Escolar Documentos
Profissional Documentos
Cultura Documentos
Chapter 2 Acid and Base
Enviado por
Kelsi Kyla PeraltaDescrição original:
Direitos autorais
Formatos disponíveis
Compartilhar este documento
Compartilhar ou incorporar documento
Você considera este documento útil?
Este conteúdo é inapropriado?
Denunciar este documentoDireitos autorais:
Formatos disponíveis
Chapter 2 Acid and Base
Enviado por
Kelsi Kyla PeraltaDireitos autorais:
Formatos disponíveis
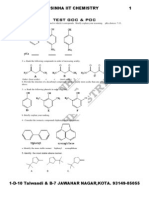
Chapter 2 - Polar Covalent Bonds; Acids and Bases
For questions 1-10 give the letter of the term that best matches the given definition.
a. Brønsted-Lowry Acid f. Ionic Bond
b. Brønsted-Lowry Base g. Covalent Bond
c. Lewis Acid h. Polar-Covalent Bond
d. Lewis Base i. Hydrophobic
e. Electronegativity j. Hydrophilic
1. Any species that can accept electrons.
Answer: c
2. A bond between two atoms differing in electronegativity by 0.5 – 2.
Answer: h
3. A term used to describe a “water loving” species.
Answer: j
4. A compound that can donate a proton.
Answer: a
5. The ability of an atom to attract the shared electrons in a covalent bond.
Answer: e
6. A term used to describe a “water fearing” species.
Answer: i
7. Any species that can donate electrons.
Answer: d
8. A bond between two atoms differing in electronegativity by < 0.5
Answer: g
9. A compound that can accept a proton.
Answer: b
10. A bond between two atoms differing in electronegativity by > 2.
Answer: f
4 Chapter 2: Polar Covalent Bonds; Acids and Bases
Calculate the formal charges on the indicated atoms in each compound below.
..
:O : 6. 7. 8.
.. ..
:Cl P Cl :
.. .. :C O:
: Cl : 5.
..
11. The formal charge on phosphorous is .
Answer: +1
12. The formal charge on oxygen is .
Answer: –1
13. The formal charge on carbon is .
Answer: –1
14. The formal charge on oxygen is .
Answer: +1
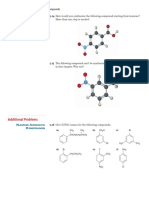
Phenylalanine is an amino acid that is essential to human nutrition. The representation below shows the
structure of phenylalanine at physiological pH. Consider this structure to answer the following questions.
A.
O
*
CH2CHC O phenylalanine
NH3 B.
15. Assign any formal charges to atoms in this representation of phenylalanine.
Answer:
O
CH2CHC O
NH3
16. The oxygen atom labeled A. has ______ non-bonding electrons.
Answer: four
17. The oxygen atom labeled B. has _____ bonding electrons.
Answer: two
Test Items for McMurry’s Organic Chemistry, Seventh Edition 5
Use the δ − /δ + convention and the crossed arrow ( ) to show the direction of the expected polarity of
the indicated bonds in the following compounds.
F
18. The C − F bond in fluorobenzene,
δ−
δ+ F
Answer:
19. The C-Si bond in tetramethylsilane, (CH3)4Si
CH3
δ+ δ−
Answer: H3C Si CH3
CH3
20. The C − O bond in furan,
O
Answer:
δ+
δ−O
Label the acid, conjugate acid, base, and conjugate base in each reaction below.
21. CH3OH + NaH CH3O- Na+ + H2
Answer:
CH3OH + NaH CH3O- Na+ + H2
acid base conjugate base conjugate acid
+ -
22. CH3OCH3 + BH3 H3C O BH3
CH3
Answer:
+ -
CH3OCH3 + BH3 H3C O BH3
base acid CH3
6 Chapter 2: Polar Covalent Bonds; Acids and Bases
Refer to the following equation to answer the questions below. Place the letter corresponding to the correct
answer in the blank.
pKa = 18 pKa = 15.7
(CH3)3C O H + K OH (CH3)3C O K + H2O
A. B. C. D.
23. The strongest Brønsted-Lowry acid in the equation is .
Answer: D
24. The strongest Brønsted-Lowry base in the equation is .
Answer: C
25. Will this reaction take place as written? Explain.
Answer: No, the reaction will not take place as written because the strongest acid reacts with the
strongest base to give the weakest conjugate acid and the weakest conjugate base. D (pKa =
15.7) is a stronger acid than A (pKa = 18).
26. An acid with a low pKa:
a. is a weak acid
b. is a strong acid
c. has a weak conjugate base
d. both b and c
Answer: d
27. Circle all the Lewis bases in the group of compounds below.
CH3 OH HCl BCl3 FeBr3
Answer:
O
CH3 OH HCl BCl3 FeBr3
Test Items for McMurry’s Organic Chemistry, Seventh Edition 7
28. Put a box around all the Lewis acids in the group of compounds below.
O
CH3 OH HCl BCl3 FeBr3
N
Answer:
CH3 OH HCl BCl3 FeBr3
29. Draw two resonance structures for the species below.
+ .. CH3
O
..
Answer:
+ CH3 + .. CH3
.. CH3 +
O O O
.. ..
30. Draw two resonance structures for the species below.
:O :
_CH
.. 2
Answer:
.. _
:O : :O : :O :
_CH
.. 2 ..
_ CH2 CH2
31. Draw two resonance structures for the species below.
_
..
O:
..
Answer:
_
..
.. _ .. _: ..
O : O: O: O
.. .. ..
_..
8 Chapter 2: Polar Covalent Bonds; Acids and Bases
Consider the acidity constants below to answer the following questions.
ACID STRUCTURE pKa
phenol OH 10.00
ethanol CH3CH2OH 16.00
water HOH 15.74
32. Which acid above will be almost completely deprotonated by NaOH?
Answer: phenol
33. Which acid has the strongest conjugate base?
Answer: Ethanol is the weakest acid (largest pKa) so its conjugate base, ethoxide, CH 3CH 2O − , will be
the strongest base.
34. Explain why phenol has a much lower pKa than ethanol.
Answer: Phenol is more acidic (has a lower pKa) than ethanol because the phenoxide anion is resonance
stabilized by the pi electrons in the ring.
Ethoxide anion has no resonance stabilization. The negative charge is borne fully by oxygen.
-H+ .. _
CH3CH2OH CH3CH2O :
..
_
..
.. -H+ .. _
OH
.. O : O :
.. ..
.. ..
O: _: O
..
_..
Test Items for McMurry’s Organic Chemistry, Seventh Edition 9
Consider the reaction below to answer the following questions.
O O
H
N .. _ _ N _
H N H + H O + O: +
H N H O + O
..
H H
35. Using the curved arrow formalism, show the flow of electrons for this reaction.
Answer:
O O
H
N .. _ _ N _
H N H + H O + O:
.. H +N H O + O
H H
36. Label the acid and the base in the reaction.
Answer:
O O
H
N .. _ _ N _
H N H + H O + O: +
H N H O + O
..
H H
base acid
Indole is pleasant smelling in highly dilute solutions and had been used in perfumery. Use the structure of
indole, below, to answer the following questions.
indole
N
H
37. Indole can function as a Bronsted-Lowry acid in the presence of strong bases. Formulate a reaction,
showing electron flow with arrows, that demonstrates this reactivity of indole.
Answer:
B + BH
N N
H
10 Chapter 2: Polar Covalent Bonds; Acids and Bases
38. Indole can function as a Lewis base in the presence of strong acid. Formulate a reaction, showing electron
flow with arrows, that demonstrates this reactivity of indole.
Answer:
H A + A
N N
H
H
H
39. The condensed structure for dimethyl ether looks symmetrical. However, dimethyl ether has a dipole
moment. Draw a structure that explains this and indicate the expected direction of the molecular dipole
moment.
CH3OCH3
dimethyl ether
Answer:
net dipole
H3C CH3
Consider the acid-base reaction below to answer the following questions.
CH3CH O + H O H
CH3
base acid
40. Using the curved arrow formalism, show the flow of electrons for this reaction.
Answer:
CH3CH O + H O H
CH3
base acid
41. Write the products of this Lewis acid - base reaction.
Answer:
CH3CH O + H O H CH3CH O H + O H
CH3 CH3
base acid
Test Items for McMurry’s Organic Chemistry, Seventh Edition 11
Você também pode gostar
- Handbook of Coordination Catalysis in Organic ChemistryNo EverandHandbook of Coordination Catalysis in Organic ChemistryAinda não há avaliações
- Organic Chemistry 32-235 Practice Questions For Exam #2: 2. Consider The SDocumento9 páginasOrganic Chemistry 32-235 Practice Questions For Exam #2: 2. Consider The Ssweta KushwahaAinda não há avaliações
- ICSE Chemistry Board Paper19 PDFDocumento9 páginasICSE Chemistry Board Paper19 PDFPrajakta DigheAinda não há avaliações
- Resonance and Inductive Effects PresentationDocumento36 páginasResonance and Inductive Effects Presentationeagl33yeAinda não há avaliações
- Module8 PDFDocumento40 páginasModule8 PDFFaizan AhmadAinda não há avaliações
- Alkenes SeatworkDocumento5 páginasAlkenes SeatworkJhefAinda não há avaliações
- Exercise With Ans FinalDocumento24 páginasExercise With Ans Finald anjilappa25% (4)
- Chemical Bonding - HybridisationDocumento3 páginasChemical Bonding - HybridisationVarsha YadavAinda não há avaliações
- Acid Base 15Documento36 páginasAcid Base 15Imranzo HsnAinda não há avaliações
- Inorganic Chemistry II (100 Items)Documento11 páginasInorganic Chemistry II (100 Items)maria jeusa matiasAinda não há avaliações
- Chapter 9 Alkynes: Answers Prof. Sivaguru JayaramanDocumento11 páginasChapter 9 Alkynes: Answers Prof. Sivaguru JayaramanRahma AshrafAinda não há avaliações
- Midterm II Key Chem 2312-003 F '12Documento7 páginasMidterm II Key Chem 2312-003 F '12acb4039Ainda não há avaliações
- All Year Chemistry Up To 2018 PDFDocumento37 páginasAll Year Chemistry Up To 2018 PDFAGAH LUCKYAinda não há avaliações
- Chapter 2 Lecture Notes - 0Documento44 páginasChapter 2 Lecture Notes - 0KirilKocevskiAinda não há avaliações
- Chapter 9 PDFDocumento12 páginasChapter 9 PDFKelsi Kyla Peralta0% (1)
- 1 Alkene Practice Problems MOC PDFDocumento8 páginas1 Alkene Practice Problems MOC PDFsamkarthik47Ainda não há avaliações
- 314 Stereochem ProbsDocumento14 páginas314 Stereochem ProbsAtul SinghAinda não há avaliações
- Aromaticity TutorialDocumento15 páginasAromaticity TutorialAlex-Mihai Ciubara100% (2)
- Organic Chemistry 2 Practice Exam 1Documento15 páginasOrganic Chemistry 2 Practice Exam 1KaybidoAinda não há avaliações
- Test1 Goc & Poc Tough by S.K.sinha See Chemistry Animations atDocumento3 páginasTest1 Goc & Poc Tough by S.K.sinha See Chemistry Animations atmyiitchemistry100% (1)
- Spectroscopy and ChromatographyDocumento7 páginasSpectroscopy and ChromatographyPa GesAinda não há avaliações
- Alcohols & Phenols:: GeneralizationsDocumento27 páginasAlcohols & Phenols:: GeneralizationsdoudoudoudouAinda não há avaliações
- Brown 5e Ch07Documento33 páginasBrown 5e Ch07Li LizAinda não há avaliações
- CHEM 331 Kraus Ihazlett 1 Chapter9Documento12 páginasCHEM 331 Kraus Ihazlett 1 Chapter9Ahmed SideegAinda não há avaliações
- Lecture 1Documento11 páginasLecture 1Fang GaoAinda não há avaliações
- Tutorial 4 - ElectrochemistryDocumento3 páginasTutorial 4 - ElectrochemistryAnis IssabellaAinda não há avaliações
- Spectroscopy WorksheetDocumento24 páginasSpectroscopy Worksheetpokemon goAinda não há avaliações
- Alkyl Halide-Jeemain - Guru PDFDocumento37 páginasAlkyl Halide-Jeemain - Guru PDFUma JadounAinda não há avaliações
- Chapter 8 Practice Test 4u1Documento1 páginaChapter 8 Practice Test 4u1helloblargAinda não há avaliações
- Chapter 12: Reactions of Arenes - Electrophilic Aromatic SubstitutionDocumento29 páginasChapter 12: Reactions of Arenes - Electrophilic Aromatic SubstitutionAnonymous Ngsu7C4aAinda não há avaliações
- Solid State-1Documento31 páginasSolid State-1ChirAgAinda não há avaliações
- Inorganic Chemistry Exam 20110331ansDocumento4 páginasInorganic Chemistry Exam 20110331ans曾鈞浩Ainda não há avaliações
- Organic Chemistry (Some Basic Principles and TechniquesDocumento30 páginasOrganic Chemistry (Some Basic Principles and TechniquesNaveen SharmaAinda não há avaliações
- Chapter 8 Nucleophilic Substitution: Answers Prof. Sivaguru JayaramanDocumento16 páginasChapter 8 Nucleophilic Substitution: Answers Prof. Sivaguru JayaramanRahma AshrafAinda não há avaliações
- Answer: (A) and (B)Documento18 páginasAnswer: (A) and (B)Germaine Manangan100% (1)
- IsomerismDocumento62 páginasIsomerismsubesinghAinda não há avaliações
- Organic Chemistry Test 1 MemorandumDocumento7 páginasOrganic Chemistry Test 1 MemorandumSandile SynthaxError Mabika0% (1)
- Organic Chemistry Problem SetDocumento10 páginasOrganic Chemistry Problem SetBermonica Alvior SatuitoAinda não há avaliações
- Vollhardt 6e Lecture PowerPoints - Chapter 11Documento58 páginasVollhardt 6e Lecture PowerPoints - Chapter 11superfr3shmAinda não há avaliações
- Reaction SummaryDocumento5 páginasReaction SummaryShafaqatRahmanAinda não há avaliações
- Test Bank For Organic Chemistry 3rd Edition Janice SmithDocumento15 páginasTest Bank For Organic Chemistry 3rd Edition Janice Smithjacobjasminekpk5Ainda não há avaliações
- The Problem Set of The Four Rounds: ProblemsDocumento29 páginasThe Problem Set of The Four Rounds: ProblemsMinh TieuAinda não há avaliações
- Chem1AA3 Lecture 1 PDFDocumento110 páginasChem1AA3 Lecture 1 PDFbhavanjeetAinda não há avaliações
- Nomenclature of Organic Compounds Chemistry QuestionsDocumento10 páginasNomenclature of Organic Compounds Chemistry QuestionsZeeshan AhmadAinda não há avaliações
- Chapter 10 PDFDocumento10 páginasChapter 10 PDFKelsi Kyla PeraltaAinda não há avaliações
- Acid-Base Practice Problems-Answers PDFDocumento5 páginasAcid-Base Practice Problems-Answers PDFSuci PrameswariAinda não há avaliações
- XI Chemistry Chapterwise Advanced Study MaterialDocumento537 páginasXI Chemistry Chapterwise Advanced Study MaterialregisAinda não há avaliações
- Qoi0809t1 ConfDocumento13 páginasQoi0809t1 ConfTahirat NasiruAinda não há avaliações
- Homework 3Documento13 páginasHomework 3polypeptideAinda não há avaliações
- APEF Electrochem MC Ans PDFDocumento2 páginasAPEF Electrochem MC Ans PDFMuhammad UsmanAinda não há avaliações
- Intermolecular Forces - Chemistry PracticeDocumento1 páginaIntermolecular Forces - Chemistry Practicewjahx8eloo ly100% (1)
- Acid Base Problems SolutionsDocumento20 páginasAcid Base Problems SolutionsAnusha PatelAinda não há avaliações
- Ch1 2 3 ExercisesDocumento11 páginasCh1 2 3 ExercisesMancini100% (1)
- Part 1: Boyle's Law: Pressure-vs-Volume: MaterialsDocumento5 páginasPart 1: Boyle's Law: Pressure-vs-Volume: MaterialsMadison IngramAinda não há avaliações
- Organic ChemistryDocumento20 páginasOrganic ChemistryGirish RaguvirAinda não há avaliações
- Practice Test 1 CHM2210C CH 1-3 PDFDocumento13 páginasPractice Test 1 CHM2210C CH 1-3 PDFkara lane100% (1)
- Enzymes: A Practical Introduction to Structure, Mechanism, and Data AnalysisNo EverandEnzymes: A Practical Introduction to Structure, Mechanism, and Data AnalysisNota: 4 de 5 estrelas4/5 (2)
- Worktext 1 - Biomol - Carbo & Lipids, PERALTADocumento10 páginasWorktext 1 - Biomol - Carbo & Lipids, PERALTAKelsi Kyla PeraltaAinda não há avaliações
- Worktext 1 - Biomol - Carbo & Lipids, PERALTADocumento10 páginasWorktext 1 - Biomol - Carbo & Lipids, PERALTAKelsi Kyla PeraltaAinda não há avaliações
- Prepared By: Group 2 Venus Prepared By: Group 2 Venus: Earth EarthDocumento2 páginasPrepared By: Group 2 Venus Prepared By: Group 2 Venus: Earth EarthKelsi Kyla PeraltaAinda não há avaliações
- Comets Comets: Comet vs. Asteroid Rock Metal Comet vs. Asteroid Rock MetalDocumento2 páginasComets Comets: Comet vs. Asteroid Rock Metal Comet vs. Asteroid Rock MetalKelsi Kyla PeraltaAinda não há avaliações
- Chapter 9 PDFDocumento12 páginasChapter 9 PDFKelsi Kyla Peralta0% (1)
- Çhapter 3Documento6 páginasÇhapter 3Kelsi Kyla PeraltaAinda não há avaliações
- Chapter 10 PDFDocumento10 páginasChapter 10 PDFKelsi Kyla PeraltaAinda não há avaliações
- Çhapter 3Documento6 páginasÇhapter 3Kelsi Kyla PeraltaAinda não há avaliações
- CEPF640/CEBF640 CEFF640: N-Channel Enhancement Mode Field Effect Transistor FeaturesDocumento4 páginasCEPF640/CEBF640 CEFF640: N-Channel Enhancement Mode Field Effect Transistor FeaturesAngel FaneitezAinda não há avaliações
- B-701 Boysen Permacoat Flat Latex2Documento7 páginasB-701 Boysen Permacoat Flat Latex2ircvpandoAinda não há avaliações
- Transient Stability of A Multi Machine Power System: Devender Kumar, Balwinder Singh SurjanDocumento4 páginasTransient Stability of A Multi Machine Power System: Devender Kumar, Balwinder Singh SurjanVerruumm AmineAinda não há avaliações
- Role of Packaging in Sales of FMCG Products and Its TrendsDocumento57 páginasRole of Packaging in Sales of FMCG Products and Its TrendsSaurabh0% (1)
- Desert Magazine 1957 DecemberDocumento44 páginasDesert Magazine 1957 Decemberdm1937Ainda não há avaliações
- Gem WK6/WK8 Service ManualDocumento18 páginasGem WK6/WK8 Service Manualalex_seidiu100% (5)
- Measuring Salinity in Crude Oils Evaluation of MetDocumento9 páginasMeasuring Salinity in Crude Oils Evaluation of Metarmando fuentesAinda não há avaliações
- Antenatally Diagnosed Kidney AnomaliesDocumento17 páginasAntenatally Diagnosed Kidney AnomalieslauraAinda não há avaliações
- Kids and Their Favourite Food Key Reading Comprehension Exercises - 34436Documento2 páginasKids and Their Favourite Food Key Reading Comprehension Exercises - 34436MonicaMartirosyanAinda não há avaliações
- Optimizing With eGaN FETsDocumento6 páginasOptimizing With eGaN FETskhsniperAinda não há avaliações
- First Periodical Exam Math 8Documento2 páginasFirst Periodical Exam Math 8Joanne88% (8)
- CP Lithium Ion BatteriesDocumento4 páginasCP Lithium Ion BatteriesvaseemalikhanAinda não há avaliações
- Worksheet 3 (Partial Pressures)Documento2 páginasWorksheet 3 (Partial Pressures)Jose Ruben SortoAinda não há avaliações
- DP16B Bench Drill PressDocumento20 páginasDP16B Bench Drill Pressalfri7370% (1)
- Liquid SizingDocumento38 páginasLiquid SizingChetan ChuriAinda não há avaliações
- Honda XL700VA9 Parts Catalogue Final PDFDocumento78 páginasHonda XL700VA9 Parts Catalogue Final PDFangeloAinda não há avaliações
- PTFE HZ-2 HoerbigerDocumento2 páginasPTFE HZ-2 HoerbigerТатьяна ВасильеваAinda não há avaliações
- Crimin Q and A Set 1Documento6 páginasCrimin Q and A Set 1Marc angelo RegnerAinda não há avaliações
- Carpentry 7&8 Quarter 4-Module 1.2Documento8 páginasCarpentry 7&8 Quarter 4-Module 1.2Mark Laurence EchaluceAinda não há avaliações
- Factorisation PDFDocumento3 páginasFactorisation PDFRaj Kumar0% (1)
- 1 N117Geriatric Nursing Lecture 1 BABY BOOMERSDocumento4 páginas1 N117Geriatric Nursing Lecture 1 BABY BOOMERSqygwfvbysgbcnntgrvbAinda não há avaliações
- BDSM List FixedDocumento4 páginasBDSM List Fixedchamarion100% (3)
- Troublesshooting Manual 3b6 PDFDocumento50 páginasTroublesshooting Manual 3b6 PDFAgnieszka Wawrzyniak Rybka100% (1)
- Jason Read, "Real Subsumption"Documento32 páginasJason Read, "Real Subsumption"Aren Z. AizuraAinda não há avaliações
- Us 5596162Documento9 páginasUs 5596162Stanley PinesAinda não há avaliações
- Method Statement of T-Beams PDFDocumento14 páginasMethod Statement of T-Beams PDFKAmi KaMranAinda não há avaliações
- Anaerobic Degradation of Palm Oil Mill Ef Uent (POME)Documento8 páginasAnaerobic Degradation of Palm Oil Mill Ef Uent (POME)HusainiAinda não há avaliações
- Gulika in Vedic AstrologyDocumento8 páginasGulika in Vedic AstrologyCatchdgreen100% (1)
- Advanced Automatic ControlDocumento26 páginasAdvanced Automatic Controlabdullah 3mar abou reashaAinda não há avaliações
- Ded Deliverable List: As Per 19-08-2016Documento2 páginasDed Deliverable List: As Per 19-08-2016Isna MuthoharohAinda não há avaliações