Escolar Documentos
Profissional Documentos
Cultura Documentos
Geometría Molecular PDF
Enviado por
GeanellaTítulo original
Direitos autorais
Formatos disponíveis
Compartilhar este documento
Compartilhar ou incorporar documento
Você considera este documento útil?
Este conteúdo é inapropriado?
Denunciar este documentoDireitos autorais:
Formatos disponíveis
Geometría Molecular PDF
Enviado por
GeanellaDireitos autorais:
Formatos disponíveis
436
Chapter 10 Chemical Bonding II: Molecular Shapes, Valence Bond Theory, and Molecular Orbital Theory
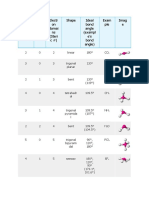
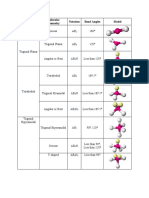
T A B L E 1 0 . 1 Electron and Molecular Geometries
Electron Bonding Lone Electron Molecular Approximate
Groups* Groups Pairs Geometry Geometry Bond Angles Example
2 2 0 Linear Linear 180° O C O
Trigonal F
3 3 0 Trigonal planar 120°
planar
F B F
3 2 1 Trigonal planar Bent <120° O S O
4 4 0 Tetrahedral Tetrahedral 109.5° H C H
H
Trigonal H N H
4 3 1 Tetrahedral <109.5°
pyramidal
H
4 2 2 Tetrahedral Bent <109.5° H O H
Cl
Cl
Trigonal Trigonal 120° (equatorial)
5 5 0
bipyramidal bipyramidal 90° (axial) P Cl
Cl
Cl
F
Trigonal <120° (equatorial)
5 4 1
bipyramidal
Seesaw
<90° (axial)
F S F
Trigonal F Br F
5 3 2 T-shaped <90°
bipyramidal
F
Trigonal
5 2 3 Linear 180° F Xe F
bipyramidal
F
F F
6 6 0 Octahedral Octahedral 90° S
F F
F
F
Square
6 5 1 Octahedral <90° F Br F
pyramidal
F F
F
Square
6 4 2 Octahedral
planar
90° F Xe F
*Count only electron groups around the central atom. Each of the following is considered one electron group: a lone pair, a single bond, a double bond, a triple bond,
or a single electron.
M10_TRO5187_04_SE_C10_426-483v4.0.2.indd 436 2015/11/20 7:15 PM
Você também pode gostar
- Mobil 1 Product GuideDocumento2 páginasMobil 1 Product GuideTudor RatiuAinda não há avaliações
- CPAA Cable Installation Manual-GJ + GL MAZDA 6Documento20 páginasCPAA Cable Installation Manual-GJ + GL MAZDA 6Viktor XAinda não há avaliações
- Configuratii ElectroniceDocumento1 páginaConfiguratii ElectroniceVasile FlaviusAinda não há avaliações
- Teorie Geometrie 5-8Documento27 páginasTeorie Geometrie 5-8Alina AlinaAinda não há avaliações
- Vsepr ChartDocumento2 páginasVsepr Chartapi-239855791Ainda não há avaliações
- 4.3-VSEPR - Shapes of MoleculesDocumento1 página4.3-VSEPR - Shapes of MoleculesStephan MinhAinda não há avaliações
- 3 AB Trigonal Planar Trigonal Planar 120 Between All BondsDocumento5 páginas3 AB Trigonal Planar Trigonal Planar 120 Between All BondsVedantAinda não há avaliações
- VSEPR TableDocumento1 páginaVSEPR TableAudrey HizonAinda não há avaliações
- Electron Groups Bonding Groups Lone Pairs Electronic Geometry Molecular Geometry Approximate Bond Angles ExampleDocumento2 páginasElectron Groups Bonding Groups Lone Pairs Electronic Geometry Molecular Geometry Approximate Bond Angles ExampleRichamille Ann RicaforteAinda não há avaliações
- Lewis Structures Molecular Geometry and Polarity 1A KEYDocumento3 páginasLewis Structures Molecular Geometry and Polarity 1A KEYrsleoAinda não há avaliações
- Bondi NG Electr On Pairs Lon e Pair S Electr On Domai Ns (Steri C#) Shape Ideal Bond Angle (Exampl E's Bond Angle) Exam Ple Imag eDocumento3 páginasBondi NG Electr On Pairs Lon e Pair S Electr On Domai Ns (Steri C#) Shape Ideal Bond Angle (Exampl E's Bond Angle) Exam Ple Imag eaadhyaAinda não há avaliações
- Chemistry-Molecular GeometryDocumento2 páginasChemistry-Molecular GeometryBubbles Bubbles100% (1)
- 2..chemical Bonding Theory-12-12Documento1 página2..chemical Bonding Theory-12-12Ashish SharmaAinda não há avaliações
- Molecular GeometryDocumento1 páginaMolecular GeometryIsraClarkeAinda não há avaliações
- Nota VSEPR PDFDocumento1 páginaNota VSEPR PDFMarlene GazconAinda não há avaliações
- KYOCERA CatalogueDocumento95 páginasKYOCERA CatalogueMANIT KUMAR BHOIAinda não há avaliações
- Electron Domains (Steric Number) Atoms Bonded To Central Atom Lone Pairs Shape Bond Angle Example ImageDocumento2 páginasElectron Domains (Steric Number) Atoms Bonded To Central Atom Lone Pairs Shape Bond Angle Example ImageBianca GuillermoAinda não há avaliações
- Shapes of Molecules & Ions: Name . . FormDocumento2 páginasShapes of Molecules & Ions: Name . . FormjnfjngsdjAinda não há avaliações
- Molecular ShapeDocumento1 páginaMolecular ShapeNUR DEENA KHALID KM-PensyarahAinda não há avaliações
- Iso DJ Milling InsertsDocumento21 páginasIso DJ Milling InsertsalphatoolsAinda não há avaliações
- θ α - alpha β - beta γ - gamma: theta Obtuse Angle Between 90 & 180Documento8 páginasθ α - alpha β - beta γ - gamma: theta Obtuse Angle Between 90 & 180charmaine padorAinda não há avaliações
- K 5Documento44 páginasK 5Ricardo Rincon Vega100% (1)
- Sa&Vol FormulaeDocumento1 páginaSa&Vol FormulaeComputer ioAinda não há avaliações
- Precision Mitre SawsDocumento4 páginasPrecision Mitre SawsTerra MachinesAinda não há avaliações
- Sample - Thickness Test Measurement For Steam DrumDocumento1 páginaSample - Thickness Test Measurement For Steam DrumAzim AsriAinda não há avaliações
- Iso Turning InsertsDocumento37 páginasIso Turning InsertsalphatoolsAinda não há avaliações
- 110022-1 Extraction Fan For VapourDocumento1 página110022-1 Extraction Fan For VapourpaulAinda não há avaliações
- Cast Iron Epoxy Hub Spigot 102221Documento15 páginasCast Iron Epoxy Hub Spigot 102221Anonymous wt2BA7uAinda não há avaliações
- LiTech DatasheetDocumento1 páginaLiTech DatasheetMadhankumarAinda não há avaliações
- Catalog of Milling Solution 2020Documento31 páginasCatalog of Milling Solution 2020vedrenne92Ainda não há avaliações
- Geometry Exercise - 1 (Redo) (Cie Cmabridge Mathematics Answer Guide)Documento11 páginasGeometry Exercise - 1 (Redo) (Cie Cmabridge Mathematics Answer Guide)Jenna HanyAinda não há avaliações
- Circular MeasureDocumento7 páginasCircular MeasureaffendeAinda não há avaliações
- Geometry - HW 31 Angles Formed by Secants and TangentsDocumento4 páginasGeometry - HW 31 Angles Formed by Secants and Tangentsroselyn panganibanAinda não há avaliações
- 8inchdrumd10 lh6Documento1 página8inchdrumd10 lh6mohammad khoraminiaAinda não há avaliações
- Part DrawingDocumento1 páginaPart DrawingAbhishek PatilAinda não há avaliações
- LiTech DatasheetDocumento1 páginaLiTech DatasheetMadhankumarAinda não há avaliações
- Tabelas Roscas TrapezoidaisDocumento49 páginasTabelas Roscas TrapezoidaisDesenvolvimento MHAinda não há avaliações
- Correlation 1 - Trigonometry NotesDocumento7 páginasCorrelation 1 - Trigonometry Notesdoni poAinda não há avaliações
- CF 10 Vbuurm KRV 22PPDocumento1 páginaCF 10 Vbuurm KRV 22PPHERNAN MESIASAinda não há avaliações
- 13C and 1H NMR (RMN 1H y 13C)Documento1 página13C and 1H NMR (RMN 1H y 13C)veromendoAinda não há avaliações
- Cazoom Maths. Lines and Angles. Angles On Parallel Lines (A) - AnswersDocumento2 páginasCazoom Maths. Lines and Angles. Angles On Parallel Lines (A) - AnswersChelsea ChikafuAinda não há avaliações
- LAS Physical-Science Week2Documento11 páginasLAS Physical-Science Week2Shekaina Faith Cuizon LozadaAinda não há avaliações
- Trapezgewinde Musterzeichnung PDFDocumento1 páginaTrapezgewinde Musterzeichnung PDFHMPGAinda não há avaliações
- EDLW DortmundDocumento17 páginasEDLW Dortmunddaniel.namendorfAinda não há avaliações
- Saegengewinde MusterzeichnungDocumento1 páginaSaegengewinde MusterzeichnunggeigerAinda não há avaliações
- Tip VDocumento1 páginaTip VKenan RamićAinda não há avaliações
- SumiSmall 2015-16 CATALOG LR PDFDocumento249 páginasSumiSmall 2015-16 CATALOG LR PDFAlbertAinda não há avaliações
- Dimensions: UnitsDocumento58 páginasDimensions: UnitsNaman MahawarAinda não há avaliações
- Square and RectangleDocumento5 páginasSquare and RectangleVishnu PrasadAinda não há avaliações
- ADESDocumento6 páginasADESjv680802Ainda não há avaliações
- Molecular GeometryDocumento1 páginaMolecular GeometryDean Joyce Alboroto100% (1)
- Ambiciclo Rampa Debaixo MMCDocumento1 páginaAmbiciclo Rampa Debaixo MMCAndréAmsAinda não há avaliações
- U-Section: Values For CalculationDocumento3 páginasU-Section: Values For CalculationEng-CalculationsAinda não há avaliações
- Tme R: Technical Data SheetDocumento5 páginasTme R: Technical Data SheetEnrique HortaAinda não há avaliações
- Insert Designation ChartDocumento10 páginasInsert Designation ChartEmba MadrasAinda não há avaliações
- 05 Geometric Details Hairpin Bends 05 Hairpin BendsDocumento1 página05 Geometric Details Hairpin Bends 05 Hairpin BendsBinod Raj GiriAinda não há avaliações
- Wallmount PDFDocumento1 páginaWallmount PDFAris RisnandarAinda não há avaliações
- Cazoom Maths. Lines and Angles. Angles On Parallel Lines (A)Documento2 páginasCazoom Maths. Lines and Angles. Angles On Parallel Lines (A)Apex GamingAinda não há avaliações
- ZYHBDocumento30 páginasZYHBGio GAinda não há avaliações
- PolygonDocumento46 páginasPolygonsheykrizzleAinda não há avaliações
- Third Quarter Examination 2017 in Science 6 (New) For DistrictDocumento6 páginasThird Quarter Examination 2017 in Science 6 (New) For DistrictMarlon Yap100% (2)
- Inductor and Flyback Transformer DesignDocumento20 páginasInductor and Flyback Transformer Designbmmostefa100% (1)
- Conduction Heat Transfer Arpaci.Documento551 páginasConduction Heat Transfer Arpaci.Abhimanyu Ghosh100% (4)
- Hess SmithDocumento26 páginasHess SmithhakimkaskusAinda não há avaliações
- JChemEduc 1983 60 112-116Documento5 páginasJChemEduc 1983 60 112-116Beto RodriguezAinda não há avaliações
- College of Engineering - Civil Engineering Department: Carig CampusDocumento6 páginasCollege of Engineering - Civil Engineering Department: Carig CampusShanaia Kate AsutenAinda não há avaliações
- Important MCQ - Magnetism and ElectromagnetismDocumento12 páginasImportant MCQ - Magnetism and ElectromagnetismNaman ChauhanAinda não há avaliações
- What Is Matrix EnergeticsDocumento3 páginasWhat Is Matrix EnergeticsJill Paxton100% (1)
- Fluid Mechanics PDFDocumento48 páginasFluid Mechanics PDFrakib hasanAinda não há avaliações
- Recent Progress Toward A Three-Dimensional Unstructured Navier-Stokes Flow SolverDocumento21 páginasRecent Progress Toward A Three-Dimensional Unstructured Navier-Stokes Flow SolverDanielle HaysAinda não há avaliações
- Composite Materials and MechanicsDocumento2 páginasComposite Materials and MechanicsroscillaAinda não há avaliações
- 3 SuperjunctionDocumento5 páginas3 SuperjunctionharibadriAinda não há avaliações
- First Year Handout March 2021Documento112 páginasFirst Year Handout March 2021King MegaAinda não há avaliações
- Questions and Answers EIMDocumento7 páginasQuestions and Answers EIMMark MarasiganAinda não há avaliações
- EAMCET Engineering Information BrochureDocumento23 páginasEAMCET Engineering Information BrochureAnweshaBoseAinda não há avaliações
- Convection Heat TransferDocumento49 páginasConvection Heat TransferAli almansouriAinda não há avaliações
- Tetrad Formulation of The Einstein Field Equations: The Newman-Penrose EquationsDocumento16 páginasTetrad Formulation of The Einstein Field Equations: The Newman-Penrose EquationsRockBrentwoodAinda não há avaliações
- Forces SlopesDocumento6 páginasForces SlopesXolani MoffatAinda não há avaliações
- s10853 011 6219 8 - DensityDocumento7 páginass10853 011 6219 8 - DensityEduardo Fernandez SanchezAinda não há avaliações
- UNIT 8 ThermodynamicsDocumento9 páginasUNIT 8 ThermodynamicsHimadhar SaduAinda não há avaliações
- TransientDocumento12 páginasTransientLester MuscaAinda não há avaliações
- Tipler and Mosca Physics For Scientists and Engineers Solutions Manual Chapter 13Documento102 páginasTipler and Mosca Physics For Scientists and Engineers Solutions Manual Chapter 13Nate LinxAinda não há avaliações
- GTE Micro Project 4th SemDocumento6 páginasGTE Micro Project 4th SemNishikant Bhure100% (3)
- Em 18 Equilibrium of A ParticleDocumento2 páginasEm 18 Equilibrium of A ParticleFattihi EkhmalAinda não há avaliações
- Glory - Phsics 32.1 ReportDocumento33 páginasGlory - Phsics 32.1 ReportGabriel Rafael S. VirayAinda não há avaliações
- Chapter 12 Notes Gas Laws NotesDocumento2 páginasChapter 12 Notes Gas Laws NotesNancy Richardson RollinsAinda não há avaliações
- Chapter 2: Water: The Solvent For Biochemical ReactionsDocumento6 páginasChapter 2: Water: The Solvent For Biochemical ReactionsMariam EidAinda não há avaliações
- Equilibrium of Rigid Bodies Unit - IIDocumento36 páginasEquilibrium of Rigid Bodies Unit - IIArkadeep MukherjeeAinda não há avaliações
- Digital Power Clamp Meter HIOKI 3286Documento4 páginasDigital Power Clamp Meter HIOKI 3286industrialindiaAinda não há avaliações