses. The inlet and outlet
temperatures 1 and 2 respectively are noted. The mass rate of flow, m is measured
NB. m is measured by collecting and measuring liquid out per second ( = )
Power supplied = rate of absorption by the liquid
= (2 1 ) + (i)
C is the s.h c of the liquid, h is the rate of heat loss
The rate of flow, is changed to m1 . The current and voltage are adjusted to V1 and I1 to bring 2 back to
original value
= (2 1 ) + .(ii)
Using equestions (i) and (ii) ,The specific heat capacity can be determined from;
= (
)(2 1 )
NB. The rate of heat loss (h) of the liquid can be calculated from either equation (i) or (ii)
Advantages of continuous flow method
1. The s.h.c of the apparatus is not required since at steady state the apparatus absorbs no mere
heat
copy right reserved by Bukenya U (+250-785783786) Page 7
2. No cooling correction is required. the heat lost to the surrounding is taken care of by repeating
the experiment
3. The temperature to be measured 1 and 2 , are static at steady state. They can therefore be
measured at leisure and accurately using a platinum resistance thermometer
Disadvantages of continuous flow method
1. Requires a liquid which is available in large quantities
2. Used only for liquids and cannot be used for solids
Example
In experiment using continuous flow, the results were as follows:
Expt 1 Expt.2
p.d (V) across the heating coil 10V 15V
current I (A) through the coil 0.60A 0.80A
0
temperature ( C ) of the incoming liquid 25.5 25.5
0
temperature ( C ) of out flowing liquid 30.5 30.5
mass (kg) of liquid collected 0.86 0.69
time (s) taken to collect the liquid 300 120
i. Calculate the s.h.c of the liquid
ii. Calculate the rate at which heat is lost to the surrounding
Exercise
A student using continuous flow calorimeter obtains the following results;
Using water which enters at 180C and leaves at 220C, the rate of flow is 20gmin-1, the current in the
heating element is 2.3A and the p.d across it is 3.3V, using oil which flows in and out at the same
temperature as water, the rate of flow is 70gmin-1, the current is 2.7A and the p.d is 3.9V
a. Taking s.h.c of water to be 4200Jkg-1K-1. Calculate clearly ;
i. The rate of heat loss from the apparatus
ii. The s.h.c of oil
b. Explain carefully how using this method , the s.h.c of oil could be measured without the
knowledge of s.h.c of water
c. Explain why readings should only be taken when steady state exists. how do you ensure that the
condition is attained
LATENT HEAT
Latent heat is that hidden heat which when supplied to the body does not change its temperature
but changes its state instead
copy right reserved by Bukenya U (+250-785783786) Page 8
Terms used
1. Specific latent heat is the amount of heat required to change the state of 1kg mass of a
substance at constant temperature
The units are Jkg-1 [heat = mass x specific latent heat]
2. Specific latent heat of vaporization (Lv) is the amount of heat required to change to change 1kg
mass of a substance from liquid to vapour at constant temperature
3. Specific latent heat of fusion (L) : this is the amount of heat required to change 1kg mass of a
substance from solid to liquid at constant temperature
Examples: [where necessary use specific latent heat of vaporization of water =2.26x106 Jkg-1 , s.h.c of
ice =2100 Jkg-1K-1 , s.h.c of water =4200Jkg-1K-1 , s.h.c of copper =400Jkg-1K-1 ]
1. Calculate the amount of heat required to change 50kg of ice water to liquid water at 00C
2. A well lagged copper calorimeter of mass 0.1kg contains 0.2kg of water and 0.05kg of ice at 00C,
steam at 1000C containing condensed water at the same temperature is passed into the mixture
until the temperature of the calorimeter and its contents is 300C. If the increase in mass of the
calorimeter and content is 0.025kg. calculate the percentage of condensed water in wet steam
3. A copper solid of mass 0.5kg and temperature 900C is placed into a mixture of ice and 0.1kg of
water contained in a vacuum flask. the final temperature of the mixture was found to be
100C.calculate the mass of ice initially in the mixture
Determination of specific latent heat of vaporization Lv of a liquid by the electrical method (Dewar
flask method)
This can be done using the setup of the apparatus below;
After the liquid has been boiling for some time it becomes
surrounded by a jacket of vapour at its boiling point and a steady
state is attained
At steady state;
i. The rate of vaporization is equal to the rate of
condensation
ii. The temperature of the apparatus remains steady and it
absorbs no more heat
copy right reserved by Bukenya U (+250-785783786) Page 9
iii. The heat supplied is used in evaporating the liquid and offsetting heat losses (mainly by radiation )
The liquid emerging from the condenser is collected for a measured time (t) and its mass (M) is
obtained. The mass rate, ( = ).
Power supplied = rate of absorption + rate of heat loss
= + ..(i)
The experiment is repeated for different I and V to have different rate of evaporation
1 1 = 1 + (ii)
Using equations (i) and (ii) , the specific latent heat of vaporization can be got from ;
1 1
=
1
NB. Rate of heat loss, h can also be obtained by back substitution in any of the equations
Advantages of this method
i. The heat capacity of the apparatus is not required
ii. Heat losses to the surrounding is taken care of by repeating the experiment
Example
In an experiment of continuous flow, a heating coil is immersed in water contained in a Dewars flask.
The water is kept boiling and the steam kept condensed by a cooling device. The p.d across the heating
coil, the heater and mass (M) of water condensed in 600seconds for various values of V and I are given
below;
V (volts) 30 40 50 60 70
I (A) 1.25 1.50 1.75 2.00 2.50
M x10-3 (kg) 8.50 14.75 22.25 31.00 46.00
Draw an appropriate graph and from it determine
i. The specific latent heat of vaporization of water
ii. The rate of heat loss to the surrounding
Determination of the specific latent heat of fusion of ice (L)
Lumps of dry ice are placed in a filter funnel and a heating system connected as shown below;
copy right reserved by Bukenya U (+250-785783786) Page 10
Power supplied by the heater + rate of absorption from surrounding =
rate of absorption by ice
+ =
Or = .(i)
The values of I and V can be adjusted to get different values for the rate
at which ice melts (m)
A graph of IV is plotted against m
m0 is the mass of ice that would melt if the heater was off. h is
the rate at which heat is absorbed from the surrounding
The specific latent heat of fusion is got by finding the slope of the
graph
HEAT TRANSFER
HEAT CONDUCTION
This is the transfer of heat energy from one place to another through a substance without the movement
of the substance as a whole
Mechanism of heat conduction
If as specimen is heated at one end, the atoms gain energy and their vibrations increase, this energy can
be passed on to another atom by inter-atomic vibration
Heat conduction in gases
Fast moving molecules pass on kinetic energy to slower-moving ones when they collide with them. In
this way, heat is slowly conducted through the gas
Heat conduction in metals
Metals contain free electrons which are in thermal equilibrium with the surrounding atoms. These
electrons travel at high speed and transfer energy quickly from one part of the metal to another by
collisions with other electrons and other atoms
copy right reserved by Bukenya U (+250-785783786) Page 11
NB. In metals heat is carried mainly by motion of free. Although it is also carried by inter-atomic
vibrations
Heat conduction in non-metals, solids and liquids
The molecules are coupled to each other by forces. There are virtually no free electrons. Heat is
transferred from one atom to another by inter-atomic vibrations. This process of heat conduction is slow
as compared to that in metals
Thermal conductivity / coefficient of thermal conductivity (k)
Consider a slab of material of thickness L and cross section area A, having temperature 1 and 2 at its
ends
Where 1 > 2
Rate of heat flow ,through the specimen depends on ;
i. The cross section area A
ii. The temperature difference between the faces 1 2
iii. The inverse of the distance L ,between the faces
iv. The nature of the material
(1 2 )
Thus; = , where k is a constant which depends on the nature of the material. Called thermal
conductivity 9coefficient of thermal conductivity
So, = . The units are Wm-1K-1
NB. Thermal conductivity (K) is the rate of heat flow through a material per unit area in a region o unit
temperature gradient
Example
1. A slab of cross section area 3600cm2 and thickness 10cm is exposed on the lower surface of
steam 1000C. a block of ice 00C rests on upper surface of the slab in 1hr, 4800g of ice is melted.
Calculate the thermal conductivity of the slab [specific latent heat of fission of ice is
3333x103Jkg-1
2. A room is maintained at 200C by a heater of resistance 20connected to 200V mains. The
temperature is uniform throughout the room and the heat is transmitted through a glass
window of area 1m2 and thickness 0.2cm. Calculate the temperature outside the room.
[Thermal conductivity of glass is 0.84Wm-1K-1]
copy right reserved by Bukenya U (+250-785783786) Page 12
3. A flat bottomed metal tank of water is dragged along a horizontal floor at the rate of 20ms-1. The
tank is of mass 20kg and contains 1000kg of water and all the heat produced in the dragging is
conducted to the water through the bottom plate of the tank. If the bottom plate has an
effective area of conductivity 1m2 and thickness 5cm and temperature of water in the tank
remains constant at 500C. calculate the temperature of the bottom surface of the tank , given
the coefficient of friction between the tank and floor is 0.343 and thermal conductivity if the
tank material is 105Wm-1K-1 and acceleration due to gravity g=9.81ms-2
Conductivity in series
Example
1. A composite metal bar of uniform section is made of length 25cm of copper, 10cm of nickel and
15cm of aluminium , each part being in perfect thermal contact with the adjoining part . The
copper end of the composite rod is maintained at 1000C and the aluminium end at 00C. The
whole rod is lagged so that is assumed no heat loss occur at the side .calculate the temperature
of copper-nickel and nickel- aluminium junctions in the steady state [given thermal conductivity
of copper, nickel and aluminium is 380Wm-1K-1, 59Wm-1K-1, 210Wm-1K-1 respectively ]
2. The rods of copper, brass and steel are welded together to form a Y-shape as shown below.
The cross section area of each rod is 4cm2. The ends of copper rod are
maintained at 1000C and the other ends of brass and steel rods at 00C.
Assume that there is no loss of heat from the surface of the rods. The
lengths of the rods are; 46cm, 13cm and 12cm for copper, brass and steel
respectively. Given that the thermal conductivity of copper, brass and
steel are 380Wm K , 107Wm-1K-1, 40Wm-1K-1 respectively]
-1 -1
i. What is the temperature of the junction points
ii. What is the heat current in copper rod?
Experiment to determine the coefficient of thermal conductivity (k)
For good conductor of heat
Thermal conductivity of a good conductor of heat, e.g copper can be determined using Searles
apparatus below. The specimen bar is heated from one end using a steam jacket and cooled at the other
end by circulating water
copy right reserved by Bukenya U (+250-785783786) Page 13
In order not to upset the temperature gradient, the cold water is allowed in through A and not through
B. When the apparatus has been running for some time, a steady state condition is attained when;
i. 1 , 2 , 3 , 4 remain constant
ii. The rate of heat flow and temperature gradient are the same for any section of the bar since
it is lagged
The steady temperatures 1 , 2 , 3 , 4 . Are recoded and the mass rate of flow, m of water obtained by
collecting the warm water flowing out of B and measuring the mass obtained in a given time.
(1 2 )
Rate of heat flow ; = (4 3 ) =
(1 2 )
This means, (4 3 ) = . Thus k can be calculated
NB. The bar should be lagged to prevent heat loss in the sides so that to have a uniform temperature
gradient
A graph of temp. Against length (L) for lagged bar
A graph of temp. Against length (L) for a non-lagged bar
copy right reserved by Bukenya U (+250-785783786) Page 14
For a poor conductor of heat e.g glass
The specimen is made in form of a disc D. the disc is placed on a thick brass slab B containing a
thermometer and heated from above by a steam chest C whose thick base also carries a thermometer
The experiment has got two parts ;
Part 1
Steam is passed until the temperature 1 and 2 are steady. Heat flows from C to B through D and then
lost to the surrounding. At steady state, the rate of heat flow equals the rate of heat loss by B at
temperature 1
(1 2 )
= .(1)
Where; , 1 , 2 , are all measured
Part 2.
The disc is removed and B is heated directly from C until the temperature is about 50C above what it was
in part 1. i.e = (1 + 50 )C is then removed and a thick felt pad is placed on top of B
Temperature time readings of B as it cools are recorded and plotted on a cooling curve
NB. The main aim of part 2, is to find the rate at which heat is lost from B at temperature 1
The temperature time graph
copy right reserved by Bukenya U (+250-785783786) Page 15
The rate of temperature fall of B at 1 is equal to the slope of the tangent at 1
(2 1 )
Where m is mass of slab B , c is s.h.c of brass . therefore ; =
Thus, k can be calculated
Thermal radiation
Thermal radiation is the electromagnetic radiation emitted by a body solely on account of its
temperature
At low temperature, the body emits mainly infrared and at high temperature the body emits visible and
ultraviolet radiations in addition to infrared radiation. Infrared is that part of the electromagnetic
spectrum extending from 0.7 to 1mm wave length
Properties of infrared
i. Moves at the same speed as speed of light in a vacuum (3.0x108ms-1)
ii. Is reflected and refracted like light
iii. Increases the temperature of a body when absorbed by matter
iv. Absorbed by glass but transmitted by rock salt and quartz
v. Causes photo-electric emission from surfaces like ceasium
vi. Affects special type of photographic plates. this enables pictures to be taken in the dark
Infrared detectors
a) The thermopile
A thermopile consists of many thermocouples connected in series
copy right reserved by Bukenya U (+250-785783786) Page 16
A- Hot junctions of the thermopile
B- Cold junctions of the thermopile
C- Metal cone with highly polished surface
D- Galvanometer
The hot junctions A, are blackened to make them good absorbers of the incident radiation. The cold
junctions B are shielded from the radiation so that an emf is set up between the hot and cold junctions
which is measured by deflection of the galvanometer. The cone C concentrates the radiation on to the
hot junction A. the magnitude of emf induced depends on the intensity of the radiation
b) Detection of infra-red in a beam of sum light
The set-up of the materials is as shown below
Light from the sun is controlled by the slit and fall on a lens made of quartz material which concentrates
light onto the prism
The prism is made of material which is opaque to visible spectrum. The infrared proportion is
transmitted and refracted. This is detected by the thermopile on the opposite side
Prevosts theory of heat exchange
A body emits radiation at a rate which is determined only by the nature of its surface and absorbs
radiation at a rate which is determined by the nature of its surface and the temperature of the
surrounding
A body at constant temperature is in a state of thermal dynamic equilibrium with its surroundings. Its
rate of absorption from the surrounding is equal to the rate of emission of radiation to the surrounding
NB. From Prevosts theory, it can be deduced that if a body is a good absorber it must be a good emitter
otherwise its temperature would rise above the surrounding
Experiment to determine which surfaces are good emitters and which are poor emitters
A cubical metal tank (Leslies cube) whose sides have a variety of polish i.e matt black, highly polished
silver e.t.c is used. The tank contains boiling water and therefore at a constant temperature
copy right reserved by Bukenya U (+250-785783786) Page 17
The thermopile is made to face the different faces of
the cube in turn and the deflection noted on the
galvanometer
NB. It is noted that a greater deflection is obtained
when the thermopile faces the black face than a silver
polished face. Therefore black bodies are good emitters (radiators) .For example car radiators are
painted black to radiate heat from the engine
Blackbody radiation
When radiation falls on a surface, three things may happen to it;
Part will be reflected (R)
Part will be absorbed (A)
Part will be transmitted (T)
Incident energy , I=R+A+T
A blackbody is a body which absorbs all radiations falling on it and reflects and transmits none. An
approximate black body absorber can be made by punching a very small hole in an enclosure (cavity)
whose inside walls are rough and black
At each reflection of incident radiation inside cavity, a high percentage of the radiation is absorbed. All
the radiation is finally absorbed after multiple reflections
NB. The cone prevents direct reflection normal to the whole
A black body radiator (cavity radiator)
The amount of radiation emitted by a body depends on three things;
The surface area of the body
The type of the surface
The temperature of the body
A black body radiator is one which emits radiation which is a characteristic of its temperature and does
not depend on the nature of its surface. It is made by surrounding the enclosure with a heating coil
copy right reserved by Bukenya U (+250-785783786) Page 18
Black body radiation is the one whose quality (wavelength) depends on the temperature of the body
Spectral curves for blackbody radiation
Spectral curves show the variation of the relative intensity emitted by a black body with wavelength
3 > 2 > 1
NB.
Relative intensity .is the energy radiated per
unit wavelength interval
Intensity E is the energy per second per square
meter
Or intensity E, is power emitted per square
meter
Laws of black body radiation
1. Weins displacement law
For each temperature T , there is a wavelength which is emitted with maximum intensity
The constant is called Weins displacement constant =2.9x10-3mK
As the temperature increases , the relative intensity at each wavelength increases ( the body becomes
brighter but the increase is more rapid for short wavelength (the color of the body changes )
Appearance of a body depends on the position of .
i.e the body changes from its color when cold ( in the infrared region ) to red-hot ( in the red
region of the visible spectrum) to yellow-hot to white-hot ( .in the middle of the visible spectrum)
and eventually to blue-hot ( .in the blue region)
2. Stefans law (Stefan-Boltzmann)
= 0 = 4
copy right reserved by Bukenya U (+250-785783786) Page 19
Thus = 4 , where, is Stefans constant , i.e = 5.7108 2 4 and E is energy
emitted per second per square meter area
The total power radiated by an area S is given by ; = = 4
= , where S is effective radiating surface area
Example
1. A tungsten filament of an electric lamp has a length of 0.5m and a diameter of 6x10-5. The power
rating of the lamp is 60W. Assuming the radiation from the filament is equivalent to 80% that of
a perfect black body radiator at the same temperature, estimate the steady temperature of the
filament (Stefans constant , = 5.7108 2 4 .)
2. Estimate the temperature Te of the Earth. assuming it is in radiative equilibrium with the sun
(radius of sun =7.0x108m, surface temperature of sun is 600K, distance of Earth from sun is
R=1.5x1011m)
Convection
This is the process whereby heat flows by the mass movement of the molecules from one place to
another
When a pot of water is heated, convection currents are set up as the heated water at the bottom of the
pot rises because of its reduced density and then replaced by cool water from above .This principle is
used in many heating systems, such as the furnace
Convection currents in hot water
Natural occurrence of convection currents are land and sea breeze
Sea breeze
During the day, the temperature of the land increases more
quickly than that of the sea because land heats up faster that
copy right reserved by Bukenya U (+250-785783786) Page 20
water. The hot air above the land reses and is replaced by colder air from the sea
Land breeze
At night the temperature of the sea is relatively higher
than that of the land because the rate at which land
loses heat is higher than that of the sea. The air above
the sea is warmer than that over the land and a
breeze blows from the land to the sea
Applications of convection currents
Ventilation
Convection is the main factor in ventilation. Air, which has been warmed by breathing tends to rise and
should be given exit near the top of the room and an inlet for cool air near the bottom. A convection
circulation will ensure that fresh air replaces that which has been used
Domestic heat supply (house heating and water supply)
A convection current of hot water from the bottom of the boiler (furnace) rises as shown
The hot water rises. This causes water to circulate in the
heating system. Hot water enters the radiator, heat is
transferred by conduction to the air and the cooled water
returns to the furnace. Pumps some times are used to
improve air circulation. The air heated by the radiator also
rises and replaced by fresh cool air
KINETIC THEORY OF GASSES
This is a simple theory which attempts to relate the macroscopic behavior of an ideal gas with the
microscopic properties of its molecules
It can be used to explain the following phenomenon;
copy right reserved by Bukenya U (+250-785783786) Page 21
A gas occupies all the volume provided to it
The temperature of a gas decreases in an adiabatic expansion
The volume of a fixed mass of a gas kept at constant temperature decreases with increase in
pressure
A gas exerts a pressure on the wall of its container
Pressure of a gas =
Qualitative explanation
Pressure of a gas is due to the bombardment of the gas molecules with the walls of the
container
When molecules bombard with the walls of the container its momentum is reversed at right
angle to the wall
Force exerted by the wall on the molecule is equal to the average rate of change of momentum
(Newtons law )
Pressure is equal to the force per unit area
Quantitative treatment
In order to allow mathematical treatment, the following simplifying assumptions are made in the
theory;
i. The intermolecular forces are negligible
ii. The volume of the molecule is negligible compared to the volume of the container
iii. The molecules are like elastic spheres and they collide perfectly elastic with themselves and with
the walls of the container and have random motion
iv. The time during collision with the wall is negligible compared to the time between collisions
Deriving Pressure of the gas [ = ]
Where p is pressure, is density of the gas, 2 is mean of the square velocities in any direction
Consider a cube of length L containing N molecules moving randomly with in the cube. The cube
contains one type of a gas so that the mass of each molecule is the same
Let be the velocity vector of a molecule at some instant
Cx , Cy and Cz are the velocities with reference to
the x, y and z axes respectively
Thus = +
+
If u, v and w, are actual velocities of the gas
Cx = u , Cy=v and Cz= w
Then 2 = 2 + 2 + 2 .(1)
copy right reserved by Bukenya U (+250-785783786) Page 22
A molecule colliding with wall x- its momentum is reversed
Change of momentum after collision =+mu-(-mu)=2mu
Time taken for a molecule to make successive collisions between opposite faces is t;
2
= or =
The force exerted by a molecule on the wall =
2 2
Or = =
2
2
The pressure exerted by a molecule on wall x ; = = .But L3=V, (volume of the cube)
3
PV = mu2
For N molecules, the total pressure is given by; = (12 + 22 + 32 + +
2
)
2
If is the mean of the square velocities of the x-component velocity
2
12 +22 ++
2 = , Thus = 2 or = 2 = 2 .(2)
(Since mN is the total mass of the gas)
With a large number of molecules of varying speeds, in random motion, the molecules show no
preference for moving in any direction parallel to any axis, so 2 = 2 = 2 thus equation
1
(1) becomes 2 = 32 then 2 = 2
3
Thus the kinetic theory expression for pressure is given by =
Interpretation of absolute temperature (T)
1
From kinetic theory ; = 2 ..(i)
3
For an ideal gas, PV = nRT ..(ii)
Combining (i) and (ii)
1
2 =
3
NB
i) For 1mole of a gas , n=1 , N=NA (NA is the Avogadros number)
3 1 3
2 = , but N=nNA . Thus; 2 =
2 2
1 3
But 2 , is average or mean kinetic energy. thus . , or . =
2 2
-23 -1
= . is called the Boltzmanns constant i.e k = 1.38x10 JK
Root mean square velocity; = 2
copy right reserved by Bukenya U (+250-785783786) Page 23
3
i.e = , for n=1 . thus
ii) R is universal gas constant i.e R = 8.314J/mole.K
iii) Avogadros number is the number of molecules in one mole of a pure substance. [i.e NA =
6.022x1023/mole
Examples
1. A vessel of volume 50cm3 contains hydrogen at a pressure 0f 1.0Pa and temperature of 270C.
Estimate
i) The number of molecules in the vessel
ii) Their root-mean square speed
[Mass of 1mole of hydrogen molecules =2.0x10-23kg/mole]
2. Calculate the root-mean square speed of the molecules of hydrogen at ;
i) 273K
ii) 373K
[Density of hydrogen at s.t.p =9x10-2kgm-3 and 1 standard atmosphere = 1.01x105Pa]
3. At what temperature, pressure remaining constant will the root mean square velocity of
hydrogen be double of its value at s.t.p?
Gas laws deduced from kinetic theory
Using kinetic theory expression = and the interpretation of temperature we can
deduce the following gas laws;
1. Boyles law
1
From = 2
3
2 1 1
= 2 , but 2
3 2 2
For a fixed mass of a gas, N is a constant. If T is constant; PV= constant, i.e P1V1 = P2V2
[Thus Boyles law states that for a fixed mass of a gas kept at a constant temperature, the
volume is inversely proportional to the pressure]
P V characteristic graph
Example
copy right reserved by Bukenya U (+250-785783786) Page 24
A container contains 10cm3of air at a pressure of 20Nm-2 if the volume of the container is
increased to 15cm3. What will be the pressure of the gas?
2. Charles law
1
From = 2
3
2 1 1
= 2 , but 2
3 2 2
1 2
For constant N and P is constant; then, , thus = ;or =
1 2
[Thus Charless law states that the volume of a fixed mass of a gas kept at constant pressure is
directly proportional to its absolute temperature]
V T characteristic graph
Example
A fixed amount of a gas occupies a volume of 40cm3 at 270C. What volume will be occupied by
the same amount of a gas if the temperature is increased to 330C, keeping the pressure constant
3. Pressure law
1
From = 2
3
2 1 1
= 2 , but 2
3 2 2
1 2
For constant N and V is constant; the , thus = ;or =
1 2
If the mass of a gas is fixed, volume is constant, then;
[Thus pressure law states that for a fixed mass of a gas, the pressure is directly proportional to
the absolute temperature if the volume is constant]
P-T graph
Example
A gas was sealed in a container at s.t p , what will be the pressure of the gas , when it is heated
to a temperature of 430C , keeping the volume of the container constant ?
NB.
i) If all the three gas laws are combined , the form the gas equation of state
=
copy right reserved by Bukenya U (+250-785783786) Page 25
ii) An ideal gas is the one which obeys the three gas laws, where the intermolecular forces is
negligible
iii) For all ideal gases , the equation of state can be written as ; PV = nRT
Example
1. A fixed mass of a perfect gas is held at 270C. It is heated so that its volume is doubled, but
the pressure constant. find the new temperature
2. 125cm3 of a gas is collected at 150C and 755mm of mercury pressure. calculate the volume
of the gas at s.t.p
4. Avogadros hypothesis: this states that equal volumes of all gases under the same conditions of
temperature and pressure , have equal number of molecules
Consider two ideal gases (1) and (2). We can write the equations
1
1 1 = 1 1 12 for gas (1)
3
1
2 2 = 2 2 22 for gas (2)
3
If their pressures, volumes and temperature are the same, then; P1 = P2 , V1 = V2
1 1
But 1 12 = 2 22 if T1 = T2, then N1 = N2
2 2
5. Daltons law of partial pressures
This states that, the pressure of a mixture of gases is the sum of the partial pressures of the
component gases
NB. Partial pressure of a gas, is the pressure a gas would have if it occupied the whole container
alone
Consider a cylinder of volume V containing a gas (1) alone
1 2 1
For gas (1): 1 = 1 1 12 = 1 1 12
3 3 2
If the cylinder is filled with gas (2)
1 2 1
2 = 2 2 22 = 2 2 22
3 3 2
If the two gases are mixed; gas(1) +gas (2)
2
= ( ) , where N = N1 + N2
3
1 1
Since the temperature is kept constant, 1 12 = 2 22 = mean kinetic energy
2 2
2 1 2 1
= 2 + 2
3 1 2 1 1 3 1 2 1 1
= 1 + 2
Thus, P = P1 + P2 . Where P1 and P2 are partial pressures of the gas (1) and gas (2) respectively
Example
Air consists of approximately 20% oxygen and 80% nitrogen. The relative masses of oxygen and
nitrogen are 32 and 28 respectively. Calculate the ratio of;
i. The mean square speed of oxygen to that of nitrogen in air
ii. The partial pressure of oxygen to that of nitrogen in air
6. Grahams law of diffusion
copy right reserved by Bukenya U (+250-785783786) Page 26
It states that the rates if diffusion of two gases are inversely proportional to the square roots of
their densities at a given temperature and pressure
Consider the case of two gases A and B having densities and respectively at a pressure P.
Let the two gases be allowed to diffuse into each other,
According to kinetic theory of gases, the root mean square velocities of the molecules will be
3 3
given by;( ) = and ( ) =
( )
Thus ( ) =
But the rates of diffusion of a gas are proportional to the root-mean square velocity (rms).
( )
Hence, ( ) = , Thus = .This is called Grahams law
Mean free path
Despite the high average speed of molecules, the gases diffuse very slowly in air. A smell even if
it is aided by convection, takes time to travel. This is because molecules suffer frequent
collisions with other molecules. At each collision the direction changes and the path follows zig-
zag steps. The progress of molecules is very low. The average length of a step between collisions
is called the mean free path
Question
Helium gas occupies a volume of 0.04m3 at a pressure of 2x105Pa and temperature of 300K.
Calculate;
i. The mass of helium
ii. The rms speed of its molecules
iii. The rms speed at 432K when the gas is heated at constant pressure to this temperature
[Relative molecular mass of helium and hydrogen is 4g and 2g respectively and molar gas
constant R=8.314Jmol-1K-1]
Real gases
Real gases have both attractive and repulsive forces. These obey the equation of state (PV=nRT) only
when they are at a very low pressure and a temperature above their critical temperature
Critical temperature (Tc) of a gas is the temperature above which a gas cannot be liquefied by mere
compression
copy right reserved by Bukenya U (+250-785783786) Page 27
In order to obtain a better equation for real gases, the two simplifying assumptions made in kinetic
theory have to be modified i.e
i. Intermolecular forces cannot be neglected
ii. The volume of the molecules themselves cannot be ignored
NB.
If the co-volume of the molecule is b, then the free volume movement is (V-b)
Molecules as they approach the wall, they experience an attractive force in the bulk so this reduces
pressure at the walls
Pressure in bulk = pressure at the wall + pressure defect
= P + P1
()2 but =
1 1
For a fixed mass of a gas , Thus 2 , So 1 = . Where, is a constant
2 2
Bulk pressure = +
2
Using the equation of state; (Van der Waals equation), PV= n RT
For real gases, it will be given by; + ( ) =
2
The P-V curves (isothermals) are as shown below
C is point of inflexion which occurs at a critical temperature
Saturated vapour
copy right reserved by Bukenya U (+250-785783786) Page 28
Terms used
Gas: this is the gaseous state of a substance above its critical temperature
Vapour: this is the gaseous state of a substance below its critical temperature
A vapour can be saturated or unsaturated
A saturated vapour is the one which is in contact with its own liquid
Unsaturated vapour is the one which is not in contact with its own liquid
NB. A saturated vapour behaves totally different from unsaturated vapour and gases. However
unsaturated vapour behave almost the same way as gases
Experiment to demonstrate saturated vapour pressure
A small quantity of a volatile liquid (e.g ether) is
introduced at the bottom of a mercury barometer; it
rises to the top of the mercury column and evaporates
into the vacuum. The pressure it exerts causes mercury
to fall
When more ether is injected, the level drops further
but stops as soon as liquid ether appears on top of the
mercury column. The ether vapour is said to be
saturated and exerting saturation vapour pressure at
the temperature of the surrounding.
Before liquid ether gathers above mercury, the ether vapour is unsaturated.
P1 is vapour pressure and P2 is saturated vapour pressure (s.v.p)
NB. The S.V.P of a substance is the pressure exerted by the vapour in equilibrium with the liquid
Vapour and gas laws
Unsaturated vapour obeys Boyles law roughly up to near saturation point (AB). Consider P-V curve
below
At B condensation of the vapour starts, liquid and
saturated vapour co-exists along BC. Since the mass of the
vapour changes, Boyles law cannot work here. So the
pressure is constant as shown in the figure above for
tubes c and d i.e P2 is constant
Unsaturated vapour approximately obey Charles law and
pressure law but saturated vapour does not
copy right reserved by Bukenya U (+250-785783786) Page 29
Consider the P-T characteristic below
A mixture of gas and unsaturated vapour obey the gas laws fairly well each of them exerting the same
pressure as it would exert if it occupied the total volume alone (Daltons law)
Example
1. A horizontal tube of uniform bore, closed at one end, has air trapped by a small quantity of
water. If the length of the closed air column is 20cm at 140, what will it be if the temperature is
raised to 400C and atmospheric pressure remains constant at 760mmHg ?[saturated vapour
pressure of water at 140C and 400C is 10.5mm and 49.5mm of mercury respectively]
2. A closed vessel contains a mixture of air and water vapour at 270C at a total pressure of
107x103Nm-2. The water vapour is just saturated at this temperature , calculate the total
pressure in the vessel if
i. The temperature is raised to 600C
ii. The temperature is lowered to 170C
[S.v.p of water at 170C and 270C is 1.9x103Nm-2 and 3.7x103Nm-2]
3. Two cylinders P and Q each of volume 1.5L are joined in the middle by a tap T and placed in a
constant temperature bath at 600C as shown below
P contains a vacuum while Q contains air and saturated water vapour. The total pressure in Q is
200mmHg, when T is opened; equilibrium is reached with water vapour remaining saturated. If
the final pressure in the cylinder is 150mmHg, calculate the saturation pressure of water at 600C
Kinetic theory explanation for the occurrence of s.v.p
Consider a liquid confined in a container with a fixed piston
copy right reserved by Bukenya U (+250-785783786) Page 30
The liquid molecules are moving randomly with mean kinetic energy determined by the liquid
temperature
The most energetic molecules overcome the attractive force with the surface and escape from
the surface to become vapour the process called evaporation and this is determined by the
liquid temperature
Molecules of vapour move randomly colliding with walls of the surface of the liquid and re-enter
the liquid the process called condensation. The rate of condensation and vapour pressure
depends on the density of the vapour
A state of dynamic equilibrium is soon attained when the rate of condensation is equal to the
rate of evaporation. The density of vapour and hence vapour pressure is then maximum and it is
called the saturated vapour pressure
Behavior of saturated vapour
Effect of volume change on s.v.p keeping temperature constant
Consider a saturated vapour contained in a cylinder with frictionless movable piston
There is dynamic equilibrium. When the volume is decreased at constant
temperature,
The rate of evaporation remains constant
The density of the vapour , the rate of condensation and the s.v.p
increases
But the increase in the rate of condensation without any change in the rate of evaporation causes the
density of the vapour, the rate of condensation and s.v.p to decrease. Hence the dynamic equilibrium is
re-attained at the original value
Boiling (ebullition)
This is a phenomenon which occurs when the s.v.p of a liquid is equal to the external atmospheric
pressure. This occurs at a fixed temperature called boiling point
The boiling point is the fixed constant temperature at which s.v.p becomes equal to the external
atmospheric pressure
Whereas evaporation occurs at the surface of a liquid at all temperatures, boiling takes place at a
temperature determined by the external atmospheric pressure. It consists of formation of bubbles of
vapour throughout the liquid and the pressure inside the bubble is s.v.p at boiling point. The pressure
outside is practically equal to atmospheric pressure in an open vessel
NB. If we neglect the hydrostatic pressure and surface tension, the liquid boils when s.v.p is equal to the
external atmospheric pressure
Factors that affect boiling point of a liquid
copy right reserved by Bukenya U (+250-785783786) Page 31
External pressure
Addition of impurities in the liquid
PROPERTIES OF MATERIALS
Terms used
Elasticity: this is the property which allows a material to regain its shape after being distorted or
deformed. The deformation can be tensile (if a stretching force is used) or compressive (if a compressing
force is used)
Elastic deformation: a material is said to be under elastic deformation if it can regain its shape after the
distorting force is reformed
Elastic limit of a material: is the maximum amount by which it can be stretched and still regaining its
shape after the distorting forces is removed. If a material is stretched beyond its elastic limit, its shape is
permanently changed
Plastic deformation: a material is said to be under plastic deformation if some extension remains when a
stretching force is removed. Plastic deformation occurs when a material is deformed beyond its elastic
limit
Ductility; this is the property of a material to allow to be hammered into other useful shapes. A ductile
material is the one which can be permanently stretched. They can be drawn into wires e.g copper. They
can undergo both elastic and plastic deformation
Brittle materials: these are materials which stretch very little and break without warning e.g glass
Stiffness: this is the resistance which a material offers to have its size / shape changed. This is measured
using Youngs modulus
Breaking stress (tensile strength): this is the maximum stress which can be applied to a material beyond
which a material will break
Strength: this is the ability of a material to withstand a force before it breaks
Toughness: this is the ability of a material to resist crack growth on its surface
Hardness: this is the ability of a material to resist plastic deformation
Tensile stress, strain and Youngs modulus
Consider a stretching force F applied to a material of original length .and cross section area A, let the
force cause an extension, e.
A. =
copy right reserved by Bukenya U (+250-785783786) Page 32
=
It is the ratio of extension to the original length. It has no units
B. =
This is the ratio of force to the cross section area. The units are Nm-2
Example
A gymnast of mass 70kg hangs by one arm from a high bar. If the gymnasts whole weight is assumed to
be taken by the humerus bone. Calculate the stress in the humerus if it has a radius 5cm
The strength of a wire
We can use a wire to investigate the strength of a material when stretched. The extension of a wire
depends on;
Stretching force (F)
Cross section area (A)
The nature of the material
Original length of the material
Youngs modulus (E)
Provided the stress is not so high that the elastic limit of proportionality is not exceeded, the ratio
stress/strain is a constant for a given material and is known as Youngs modulus
= = , giving = ..(1)
0
Thus, this means force is proportional to extension (Hookes law)
The units of Youngs modulus are Nm-2 and its dimensions are ML-1T-2
Hookes law
It states that the extension caused in a material is directly proportional to the force applied if the elastic
limit is not exceeded
Or stress is directly proportional to strain if the elastic limit is not exceeded
Thus , , or = ; where k is called the force constant of the material . from equation (1) it
means that ; = . The units of k are Nm-1
copy right reserved by Bukenya U (+250-785783786) Page 33
NB. The units of Youngs modulus are Nm-2 and the dimensions are MT-1T-2
Examples
1. A rod of original length 1.2m and cross section area 1.5x10-4m2 is extended by 3.0mm when the
stretching tension is 6.0N; calculate the Youngs modulus of the material rod
2. A wire of length 1.2 has a diameter of 10mm. if a force of 20N is applied to it, it expands by
25cm. calculate ;
i. Force constant of the wire
ii. Youngs modulus of the wire
3. A steel wire 30m long has a cross section of 0.5mm2, Youngs modulus for steel is 2x1011Pa,
calculate the force constant of the wire
The stress strain curve for a ductile material
Regions
OA is Hookes law region. Stress is directly
proportional to strain. Youngs modulus can only be
defined in this region
CD is plastic deformation region
Points:
B: Elastic limit. Up to B, the wire returns to its
original length when the stress is reduced to zero
Y: yield point: beyond this point, permanent or
plastic deformation occurs. Any given increase in
stress gives a greater increase in strain than before.
If the is gradually reduced to zero the graph follows the dotted line, leaving permanent strain (s)
E: breaking point, without any further increase in stress, the wire begins to undergo physical changes, its
thins out and finally breaks
Comparison of stress-strain graphs for different materials
Glass being brittle has only a small elastic region and does not
undergo plastic deformation. This is due to existence of cracks
in its surface. The high concentration of stress at the crack
makes the glass break
Rubber has a greater range of elasticity and does not undergo
plastic deformation .unstretched rubber has coiled molecules.
copy right reserved by Bukenya U (+250-785783786) Page 34
When stretched, they unwind and become straight
Energy stored in an elastic material
Work done in stretching an elastic material is stored as elastic potential energy in the material
Consider a material stretched from zero extension to extension, e
Work done =area under the force extension curve
1 1
= () = 2 , but = thus stored energy is given
2 2
1
by; = 2 .
2
This is also called strain energy or elastic potential energy
Example
A uniform wire has unstretched length of 2.49m and uniform cross section area 0.5x10-6m2. If the wire is
loaded, it expands and the length becomes 2.52m. Calculate the energy stored in the wire if Youngs
modulus is 2x1011Nm-2
Energy per unit volume (U)
Strain energy per unit volume of a wire (U) is the work done in stretching an elastic material per unit
volume
Consider the length of wire
Original volume of the wire(v)= 0
Work done to stretch the material by extension e is w
1
= 2 ; then = = 2 = ( )()2
2 202 2
U, is also called elastic potential energy per unit volume
Examples
copy right reserved by Bukenya U (+250-785783786) Page 35
1. A wire has uniform diameter of 2mm and length of 5cm. it supports a load of 500g. if the wire
expands by 2cm. calculate the elastic potential energy per unit volume stored in the wire
2. If the Youngs modulus of steel is 2x1011Nm-2. Calculate the work done in stretching the wire
100cm in length and cross section area 0.03cm2when a load of 100N is slowly applied and the
elastic limit is not exceeded
3. A uniform wire has unstretched length of 2.49m. The wire has a uniform cross section area of
0.5x10-6m2. If the wire is loaded, it expands and the length becomes 2.52m. calculate the energy
stored in the wire [youngs modulus of wire is 2x1011Nm-2]
Force in a bar due to thermal expansion
If a metal bar is heated, it will expand, but if it is prevented from expanding by fixing its ends, a large
force will be set up with in the bar
The force , = ; is the expansion if the bar was free to expand . when a material expands due to
heat , the coefficient of linear expansivity is given by ()
i.e = = ; The units of () are K-1 or /0
thus =
from = = . thus =
Examples
1. A steel bar with cross section area of 2cm2 is heated raising its temperature by 1200C and
prevented from expanding. Calculate the resulting force in the bar. Youngs modulus of steel is
2x1011Nm-2
2. A cylindrical copper rod of length 0.5m and diameter 4x102m is fixed between two rigid supports
at a temperature of 200C . the temperature of the rod is increased to 700C
i. Calculate the force exerted on the rigid support at 700C
ii. What is the energy stored in the rod at 700C
[Youngs modulus for copper =1.2x1011Nm-2 and linear expansivity of copper =1.7x10-5K-1]
THERMODYNAMICS
copy right reserved by Bukenya U (+250-785783786) Page 36
Thermodynamics deals with processes that cause energy changes as a result of heat flow to or from a
system and work is done on or by the system
Heat engines such as petrol systems (engines), steam turbines and jet engines all contain
thermodynamic systems designed to transform heat into mechanical work
Heat pumps and refrigerators are thermodynamic devices for transforming energy from cold body to a
hotter one
First law of thermodynamics
It states that, the total energy of a closed system is a constant
Or it states that, the heat supplied to a system may raise its internal energy or enable it to expand and do
external work
NB. The first law is considered from the point of view of a gas. Consider a gas in a vessel fitted with a
piston
If the vessel is heated (heat supplied), then;
= +
Where heat is supplied, is increase in internal energy and is
external work done
NB: if we have ;
+ , Then system gains heat
, Then system loses heat
+ , Means increase in internal energy
, Decrease in internal energy
+ , System does work
, Work is done on the system
Internal energy (U) of a gas
Real gases
The internal energy has two components;
copy right reserved by Bukenya U (+250-785783786) Page 37
The kinetic energy component due to motion of its molecules (K.e is directly proportional to
temperature )
Potential energy component due to the intermolecular forces . this depends on the separation
of the molecules (volume of the gas)
Ideal gases; an ideal gas has no intermolecular forces, so its internal energy has no potential energy
component. The internal energy is independent of the volume but depends on its temperature
, where is change in temperature
WORK DONE BY A GAS WHEN EXPANDING ()
Consider a gas at a pressure p, volume V enclosed in a cylinder with frictionless movable piston
Suppose the gas expands and pushes the piston outwards by
a distance , the volume of the gas increases by
= .and the pressure reduces by
If is very small, ,can be ignored and pressure considered
to be constant . the small work done by the gas is given by
= = =
Total work done in changing volume from V1 to V2 is given by ;
= 2 .(1)
1
NB. This is the area below the P-V curve
The total work done is given by ; = 2
1
If we know the relationship between P and V , we can evaluate the
expression . i.e if pressure is taken constant ; W = P(V2V1)
Specific heat capacities of gases
Gases, unlike solids and liquids have a number of specific heat capacities
The change in temperature will produce a large change in pressure and volume (for solids, pressure can
be neglected). There are two principle heat capacities i.e one at constant pressure and the other at
constant volume
1. Principal specific heat capacity at constant volume (cv). This is the amount of heat required to
change the temperature of 1kg of a gas at constant volume by 1K. it is measured in Jkg-1K-1
copy right reserved by Bukenya U (+250-785783786) Page 38
2. Principal specific heat capacity at constant pressure (cp). This is the amount of heat required to
change the temperature of 1kg of a gas at constant pressure by 1K. it is measured in Jkg-1K-1
3. Molar principal heat capacity at constant volume (Cv).this is the amount of heat required to
change the temperature of one mole of a gas by 1K at constant volume. its units are Jmol-1K-1
4. Molar principal heat capacity at constant pressure (Cp).this is the amount of heat required to
change the temperature of one mole of a gas by 1K at constant pressure. its units are Jmol-1K-1
Relationship between CP and Cv
Consider one mole of an ideal gas heated at constant volume, so that its temperature changes by 1K
Cv is molar heat capacity at constant volume , = 1
= 2 = 0 , since v is constant
1
= +
= + 0 = .(2)
Also heat supplied ; =
= ..(3) for 1mole
Compare (2) and (3)
= .(4)
Consider one mole of an ideal gas heated at constant pressure. The gas must be enclosed in a cylinder
with a frictionless movable piston
= .(5)
The gas does external work; = 2 = (2 1 )
1
= + ; but =
Thus , = + (2 1 ) (6)
Compare equation (5) and (6)
= + (2 1 ) .(7)
For ideal gas equation ( for one mole)
1 = and 2 = ( + )
By substituting in equation (7)
copy right reserved by Bukenya U (+250-785783786) Page 39
It gives ; = .(8)
Where R, is molar gas constant
NB. Cp is greater than Cv , because when the gas is heated at constant pressure , eternal work must be
done by the gas in addition to increasing its internal energy
Particular gas changes
a) Isovolumetric process /isochoric (V is constant)
The gas must be contained in a sealed vessel
Since = 0, = 0
From , = = , n is number of moles, m is molar
mass
Or , = =
b) Isobaric process (P is constant )
The gas must be enclosed in a cylinder with a frictionless movable piston. At any instant the pressure of
the gas is equal to the external pressure
From 1st law , = 2 = (2 1 ),
1
= + .
Thus; = + (2 1 )
Example
1. At a temperature of 1000C and pressure of 1.01x105Nm-2, 1kg of steam occupies 1.63m3 but the
same mass of water occupies only 1.04x10-3m3. The specific latent heat of vaporization of water
at 1000C is 2.26x106Jkg-1. For system of 5kg of water changing to steam at 1000C at a pressure of
1.01x105Nm-2, find
i. The heat supplied to the system
ii. The work done by the system
iii. The increase in internal energy of the system
Solution
i) =
copy right reserved by Bukenya U (+250-785783786) Page 40
ii) = 2 = (2 1 )
1
iii) = +
2. A cylinder contains 4moles of oxygen gas at a temperature of 270C , the cylinder is provided with
a frictionless piston , which maintains a constant pressure of 1.0x105Pa , the gas is heated until
its temperature increases to 1270C
i. Calculate the amount of heat supplied to the gas
ii. What is the change in the internal energy of the gas ?
iii. The work done by the gas
[Cp=29.4Jmol-1K-1, gas constant R=8.31Jmol-1K-1]
Solution
i) =
ii) =
iii) = +
3. Nitrogen gas is trapped in a container by a movable piston . if the temperature of the gas is
raised from 00C to 500C , at a constant pressure of 4x105Pa and the total heat added is 3x104J .
calculate the work done by the gas [Cp =29.1Jmol-1K-1 , = 1.4]
Solution
= +
= , =
=
= +
Thus , = +
c) Isothermal process (constant temperature)
Using the first law ; = + , since = 0 for = 0
Either ,
+ = + , heat supplied does external work
Or , = , work done on gas is converted into heat
In order to achieve this , the rate at which heat is supplied must be equal to the rate of doing work
i.e + =+
Realization of isothermal process in practice
The gas must be contained in a cylinder with thin conducting walls , having frictionless piston
The gas cylinder must be surrounded by a constant temperature reservoir
The process must be carried out infinitely slowly to allow enough time for heat exchange
copy right reserved by Bukenya U (+250-785783786) Page 41
Equations for isothermal processes
For isothermal expansion from, V1 to V2 we use the equation of state
PV = nRT or PV = constant i.e for constant temperature T
Isothermal process obeys Boyles law
NB. Graphs showing variation of P with V at constant temperatures are called isothermals
Isothermal are drawn by dotted lines and the process by solid
lines
Consider an isothermal process (from V1 to V2)
Along AB; = = , or 1 1 = 2 2 =
= 2
1
2 1
But , = , then ; = 2 = ln = ln
1 1 2
d) Adiabatic process ( = 0)
This is the process where there is no heat exchange between the gas and the surrounding
From 1st law; = + , = 0.
Either; + = (gas does work at expense of internal energy)
Or, = + (work done on the gas increases internal energy)
An adiabatic expansion is accompanied by temperature decrease
(1 , 1 , 1 ) (2 , 2 , 2 )
Realization of an approximate adiabatic process in practice
copy right reserved by Bukenya U (+250-785783786) Page 42
The gas must be contained in a thick-walled poorly conducting cylinder and piston
The process must be carried out in very rapid but very small change in volume step to
minimize the escape of heat through the walls
Equations for adiabatic process
i. Relationship between p and v
=
Where = = , is the ratio of the principal heat capacities which depends on the
atomicity of the gas
Atomicity
Monatomic 1.67
Diatomic 1.40
Polyatomic 1.30
ii. Relationship between T and v
= ..(1) for adiabatic process
= ...(2) for any process
Divide (1) by (2) it gives ;
=
iii. Relationship between p and T
Raising (2) by we get ;
= .(3)
Divide (3) by (1) it gives ;
Work done in adiabatic process
Along AB, = = ..(1)
1 1
= , = 2 = 2 1 .(2)
1 1
Combining (1) and (2) gives ;
( )
=
NB. All these processes are reversible. i.e for expansion , the reverse is compression and the work
done in compression is negative since work is done on the gas
copy right reserved by Bukenya U (+250-785783786) Page 43
Consider the figure below;
, isobaric expansion
, isobaric compression
, isothermal expansion
, isothermal compression
, Adiabatic expansion
, Adiabatic compression
, isovolumetric cooling
,isovolumetric heating
Examples
1. Calculate the two principal molar heat capacities of oxygen if their ratio is 1.40
The density of oxygen at stp is 1.43kgm-3 and one standard atmosphere is 1.01x105Pa take the
molar mass of oxygen to be 32x10-3kgmol-1
2. A mass of an ideal gas of volume 400cm3 at 288K expands adiabatically and its temperature falls
to 273K
a. What is the new volume if = 1.40?
b. If it is then compressed isothermally until the pressure returns to its original value , calculate
the final volume of the gas
3. a) A cylinder with a piston contains one mole of gas at a pressure of 1.0x105Pa and temperature
of 300K. The gas is heated at constant pressure until its volume doubles. it is then compressed
isothermally back to its original volume and finally it is cooled at constant volume to its original
state
i. represent the above processes on a pv-diagram
ii. calculate the work done by the gas in the cycle (R=8.31Jmol-1K-1)
b) A gas with specific heat capacity ratio = 1.67 is made to expand adiabatically from a
volume of V1= 1.2x104cm3 and a pressure of P1= 2.5x105Nm-2. If the final pressure is 1.9x105Nm-2
find the final volume of the gas
c) Air in a cylinder has a volume of V1= 1000cm3at an initial temperature 300K and pressure
p1=1.01x105Pa. a sequence of changes are imposed on the air, assumed to behave as an ideal
gas , as shown below;
copy right reserved by Bukenya U (+250-785783786) Page 44
i. AB, the air is heated to 375K at constant pressure , calculate new volume (V2)
ii. BC , the air is compressed isothermally to a volume (V1), calculate the new pressure (P2)
iii. CA, the air is cooled at constant volume to (P1), calculate the work done on the gas
during the cycle
THE SECOND LAW
The first law stresses conservation of energy but many processes which conserve energy are not realized
in nature. The second law is a statement about which processes occur in nature and which do not. It can
be stated in a number of ways but all of which are equivalent.
Heat can flow spontaneously from a hot object to a cold one but the reverse is not spontaneous. This
statement was based partly on the study of heat engines such as steam engines, internal combustion
engines for automobiles
The heat engines (steam and internal combustion engine)
A heat engine is a device used for changing thermal energy by means of heat flow between two
different temperatures into useful work
The process of thermodynamics in a heat engine is as follows
Heat flows from the source at temperature TH to engine which
transforms part of this energy into mechanical work (W) and transfers
the remainder (Qout) to the sink (lower temperature TC)
By energy conservation; = +
copy right reserved by Bukenya U (+250-785783786) Page 45
Second law version of heat engine
No continually working heat engine can take heat from a source and use it all for work
Or ; = +
Efficiency of engine (e)
This is the ratio of the work done by the engine to the heat input at high temperature
=
This efficiency can be expressed as a percentage. It can only be 100% if no heat were exhausted to the
environment. The efficiency of the engine increases as the temperature ratio , increases . So
materials which can withstand high temperatures like lead must be used to improve on the efficiency of
the engine
NB.
i. A reservoir is the source or sink of infinite heat capacity. Its temperature remains constant
however much heat supplies or it receives
ii. In a petrol engine, the hot reservoir is got by combustion of fuel or steam in steam turbine.
The petrol or steam is called a working substance. The temperatures TH and Tc are called
working temperatures
Examples
An automobile engine has efficiency of 30% and produces an average of 20000J of mechanical work per
second during operation
i. Calculate the heat input from the reservoir per second
ii. Calculate the heat discharged in the exhaust
Internal combustion engine
These are engines which burn fuel inside the engine. e.g petrol engine and diesel engine
NB. Steam engines are not internal because its fuel combustion takes place outside the engine in
the steam boiler
The petrol engine (four strokes)
copy right reserved by Bukenya U (+250-785783786) Page 46
The petrol engine gets its energy from an exploded mixture of air and petrol vapour. It is called four
strokes because four piston strokes or movements inside the cylinder repeat themselves
continuously
The strokes are in the order; intake, compression, power and exhaust stroke as shown below
A. Intake / induction (isobaric expansion)
Inlet valve open
Piston moves down
Petro-air mixture enters the cylinder from carburetor by
atmospheric pressure
B. Compression stroke (adiabatic compression)
Piston moves up
All valves close
Petrol air mixture is compressed to about one seventh of its volume at
the top of the stroke
The mixture explodes by the spark plug and ignition takes place
C. Power stroke /expansion (isothermal expansion)
The burnt gases expand forcing the piston downwards and a power stroke is
obtained and transmitted to the crankshaft connected to the wheels
copy right reserved by Bukenya U (+250-785783786) Page 47
D. Exhausted stroke (adiabatic compression )
The piston moves up, the exhaust valve is opened and the burnt or
exhaust gases are expelled from the cylinder. The cycle is the
repeated
NB. In the four stroke engine, a power stroke is obtained in a cylinder
once every four strokes which correspond to two revolutions of the
crankshaft. With an engine of say four cylinders, the power strokes in
each cylinder are arranged following each other so that the engine
works smoothly
The spark plug
The diesel engine (four strokes)
This is also a four stroke cycle engine. During induction, only air enters the cylinder by atmospheric
pressure as the piston descends
During the compression stroke, the upstroke compresses the air to about one-sixteenth of its original
volume (which is twice the compression of petrol engine). This very high compression increases the
temperature of air considerably. At the end of the compression fuel is pumped into the cylinder by the
fuel injector and it ignites automatically
During the power stroke, oil-air mixture burns and expands pushing the piston down
During the exhaust stroke, burnt gases are expelled through exhaust stroke pot as the piston moves
upwards
Carnot engine
Sadi Carnot imagined an ideal engine which is more efficient than other engine but on the following
assumptions;
A more efficient engine using ideal substance moving in four reversible processes i.e two isothermal
and two adiabatic processes is called Carnot engine. And its cycle is called the Carnot cycle
copy right reserved by Bukenya U (+250-785783786) Page 48
Each process was done slowly that the whole process could be considered a series of equilibrium states.
The whole process could be done with no change in the magnitude of work done or heat exchange
AB the substance expands isothermally absorbing heat Qin from source temperature TH and doing
external work
BC adiabatic expansion, more work is done by substance and temperature decrease to Tc
CD isothermal compression, work is done on substance and heat is rejected to a sink at temperature Tc
DA adiabatic compression, more work is done on the substance
The total external work done by the substance is given by ; =
Efficiency, =
Using an ideal gas as working substance, the efficiency for Carnot engine is also given by , =
NB. Reversible processes are ideal and most efficient. However there is an upper theoretical limit which
exists and can be reduced by the practical factor
copy right reserved by Bukenya U (+250-785783786) Page 49
e.g for a steam turbine with working temperatures of 830K and 303K has efficiency 63% higher than the
actual 30%. The petrol engine is 25% and for a large diesel engine is about 40%
Limitations of Carnot engine
Carnot engine is considered to be unrealistic because of the following limitations
A real processes occurs more quickly and there would be turbulence in the gas and magnitude
of work done would change
Friction cannot be eliminated completely and the processes of friction cannot be reversed. thus
the process is irreversible
Example
1. A steam engine operates between 5000C and 2700C. what is the maximum efficiency of the
engine
2. An engine manufacturer makes the following claims on his products. An engines heat input per
second is 9.0KJ per second at 435K. The heat output per second is 4.0KJ per second at 285K.
prove the correctness of this claim
Refrigerators, air conditioners and heat pumps
The operation of refrigerators, air conditioners and heat pumps is just the reverse of heat engines. Each
operates to transfer heat out of a cool environment into a warm environment
Energy transfer in a refrigerator or air conditioner
Heat is taken from a low temperature region Tc and greater
amount of heat is exhausted at a high temperature TH
The work (w) is usually done by an electric compressor motor
which compresses a fluid
From the diagram ; = +
This is the second of thermodynamic in relation to the
refrigerator and heat pumps
Second law version of refrigerator
Heat cannot be transferred continually from one body to another at high temperature unless external
work is done
copy right reserved by Bukenya U (+250-785783786) Page 50
Coefficient of performance (COP)
This is defined as the ratio of the heat removed from low temperature Qin to the work done to remove
the heat
= =
For an ideal refrigerator (Carnot refrigerator)
=
NB.
i. An air conditioner works very much like a refrigerator, although the actual construction
details are different. the air conditioner takes heat Qc from inside room at low temperature
and deposits heat QH outside to the environment at higher temperature
ii. Heat pump is a device that can heat a house in winter by using an electric motor and does
work (w), to take heat Qc from outside at low temperature and delivers heat QH to the
warmer inside of the house. Since the sole function is to heat the room,
= ..(it is necessarily greater than 1)
Some heat pumps can be turned around to work as air conditioners in summer
Structure and working of a refrigerator
Cooling in a refrigerator takes place
when a volatile liquid (Freon)
evaporates inside a copper coil
surrounding the freezing box as show
below
Inside the cooling box, the liquid Freon
absorbs heat from the surrounding
and evaporates. The vapour flows in
the copper tube s to the compressor
run by an electric motor.
The compressor compresses the
vapour through the outlet valve to the
condenser tubes at high pressure
forcing heat out by conduction
through the cooling fins. When heat is
lost, the gas condenses to liquid but at
high pressure and forced to rise into
copy right reserved by Bukenya U (+250-785783786) Page 51
the capillary tube back to the cooling box. The process repeats its self
Examples
1. A freezer has a COP of 3.8 and uses 200W of power. How long would it take to freeze an ice-
cube tray that contains 600g of water at 00C. specific latent heat of fusion of ice is 333KJkg-1
2. A heat pump has coefficient of performance of 3.0 and is rated to do work at 1500W
a) How much heat can it add to a room per second?
b) If the heat pump were turned around to act as an air conditioner in the summer, what
would be the expected coefficient of performance , assuming other factors kept constant
copy right reserved by Bukenya U (+250-785783786) Page 52
Você também pode gostar
- Mechanics of the Household: A Course of Study Devoted to Domestic Machinery and Household Mechanical AppliancesNo EverandMechanics of the Household: A Course of Study Devoted to Domestic Machinery and Household Mechanical AppliancesAinda não há avaliações
- Physics 2: Heat TransferDocumento22 páginasPhysics 2: Heat TransferAndreana Amor GulayAinda não há avaliações
- Chemistry For Engineers - 1 Energy - Topic 02 - Sensible Heat-Heat Capacity-CalorimetryDocumento7 páginasChemistry For Engineers - 1 Energy - Topic 02 - Sensible Heat-Heat Capacity-CalorimetryJacob JimenezAinda não há avaliações
- Analytical Chemistry of Zirconium and Hafnium: International Series of Monographs in Analytical ChemistryNo EverandAnalytical Chemistry of Zirconium and Hafnium: International Series of Monographs in Analytical ChemistryAinda não há avaliações
- Specific HeatDocumento2 páginasSpecific HeatAina Beñasfre RafalesAinda não há avaliações
- Phys 211 11a.law of Heat Exchange and Change of PhaseDocumento6 páginasPhys 211 11a.law of Heat Exchange and Change of PhaseRegina May Dela CruzAinda não há avaliações
- Activity 6 For Laboraory (Bautista)Documento3 páginasActivity 6 For Laboraory (Bautista)Dan Patrick BautistaAinda não há avaliações
- Heat Transfer WorksheetDocumento5 páginasHeat Transfer Worksheetshahab waheedAinda não há avaliações
- Thermal 2 Specific HeatDocumento13 páginasThermal 2 Specific HeatBrian TotmanAinda não há avaliações
- Heat Capacity and Specific HeatDocumento6 páginasHeat Capacity and Specific Heatcrazyen28100% (2)
- Slaid Topik 6-Temperature and Heat2Documento46 páginasSlaid Topik 6-Temperature and Heat2Md KhairulAinda não há avaliações
- Thermodynamics RevisionDocumento11 páginasThermodynamics RevisionbobAinda não há avaliações
- Thermal Physics 2 of 3Documento47 páginasThermal Physics 2 of 3Kyoko LevitaAinda não há avaliações
- TranscriptDocumento29 páginasTranscriptNurAfifah OsmanAinda não há avaliações
- L10 - Temperature, Heat, ThermodynamicsDocumento9 páginasL10 - Temperature, Heat, ThermodynamicsKristineLyra BucayoAinda não há avaliações
- TemperatureDocumento50 páginasTemperatureJohn HobanAinda não há avaliações
- A2 53a ThermalenergyDocumento39 páginasA2 53a ThermalenergyHany ElGezawyAinda não há avaliações
- Phys 211 11.temperatureDocumento5 páginasPhys 211 11.temperatureRegina May Dela CruzAinda não há avaliações
- G10 CH 8 Change of State11May07Documento9 páginasG10 CH 8 Change of State11May07Tun Lin AungAinda não há avaliações
- 19 - Thermal EnergyDocumento22 páginas19 - Thermal EnergyAM - 12CA 812699 Louise Arbour SSAinda não há avaliações
- Lab 4 Phy400Documento11 páginasLab 4 Phy400AUNI SAKINAH BINTI NOR HASNAN KM-PelajarAinda não há avaliações
- Module 4.1 CalorimetryDocumento8 páginasModule 4.1 CalorimetryCanskie Gabucay - CoEd100% (1)
- Physics 2 Lab 1Documento5 páginasPhysics 2 Lab 1Peter ZawyckyAinda não há avaliações
- Exercise - III: (Subjective Problems)Documento3 páginasExercise - III: (Subjective Problems)RishabhAinda não há avaliações
- Grammar College, Kano: Sss Two (2) Week EightDocumento7 páginasGrammar College, Kano: Sss Two (2) Week EightMusa IsyakuAinda não há avaliações
- Thermal 2 Specific HeatDocumento13 páginasThermal 2 Specific HeatOmaru NimagaAinda não há avaliações
- Module Physics. LastDocumento19 páginasModule Physics. LastjlysntsAinda não há avaliações
- Experiment 4: Specific Heat CapacityDocumento8 páginasExperiment 4: Specific Heat Capacitysyaf100% (1)
- Calorimetry LabDocumento6 páginasCalorimetry LabchristineAinda não há avaliações
- 02 - HeatDocumento16 páginas02 - HeatAshmiVeeraAinda não há avaliações
- The Effects of Heat On MatterDocumento5 páginasThe Effects of Heat On MattercirclelineAinda não há avaliações
- 3.9 Quantity of HeatDocumento38 páginas3.9 Quantity of Heatcicilywairimu25Ainda não há avaliações
- Physics Lesson Note SS2 2nd Term 2022 - 1Documento103 páginasPhysics Lesson Note SS2 2nd Term 2022 - 1isaac bakare100% (7)
- Calorimetry Villanueva CristyMaeDocumento11 páginasCalorimetry Villanueva CristyMaeCristy Mae U. VillanuevaAinda não há avaliações
- 10.4.2 HEAT (Section 2 and 3) - TeacherDocumento16 páginas10.4.2 HEAT (Section 2 and 3) - TeacherKurdishAinda não há avaliações
- Heat Science Study Note Question and AnswerDocumento4 páginasHeat Science Study Note Question and AnswerdenyAinda não há avaliações
- Heat TransferDocumento57 páginasHeat TransferSatyam PandyaAinda não há avaliações
- Heat Transfer AnsDocumento8 páginasHeat Transfer AnsejAinda não há avaliações
- 1 - Electrochemical Energy-1aDocumento3 páginas1 - Electrochemical Energy-1aMae TadaAinda não há avaliações
- Lab 12Documento15 páginasLab 12Saddam HossainAinda não há avaliações
- Heat P1P2Documento4 páginasHeat P1P2Sharvinder SinghAinda não há avaliações
- Lab ReportDocumento7 páginasLab ReportAristotle LeventidisAinda não há avaliações
- Physics2 SheetDocumento6 páginasPhysics2 SheetBola AkramAinda não há avaliações
- Heat and Thermodynamics: By: Arra C. QuitanegDocumento16 páginasHeat and Thermodynamics: By: Arra C. Quitaneg2FPharmacyAinda não há avaliações
- Chemistry 30 Thermochemical Changes Practice 1Documento9 páginasChemistry 30 Thermochemical Changes Practice 1Alaa mohamed Shaat BAAinda não há avaliações
- HKDSE Phy 1A A2 - AllDocumento8 páginasHKDSE Phy 1A A2 - Alljackson wongAinda não há avaliações
- Lab Report Physic210Documento5 páginasLab Report Physic210Ahmed FiziAinda não há avaliações
- 9 Heat & Thermal EnergyDocumento80 páginas9 Heat & Thermal Energy2FPharmacyAinda não há avaliações
- Measuring The Enthalpy of Combustion of Ethanol IB Chemistry SL Lab ReportDocumento7 páginasMeasuring The Enthalpy of Combustion of Ethanol IB Chemistry SL Lab Reportxavier bourret sicotte76% (17)
- 13B Heat of FusionDocumento8 páginas13B Heat of FusionManish KumarAinda não há avaliações
- Thermochemistry (Solutions)Documento16 páginasThermochemistry (Solutions)MarikAinda não há avaliações
- Temp and Heat Chapter 10Documento18 páginasTemp and Heat Chapter 10erikaAinda não há avaliações
- Thermochem Solved Practice Problems PDFDocumento9 páginasThermochem Solved Practice Problems PDFRez Balayo JanabanAinda não há avaliações
- Lab Experiment: Specific Heat of MetalDocumento13 páginasLab Experiment: Specific Heat of MetalHafiz Mohd Basir100% (2)
- The Study of Heat and Work and State FunctionsDocumento39 páginasThe Study of Heat and Work and State Functions翁绍棠Ainda não há avaliações
- Thermochemistry: Prepared By: Ron Eric B. LegaspiDocumento42 páginasThermochemistry: Prepared By: Ron Eric B. LegaspiRon Eric Legaspi100% (1)
- Temperature and Its Measurement: - When The Physical Properties Are No Longer Changing, The Objects Are Said To Be inDocumento17 páginasTemperature and Its Measurement: - When The Physical Properties Are No Longer Changing, The Objects Are Said To Be inPriyanga PremAinda não há avaliações
- Heat Capacities Evaporation Freezing and Linear ExpansivityDocumento19 páginasHeat Capacities Evaporation Freezing and Linear Expansivitykasumbaashraf3Ainda não há avaliações
- Lesson Plan 7 - HeatDocumento9 páginasLesson Plan 7 - Heatjeanette Prades91% (23)
- Fiber Optic Chemical Sensors Biosensors PDFDocumento2 páginasFiber Optic Chemical Sensors Biosensors PDFKatreenaAinda não há avaliações
- Flash and Fire Point of Lubricant ExperimentDocumento5 páginasFlash and Fire Point of Lubricant ExperimentShanti Kiran Z100% (1)
- Cambridge IGCSE™: Combined Science 0653/42 October/November 2020Documento9 páginasCambridge IGCSE™: Combined Science 0653/42 October/November 2020keyzainbAinda não há avaliações
- Tugas Teknik Reaksi Kimia Lanjut EXAMPLE 10.2 & PROBLEM 10.3Documento13 páginasTugas Teknik Reaksi Kimia Lanjut EXAMPLE 10.2 & PROBLEM 10.3Andre Fahriz Perdana HarahapAinda não há avaliações
- Chemistry Paper 1 2009Documento7 páginasChemistry Paper 1 2009Robert EdwardsAinda não há avaliações
- Tutorial 8Documento3 páginasTutorial 8CHANDAN RAJAinda não há avaliações
- Bipyramid-Templated Synthesis of MonodisperseDocumento9 páginasBipyramid-Templated Synthesis of MonodisperseAdrianoDSAinda não há avaliações
- FHGGHDocumento3 páginasFHGGHsangeethaAinda não há avaliações
- Aplication Fat Extraction FoodDocumento6 páginasAplication Fat Extraction FoodKu BonAinda não há avaliações
- Ma. Jade R. Agres - Pre Lab Experiment No.3Documento8 páginasMa. Jade R. Agres - Pre Lab Experiment No.3Ma. Jade AgresAinda não há avaliações
- Longuet-Higgins: Studies in Molecular Orbital Theory I: Resonance & Molecular Orbitals in Unsaturated HydrocarbonsDocumento11 páginasLonguet-Higgins: Studies in Molecular Orbital Theory I: Resonance & Molecular Orbitals in Unsaturated HydrocarbonsvanalexbluesAinda não há avaliações
- Quantitative Determination of Total Hardness of WaterDocumento11 páginasQuantitative Determination of Total Hardness of WaterJoshua Oliveros50% (2)
- QB PDFDocumento18 páginasQB PDFShivani0% (1)
- Experiment 5: Chemical Kinetics - The Iodine Clock ReactionDocumento10 páginasExperiment 5: Chemical Kinetics - The Iodine Clock ReactionLero LeroAinda não há avaliações
- Thermo Sheet7 NewDocumento3 páginasThermo Sheet7 NewRodney SalazarAinda não há avaliações
- Jee Main 2021 Mar 18 First Shift PaperDocumento15 páginasJee Main 2021 Mar 18 First Shift Papershivam singhAinda não há avaliações
- Annotated-Acid-base EquilibriaDocumento11 páginasAnnotated-Acid-base EquilibriaVECNAAinda não há avaliações
- Prob Set 10Documento5 páginasProb Set 10Payal SAinda não há avaliações
- Aspen AdsimDocumento307 páginasAspen Adsimkiny81100% (3)
- Compositional and Structural Characterization of Waxes Isolated From BitumensDocumento8 páginasCompositional and Structural Characterization of Waxes Isolated From BitumensMa JamieAinda não há avaliações
- Material Property Charts-W4Documento15 páginasMaterial Property Charts-W4Fara AsilaAinda não há avaliações
- Bs6088-Aiipl 202203011546377064Documento4 páginasBs6088-Aiipl 202203011546377064Vijendra KumarAinda não há avaliações
- Isentropic Process PDFDocumento2 páginasIsentropic Process PDFJeebee Logroño AloAinda não há avaliações
- Leak CalcDocumento3 páginasLeak Calckisan singhAinda não há avaliações
- Saft MieDocumento93 páginasSaft MiePatrice PariAinda não há avaliações
- Chapter 10 - Atoms in Combination, The Chemical BondDocumento40 páginasChapter 10 - Atoms in Combination, The Chemical BondMade SandhyanaAinda não há avaliações
- FinalDocumento4 páginasFinalSimge DemirAinda não há avaliações
- Acid Base Titration Post LabDocumento3 páginasAcid Base Titration Post Labapi-532693623Ainda não há avaliações
- Unit-Wise Weightage From NEET Exam 2020Documento5 páginasUnit-Wise Weightage From NEET Exam 2020pratikAinda não há avaliações