Escolar Documentos
Profissional Documentos
Cultura Documentos
Conductometric Titration: Determination of The Strength of A Solution of Hydrochloric Acid (HCL) by A Standard Solution of Sodium Hydroxide (Naoh)
Enviado por
Paolo NaguitTítulo original
Direitos autorais
Formatos disponíveis
Compartilhar este documento
Compartilhar ou incorporar documento
Você considera este documento útil?
Este conteúdo é inapropriado?
Denunciar este documentoDireitos autorais:
Formatos disponíveis
Conductometric Titration: Determination of The Strength of A Solution of Hydrochloric Acid (HCL) by A Standard Solution of Sodium Hydroxide (Naoh)
Enviado por
Paolo NaguitDireitos autorais:
Formatos disponíveis
CONDUCTOMETRIC TITRATION: DETERMINATION OF THE STRENGTH OF A
SOLUTION OF HYDROCHLORIC ACID (HCl) BY A STANDARD SOLUTION OF
SODIUM HYDROXIDE (NaOH)
Aguilar, J., Antazo, F., Lumayag, L., Naguit, P., Ragos, E.
Department of Chemistry, College of Science
University of Santo Tomas
ABSTRACT
Conductivity the degree to which a specified material conducts electricity, calculated as the ratio
of the current density in the material to the electric field that causes the flow of current. It is the
reciprocal of the resistivity. Conductometric titration is a type of titration in which the electrolytic
conductivity of the reaction mixture is continuously monitored as one reactant is added. The
equivalence point is the point at which the conductivity undergoes a sudden change. In this
experiment, HCl is titrated with NaOH. Based from the graph, the conductivity decreases until
the end point is reached, where excess NaOH brings about a gradual increase in conductivity.
The strength of the acid was calculated with a value at 0.085 N. In conclusion with these
experiment, conductometric titration refers to the species in the solution are converted to non-
ionic for by neutralization, precipitation, etc. are of more value.
INTRODUCTION conductance are associated with the
change in concentration of the two most
Conductometric titration is a method in highly conducting ions-hydrogen and
which we determine the point where hydroxyl ions. As the hydrochloric acid is
reaction is completed with the help of titrated with the sodium hydroxide, the
conductometer that measures the changes hydrogen ions are progressively replaced by
in conductance of solution produced by the the sodium ions decreasing the
ions in the solution[1]. This type of titration conductance of the solution. After the end
is where the electrolytic conductivity of the point, the conductance of the solution
reaction mixture is continuously monitored increases due to the presence of excess
as once reactant is added. This titration hydroxide ions [2].
makes use of a conductometer in which the
principle of ohm's law is observed where the METHODOLOGY
current strength flowing through a conductor
Upon performing the conductometric
is directly proportional to the potential
titration, the needed materials, glasswares
difference and inversely proportional to the
and primary equipment namely the
resistance of the conductor [1].
conductometer were prepared. The
conductivity cell was rinsed off a few times
with distilled water. Then, 20 milliliters of 0.1
M HCl was pipetted into a beaker
consequently, the conductivity cell was
The equivalence point is where the
dipped onto the solution. the students made
conductivity undergoes a sudden change
sure that the temperature of the sample
[2]. An increase or decrease in the
solution control was set accordingly at a Figure 1. Schematic presentation of the
room temperature. Addition of NaOH titration curves for the titration of HCl
solution from the burette was done by against NaOH
dropping a few amounts of the solution.
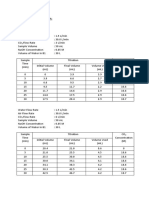
Measurement of the conductance using the The table below shows the results of
conductometer was determined every after the resulting conductance measured on the
addition of the NaOH solution to the Analyte amount of NaOH.
containing the HCl solution. The
conductance of each addition of the NaOH
solution was noted. Subsequently, a graph Table 1. Amount of NaOH and resulting
between conductance versus volume of the conductance
titrant specifically, NaOH solution was Volume Conductance Volume Conductance
plotted. NaOH NaOH
(mL) (mL)
RESULTS AND DISCUSSION
0.00 19.96 9.5 12.41
Conductometric titration is a 0.5 19.96 10.0 11.93
scientific process in determining the quantity
of a sample by measuring the conductance 1.0 19.95 10.5 11.48
of the solution until the end point is
1.5 19.94 11.0 10.86
reached.[1] The end point of the titration is
indicated by an indicator which shows that 2.0 19.92 11.5 10.35
the amount of the reactant that is necessary
for the reaction to be complete has been 2.5 19.89 12.0 9.82
added to the solution.[2] 3.0 19.86 12.5 9.28
In the titration of HCl by NaOH, the 3.5 19.80 13.0 8.81
conductivity decreases until the end point is
4.0 19.71 13.5 8.35
reached, where excess NaOH brings about
a gradual increase in conductivity as shown 4.5 19.30 14.0 7.77
in the figure below. [3]
5.0 18.78 14.5 7.09
5.5 18.02 15.0 6.75
6.0 17.25 15.5 6.22
6.5 16.72 16.0 5.87
7.0 15.96 16.5 5.37
7.5 14.98 17.0 5.03
8.0 14.57 17.5 5.32
8.5 13.96 18.0 5.54
9.0 13.36 18.5 5.74
V1S1=V2S2
19.0 5.91 22.5 7.17
Where V1= Volume of HCl taken
19.5 6.10 23.0 7.32 S1= Strength of Acid
20.0 6.30 23.5 7.52 V2= Volume NaOH at equivalence
point
20.5 6.48 24.0 7.63 S2= Strength of NaOH (0.1 x N)
21.0 6.64 24.5 7.75
Therefore, when rearranging the equation to
21.5 6.82 25.0 7.9 isolate S1, the equation is,
S1= (V2S2/20) (N)
22.0 7.02
Based on the equation, the strength
The table showed that the of the HCl acid was 0.085 N.
equivalence point of the titration was when
17.0 mL of NaOH was added. Furthermore, CONCLUSION
the graph below shows that the
conductance decreased upon the addition The electrical conductance of a
of NaOH in the HCl solution which signifies solution is a measure of its currents carrying
that the Na+ ions are slowly replacing the capacity and therefore determined by the
fast H+ ions in the solution.[3] total ionic strength. It is a non-specific
property and for this reason direct
conductance measurement are of little use
unless the solution contains only the
electrolyte to be determined or the
concentrations of other ionic species in the
solution are known. Shown from data
gathered the end point lies on 17.0 mL with
a conductance at 5.03 where there is a
decrease in conductance and a gradual
increase in the conductance of the solution
Figure 2. Graph on the titration curve for caused by an excess NaOH. Furthermore,
the titration of HCl against NaOH the strength of the acid was calculated with
a value at 0.085 N. In addition,
The decrease in the conductance of conductometric titrations, in which the
the solution continues until it reached the species in the solution are converted to non-
end point where the decrease in ionic for by neutralization, precipitation, etc.
conductance stops and excess NaOH in the are of more value.
solution caused a gradual increase in the
conductance of the solution.[3]
The strength of the acid was also
calculated using the equation,
REFERENCES
[1] Werkmeister, P., & Cleveland State
University. (1974). Automation of a
conductometric titration. Cleveland:
Werkmeister.
[2] Himpler, H. A., Schweikert, E. A.,
Barnes, G. E., Pettersen, R. C., Rowe, M.
W., & Texas A & M University,. (1979).
Analytical applications of electrode sensors.
[3] Royal Society of Chemistry (2018).
Conductometric titration. Retrieved March
15, 2018 from
http://www.rsc.org/publishing/journals/prosp
ect/ontology.asp?id=CMO:0002143&MSID=
C3PY00181D
[4] Author (n.d). End point. Retrieved March
15, 2018 from
http://www.chem.purdue.edu/jmol/gloss/end
point.html
[5] Banica, F.G. (2011). Conuctometric
Titration. Retrieved March 15, 2018 from
http://folk.ntnu.no/floban/KJ%20%202051/C
onductometry.pdf
Você também pode gostar
- Chem 214 Homework #2 Due February 22, 2021 by 11:59 PM On MoodleDocumento2 páginasChem 214 Homework #2 Due February 22, 2021 by 11:59 PM On MoodleNguyệt BìnhAinda não há avaliações
- 11chem301 Argentometric MethodsDocumento16 páginas11chem301 Argentometric MethodsPaolo NaguitAinda não há avaliações
- Feed Analysis Standard Operation (Analysis) Procedure SOPDocumento27 páginasFeed Analysis Standard Operation (Analysis) Procedure SOPZiauddeen Noor100% (1)
- Bab 8Documento28 páginasBab 8Mohammad Farhan S0% (1)
- CHM170L - Final Report 3Documento5 páginasCHM170L - Final Report 3Katrina BucudAinda não há avaliações
- Safety Hazards of Common ChemicalsDocumento5 páginasSafety Hazards of Common ChemicalsPorkkodi SugumaranAinda não há avaliações
- CELCHA2 Study GuidesDocumento7 páginasCELCHA2 Study GuidesEsther100% (1)
- 4500 NO2 - NitritoDocumento3 páginas4500 NO2 - NitritoPenelope MeloAinda não há avaliações
- Enplate Ni 434 EDocumento10 páginasEnplate Ni 434 EMagda FaragAinda não há avaliações
- Lab Report Exp 6 - Determination of The Percentage of Ligands in Coordination CompoundsDocumento11 páginasLab Report Exp 6 - Determination of The Percentage of Ligands in Coordination CompoundsNabilah Hairman100% (20)
- Sedimentation PaperDocumento4 páginasSedimentation Paperapi-3702623100% (4)
- Diffusion in Gases and LiquidsDocumento39 páginasDiffusion in Gases and LiquidsJocelyn CorpuzAinda não há avaliações
- Packed Distillation Column ExperimentDocumento20 páginasPacked Distillation Column ExperimentChan Chun ChenAinda não há avaliações
- Chemical Engineering Principles Recycle Process OptimizationDocumento8 páginasChemical Engineering Principles Recycle Process OptimizationJakaAbuAinda não há avaliações
- Volume 6 Solns ManualDocumento8 páginasVolume 6 Solns ManualshaaaaaaaaareeeeeeeAinda não há avaliações
- 7405Documento8 páginas7405Ebby OnyekweAinda não há avaliações
- Experiment 2: Determination of A Mixture of Xylene Isomers Using Infrared (Ir) SpectrometerDocumento8 páginasExperiment 2: Determination of A Mixture of Xylene Isomers Using Infrared (Ir) SpectrometerNur Atiqah Mohd RedzuanAinda não há avaliações
- CTD II Deg Mid (April-2019)Documento1 páginaCTD II Deg Mid (April-2019)Ramesh BabuAinda não há avaliações
- PDFDocumento88 páginasPDFMuralidharanAinda não há avaliações
- Ch12P1 VLE Models by Margules Van Laar and Wilson EquationsDocumento2 páginasCh12P1 VLE Models by Margules Van Laar and Wilson Equationshana faqihAinda não há avaliações
- 練習單3 2Documento8 páginas練習單3 2Marco RezendeAinda não há avaliações
- Lecture 9Documento4 páginasLecture 9Asif AliAinda não há avaliações
- Lab Report 2Documento14 páginasLab Report 2SyafiyatulMunawarahAinda não há avaliações
- Identifying Flow Type Using Reynolds ApparatusDocumento5 páginasIdentifying Flow Type Using Reynolds ApparatusKonem SolutionsAinda não há avaliações
- Chemical Engineering Laboratory - AgitationDocumento6 páginasChemical Engineering Laboratory - AgitationRab SangcalAinda não há avaliações
- Exp 4 Batch Evaporative Crystallization PDFDocumento9 páginasExp 4 Batch Evaporative Crystallization PDFmirza farhanAinda não há avaliações
- Tutorial Reactive SystemsDocumento33 páginasTutorial Reactive Systemssiti azilaAinda não há avaliações
- Motion of Particles Through Fluids 221Documento27 páginasMotion of Particles Through Fluids 221SAYEEAinda não há avaliações
- TK-315 CPI2 - 1 - NonReacting SystemsDocumento34 páginasTK-315 CPI2 - 1 - NonReacting SystemsMuhammad IkbalAinda não há avaliações
- Tgs LeachingDocumento1 páginaTgs LeachingShanaz100% (1)
- Reynolds QuestionDocumento1 páginaReynolds QuestionAstrJohn ObikaonuAinda não há avaliações
- Laboratory Manual For Determining Oil and Grease For Raw and Treated EffluentDocumento3 páginasLaboratory Manual For Determining Oil and Grease For Raw and Treated EffluentAnas YuzairiAinda não há avaliações
- Cell kinetics-IIIDocumento12 páginasCell kinetics-IIIVineet Kumar ThakurAinda não há avaliações
- Chapter 4Documento43 páginasChapter 4aliAinda não há avaliações
- Wetted-Wall Column: Separation ProcessDocumento3 páginasWetted-Wall Column: Separation ProcessHuzaifa AftabAinda não há avaliações
- Soal PR TermodinamikaDocumento10 páginasSoal PR TermodinamikaanjaniAinda não há avaliações
- IYOHA COLLINS 16CF020531 Batch Reactor ReportDocumento19 páginasIYOHA COLLINS 16CF020531 Batch Reactor ReportDavid OvieAinda não há avaliações
- Unit Operation 1: CHE 321 (3 Units)Documento70 páginasUnit Operation 1: CHE 321 (3 Units)Glory UsoroAinda não há avaliações
- Atkkkkkkkk BaruuuuuDocumento27 páginasAtkkkkkkkk BaruuuuuSavira AAinda não há avaliações
- Problem 3.17Documento6 páginasProblem 3.17Ralph Evidente100% (1)
- Seatwork Sepa Graph Number 3Documento3 páginasSeatwork Sepa Graph Number 3Yasmin KayeAinda não há avaliações
- CAP 10 Principles of Chemical Engineering Processes Material and Energy Balances Second Edition T.LDocumento7 páginasCAP 10 Principles of Chemical Engineering Processes Material and Energy Balances Second Edition T.LAnel Viridiana Alfonso BocarandoAinda não há avaliações
- EVAPORATORDocumento50 páginasEVAPORATORnur irfana mardiyah100% (1)
- Solution: 0.05 Moisture (Feed in Tons/hr) 0.95 Dry 9.8 (Tons/hr Dry Solid) Fdry 124.45 P 114.65Documento7 páginasSolution: 0.05 Moisture (Feed in Tons/hr) 0.95 Dry 9.8 (Tons/hr Dry Solid) Fdry 124.45 P 114.65api-3728602Ainda não há avaliações
- Drying: Merry Jessah S. TorresDocumento6 páginasDrying: Merry Jessah S. TorresFrancis Val FranciscoAinda não há avaliações
- Activation Energy of an Ionic ReactionDocumento10 páginasActivation Energy of an Ionic ReactionHanif YusofAinda não há avaliações
- Mechanical Phase Separations Chapter Explains Particle Separation DevicesDocumento36 páginasMechanical Phase Separations Chapter Explains Particle Separation DevicesIvan MarmilichAinda não há avaliações
- Mass Transfer - II: Distillation Processes ExplainedDocumento39 páginasMass Transfer - II: Distillation Processes ExplainedSMIT CHRISTIANAinda não há avaliações
- Conclusion, Recoomendation, Reffenrence, Lab 2, CHE 485Documento2 páginasConclusion, Recoomendation, Reffenrence, Lab 2, CHE 485MOHD MU'IZZ BIN MOHD SHUKRIAinda não há avaliações
- Heat of ReactionDocumento8 páginasHeat of ReactionNece Jean Tagam83% (6)
- Lab Report GCDocumento8 páginasLab Report GCAyish MataAinda não há avaliações
- Gas Absorption Lab ReportDocumento3 páginasGas Absorption Lab ReportNur Shaffikha Azmi100% (1)
- Duhem's theorem determines equilibrium stateDocumento1 páginaDuhem's theorem determines equilibrium stateZafirahAhmadFauzi100% (1)
- Assignment II - CL 308Documento2 páginasAssignment II - CL 308Satyam SaranAinda não há avaliações
- CLO-2 Packed and Fluidized BedsDocumento44 páginasCLO-2 Packed and Fluidized BedskhalifaAinda não há avaliações
- Chapter 5 .Molecular Diffusion in BiologicalDocumento31 páginasChapter 5 .Molecular Diffusion in BiologicalNur FarazielaAinda não há avaliações
- Reactor Design Equations and ExamplesDocumento25 páginasReactor Design Equations and ExamplesTÍN Phạm Nguyễn TrọngAinda não há avaliações
- Un1 MergedDocumento467 páginasUn1 MergedHariHaran KAinda não há avaliações
- Water Treatment Water Treatment: Lecture 7: DisinfectionDocumento17 páginasWater Treatment Water Treatment: Lecture 7: DisinfectionAbo-Khaled MohammedAinda não há avaliações
- CHE485 Chemistry Laboratory Lab 4 Stoichiometric Analysis of Thermal Decomposition Reaction of Sodium BicarbonateDocumento11 páginasCHE485 Chemistry Laboratory Lab 4 Stoichiometric Analysis of Thermal Decomposition Reaction of Sodium BicarbonatetuesdayofjulyAinda não há avaliações
- Process Engineering Thermodynamics: Dr. Dharmendra Kumar Bal Assistant Professor (SR.) ScaleDocumento50 páginasProcess Engineering Thermodynamics: Dr. Dharmendra Kumar Bal Assistant Professor (SR.) ScaleAABID SHAIKAinda não há avaliações
- Lab 1Documento12 páginasLab 1JoeJeanAinda não há avaliações
- Aspirin (Conducto) ProductionDocumento11 páginasAspirin (Conducto) Productionapi-26797747100% (1)
- Practica ConductimetriaDocumento21 páginasPractica Conductimetriajoss villagomezAinda não há avaliações
- 1.18 Radiologic Anatomy of The Head and NeckDocumento2 páginas1.18 Radiologic Anatomy of The Head and NeckPaolo NaguitAinda não há avaliações
- 1.14 ANATOMY - The Pharynx Landmarks - MusclesDocumento3 páginas1.14 ANATOMY - The Pharynx Landmarks - MusclesPaolo NaguitAinda não há avaliações
- 1.15 ANATOMY - The LarynxDocumento2 páginas1.15 ANATOMY - The LarynxPaolo NaguitAinda não há avaliações
- Vector 3Documento55 páginasVector 3Paolo NaguitAinda não há avaliações
- 1.17 Anatomy-Neurovascular Structures of Head and NeckDocumento2 páginas1.17 Anatomy-Neurovascular Structures of Head and NeckPaolo NaguitAinda não há avaliações
- VectorDocumento39 páginasVectorPaolo Naguit100% (1)
- 1.12 ANATOMY - The Ears Surface Anatomy and Landmarks, External, Middle and Inner Portions, Blood Vessels and NervesDocumento5 páginas1.12 ANATOMY - The Ears Surface Anatomy and Landmarks, External, Middle and Inner Portions, Blood Vessels and NervesPaolo NaguitAinda não há avaliações
- 1.13 ANATOMY - The Teeth, Palate, Internal Auditory Tube and TonsilsDocumento2 páginas1.13 ANATOMY - The Teeth, Palate, Internal Auditory Tube and TonsilsPaolo NaguitAinda não há avaliações
- 1.11 ANATOMY - The Nose and Paransal SinusesDocumento4 páginas1.11 ANATOMY - The Nose and Paransal SinusesPaolo NaguitAinda não há avaliações
- 1.10 ANATOMY - The Eyeball - Surface Anatomy - Landmarks - Extrinsic - Intrinsic MusclesDocumento3 páginas1.10 ANATOMY - The Eyeball - Surface Anatomy - Landmarks - Extrinsic - Intrinsic MusclesPaolo NaguitAinda não há avaliações
- Titration Curve of Amino AcidsDocumento3 páginasTitration Curve of Amino AcidsPaolo NaguitAinda não há avaliações
- 1.16 The TongueDocumento2 páginas1.16 The TonguePaolo NaguitAinda não há avaliações
- 12chem301 EDTA Titration PDFDocumento17 páginas12chem301 EDTA Titration PDFPaolo NaguitAinda não há avaliações
- Smith1985 PDFDocumento10 páginasSmith1985 PDFLizeth Paola TellezAinda não há avaliações
- Coulometric Titration of Ascorbic Acid With Electrogenerated IodineDocumento5 páginasCoulometric Titration of Ascorbic Acid With Electrogenerated IodinePaolo NaguitAinda não há avaliações
- BCA Assay Paper - 1988Documento7 páginasBCA Assay Paper - 1988Paolo NaguitAinda não há avaliações
- Dry About 1Documento1 páginaDry About 1Paolo NaguitAinda não há avaliações
- BradfordDocumento7 páginasBradfordJonatas LopesAinda não há avaliações
- PDFDocumento124 páginasPDFratno budiyantoAinda não há avaliações
- HBB Hemoglobin, Beta (Bos Taurus (Cattle) )Documento1 páginaHBB Hemoglobin, Beta (Bos Taurus (Cattle) )Paolo NaguitAinda não há avaliações
- PDFDocumento124 páginasPDFratno budiyantoAinda não há avaliações
- Hba-A1 Hemoglobin Alpha, Adult Chain 1 (Mus Musculus (House Mouse) )Documento1 páginaHba-A1 Hemoglobin Alpha, Adult Chain 1 (Mus Musculus (House Mouse) )Paolo NaguitAinda não há avaliações
- PV92 PCR Kit Manual PDFDocumento104 páginasPV92 PCR Kit Manual PDFPaolo NaguitAinda não há avaliações
- HBZP1 Hemoglobin Subunit Zeta Pseudogene 1 (Homo Sapiens (Human) )Documento1 páginaHBZP1 Hemoglobin Subunit Zeta Pseudogene 1 (Homo Sapiens (Human) )Paolo NaguitAinda não há avaliações
- BFG Chapter2 AccessInformation v04Documento67 páginasBFG Chapter2 AccessInformation v04Paolo NaguitAinda não há avaliações
- Hbb-Ar Hemoglobin, Activating Region (Mus Musculus (House Mouse) )Documento7 páginasHbb-Ar Hemoglobin, Activating Region (Mus Musculus (House Mouse) )Paolo NaguitAinda não há avaliações
- HBA Hemoglobin, Alpha 2 (Bos Taurus (Cattle) )Documento1 páginaHBA Hemoglobin, Alpha 2 (Bos Taurus (Cattle) )Paolo NaguitAinda não há avaliações
- Transcription: The Path and Control of Gene ExpressionDocumento24 páginasTranscription: The Path and Control of Gene ExpressionPaolo NaguitAinda não há avaliações
- Experiment 6: Determination of Alkali Content in Antacid Tablet Using HCLDocumento6 páginasExperiment 6: Determination of Alkali Content in Antacid Tablet Using HCLVivek SehgalAinda não há avaliações
- Worksheet Acid Base Titration CurveDocumento2 páginasWorksheet Acid Base Titration CurveMark MagoAinda não há avaliações
- A Colorimetric Assay For Detecting Haloalkane Dehalogenase ActivityDocumento6 páginasA Colorimetric Assay For Detecting Haloalkane Dehalogenase ActivityshuklagAinda não há avaliações
- Class 12th Chemistry Project On Investigatory Test On GuavaDocumento21 páginasClass 12th Chemistry Project On Investigatory Test On GuavaSulogna Dutta0% (1)
- 0620 Chemistry: MARK SCHEME For The May/June 2011 Question Paper For The Guidance of TeachersDocumento7 páginas0620 Chemistry: MARK SCHEME For The May/June 2011 Question Paper For The Guidance of TeachersAYSHAMOHD88Ainda não há avaliações
- Preparation of Reagents SolutionsDocumento2 páginasPreparation of Reagents Solutionsmelissa acostaAinda não há avaliações
- Titrasi Asam Basa Dan Reaksi RedoksDocumento40 páginasTitrasi Asam Basa Dan Reaksi RedoksPutri RiduanAinda não há avaliações
- AP Chemistry Review Questions Cover Oxidation States, Lewis Structures, StoichiometryDocumento16 páginasAP Chemistry Review Questions Cover Oxidation States, Lewis Structures, StoichiometryGernanAinda não há avaliações
- Titrimetric Methods of AnalysisDocumento34 páginasTitrimetric Methods of Analysisgizachew100% (1)
- Iso 6557-2-1984 - (2018-08-02 - 07-54-35 Am) PDFDocumento8 páginasIso 6557-2-1984 - (2018-08-02 - 07-54-35 Am) PDFbüşraAinda não há avaliações
- Practical Chem (CHM 107)Documento53 páginasPractical Chem (CHM 107)nuruddeenabdulhakeem070Ainda não há avaliações
- Determine Carbonate and Bicarbonate Using Acid-Base TitrationDocumento4 páginasDetermine Carbonate and Bicarbonate Using Acid-Base TitrationRisfanali RajaAinda não há avaliações
- Common Laboratory OperationsDocumento20 páginasCommon Laboratory OperationsJose Barrera Galera0% (1)
- Vitamin LABDocumento3 páginasVitamin LABLeonardo WalcottAinda não há avaliações
- App Note 016 Standardization of Iodine Titrant For Ripper Titration of WinesDocumento2 páginasApp Note 016 Standardization of Iodine Titrant For Ripper Titration of WinesfonsynickAinda não há avaliações
- Statistics in Analytical Chemistry-Part 2: Instructor: Nguyen Thao TrangDocumento44 páginasStatistics in Analytical Chemistry-Part 2: Instructor: Nguyen Thao TrangLeo PisAinda não há avaliações
- Topic 2 Precipitation of Barium (II) Sulphate & 3 Determination of The Amount of Dissolved Oxygen in A Water Sample by IodometryDocumento4 páginasTopic 2 Precipitation of Barium (II) Sulphate & 3 Determination of The Amount of Dissolved Oxygen in A Water Sample by IodometryChuan Ning LeAinda não há avaliações
- D664 PDFDocumento7 páginasD664 PDFROHITAinda não há avaliações
- J. Electrochem. Soc. 1978 Butler 228 32Documento5 páginasJ. Electrochem. Soc. 1978 Butler 228 32法林Ainda não há avaliações
- Universiti Sains Malaysia: EBS 336/3 - Analytical ChemistryDocumento15 páginasUniversiti Sains Malaysia: EBS 336/3 - Analytical ChemistryKrystel Monica ManaloAinda não há avaliações
- LabDocumento4 páginasLabapi-2818620680% (1)
- Novel Approach To Job's Method: An Undergraduate ExperimentDocumento6 páginasNovel Approach To Job's Method: An Undergraduate ExperimentClaudia TocuyoAinda não há avaliações
- Potentiometric TitrationsDocumento16 páginasPotentiometric Titrationsshal2006Ainda não há avaliações
- AP Chemistry Unit 2 Chemical Reactions Problem Sets CDODocumento7 páginasAP Chemistry Unit 2 Chemical Reactions Problem Sets CDOcgp7c648srAinda não há avaliações
- Ascorbic Acid As A Standard For Iodometric TitrationsDocumento5 páginasAscorbic Acid As A Standard For Iodometric TitrationsniltonAinda não há avaliações
- Chapter 4 MethodologyDocumento18 páginasChapter 4 Methodologyit rewaAinda não há avaliações