Escolar Documentos
Profissional Documentos
Cultura Documentos
Acid Dew Point - Encyclopedia Article - Citizendium
Enviado por
insult2injuryDescrição original:
Direitos autorais
Formatos disponíveis
Compartilhar este documento
Compartilhar ou incorporar documento
Você considera este documento útil?
Este conteúdo é inapropriado?
Denunciar este documentoDireitos autorais:
Formatos disponíveis
Acid Dew Point - Encyclopedia Article - Citizendium
Enviado por
insult2injuryDireitos autorais:
Formatos disponíveis
Acid dew point - encyclopedia article - Citizendium
http://en.citizendium.org/wiki/Acid_dew_point
From Citizendium, the Citizens' Compendium
This article is about Acid dew point. For other uses of the term Dew point, please see Dew point (disambiguation). The acid dew point (also acid dewpoint) of a flue gas (i.e., a combustion product gas) is the temperature, at a given pressure, at which any gaseous acid in the flue gas will start to condense into liquid acid.[1][2][3] The acid dew point of a flue gas, at a given pressure, is often referred to as the point at which the flue gas is "saturated" with gaseous acid, meaning that the flue gas cannot hold any more gaseous acid.
In many industrial combustion processes, the flue gas is cooled by the recovery of heat from the hot flue gases before they are emitted to the atmosphere from the final flue gas stack (commonly referred to as a chimney). It is very important not to cool the flue gas below its acid dew point because the resulting liquid acid condensed from the flue gas can cause serious corrosion problems for the equipment used in transporting, cooling and emitting the flue gas.
1 Chemistry and mechanism 1.1 Sulfuric acid dew point 1.2 Other acid dew points 1.2.1 Sulfurous acid 1.2.2 Nitric acid 1.2.3 Hydrochloric acid 2 Prediction of acid dew points 2.1 Predicting the sulfur trioxide content of flue gases 3 References
Sulfuric acid dew point
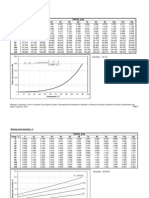
As a broad generality, flue gases from the combustion of coal, fuel oil, natural gas, or biomass are primarily composed of gaseous carbon dioxide (CO2) and water vapor (H2O) as well as gaseous nitrogen (N2) and excess oxygen (O2) remaining from the intake combustion air. Typically, more than two-thirds of the flue gas is nitrogen. The combustion flue gases may also contain small amounts of particulate matter, carbon monoxide, nitrogen oxides, and sulfur oxides in the form of gaseous sulfur dioxide (SO2) and gaseous sulfur trioxide (SO3). The SO3 is present because a portion of the SO2 formed in the combustion of the sulfur (S) compounds in the combustion fuel is further oxidized to SO3. The gas phase SO3 then combines the vapor phase H2O to form gas phase sulfuric acid H2SO4: H2O + SO3 H2SO4 water + sulfur trioxide sulfuric acid Because of the presence of gaseous sulfuric acid, the sulfuric acid dew point of most flue gases is much higher than the water dew point of the flue gases. For example, a flue gas with 12 volume % water vapor and containing no acid gases has a water dew point of about 49.4 C (121 F). The same flue gas with the addition of only 4 ppmv (0.0004 volume %) of SO3 will have a sulfuric acid dew point of about 130.5 C (267 F). The acid dew point of a combustion flue gas depends upon the composition of the specific fuel being burned and the resultant composition of the flue gas. The adjacent graph depicts how the amounts of water vapor and gaseous SO3 present in a flue gas affect the sulfuric acid dew point of the flue gas.
1 of 4
11/15/2011 10:56 AM
Acid dew point - encyclopedia article - Citizendium
http://en.citizendium.org/wiki/Acid_dew_point
Given a flue gas composition, its acid dew point can be predicted fairly closely. As an approximation, the sulfuric acid dew points of flue gases from the combustion of fuels in thermal power plants range from about 120 C to about 150 C (250 to 300 F).
Other acid dew points
Sulfurous acid Some of the sulfur dioxide in flue gases will also combine with water vapor in the flue gases and form gas phase sulfurous acid (H2SO3): H2O + SO2 H2SO3 water + sulfur dioxide sulfurous acid Nitric acid
(PD) Graph: Milton Beychok
Calculated sulfuric acid dew points of typical combustion flue gases, as a function of SO3 content, and water vapor content [4]
The nitrogen in flues gases is derived from the combustion air as well as from nitrogen compounds contained in the combustion fuel. Some small amount of the nitrogen is oxidized into gaseous nitrogen dioxide (NO2) and some of that gas phase nitrogen oxide then combines with water vapor to form gas phase nitric acid (HNO3): H2O + NO2 H2NO3 water + nitrogen dioxide nitric acid Hydrochloric acid Some flue gases may also contain gaseous hydrochloric acid (HCl) derived from chloride compounds in the combustion fuel. For example, municipal solid wastes contain chloride compounds and therefore the flue gases from municipal solid waste incinerators may contain gaseous hydrochloric acid which will condense into liquid hydrochloric acid if those flue gases are cooled to a temperature below the acid dew point of hydrochloric acid.
These equations can be used to predict the acid dew points of the four acids that most commonly occur in typical combustion product flue gases: Sulfuric acid (H2SO4) dew point:[5][6] (1) or this equivalent form:[2][4][7] (2) Sulfurous acid (H2SO3) dew point:[2][7][8] (3)
2 of 4
11/15/2011 10:56 AM
Acid dew point - encyclopedia article - Citizendium
http://en.citizendium.org/wiki/Acid_dew_point
Hydrochloric acid (HCl) dew point:[2][7][8] (4) Nitric acid (HNO3) dew point:[7][8] (5) where: = The acid dew point temperature for the indicated acid, (K) = Partial pressure, (atm for equation 1 and mmHg for equations 2, 3, 4 and 5) Compared with published measured data, the acid dew points predicted with equations 3, 4 and 5 are said to be within 6 kelvins, and within 9 kelvins for equations 1 and 2.[2]
Predicting the sulfur trioxide content of flue gases
As can be seen in the above equation for the sulfuric acid dew point of a flue gas, the partial pressure of sulfur trioxide in the flue gas is required. That partial pressure can be readily determined given the total pressure of the flue gas and the volume percent of sulfur trioxide in the flue gas, since the partial pressure of any component of a gaseous mixture may be obtained by simply multiplying the total gas pressure by the component's volume fraction of the gaseous mixture. Determining the volume percent of sulfur trioxide in a flue gas by theoretical calculations is quite difficult and unreliable. However, the volume fraction of the sulfur dioxide in the flue gas can be determined by assuming that 90 percent or more of the sulfur in the combustion fuel will be oxidized into gaseous sulfur dioxide when the fuel is combusted. Then it is commonly assumed that about 1 to 5 percent of the sulfur dioxide will be further oxidized into sulfur trioxide. In other words, if the sulfur dioxide in the flue gas is determined to be 0.3 volume percent and it is assumed that 3 percent of that will be further oxidized to sulfur trioxide, the volume fraction of sulfur trioxide in the flue gas will be (0.003)(0.03) = 0.00009 and, if the flue gas pressure is essentially 1 atm (760 mmHg), the partial pressure of the sulfur trioxide will be (0.00009)(760) = 0.0684 mmHg.
1. David A. Lewandowski (2000). Design of Thermal Oxidation Systems for Volatile Organic Compounds, 1st Edition. CRC Press. ISBN 1-56670-410-3. Available here (http://books.google.com/books?id=L-lKUWd-QOwC&printsec=frontcover& dq=intitle:Design+intitle:of+intitle:Thermal+intitle:Oxidation+intitle:Systems+intitle:for+intitle:V olatile+intitle:Organic+intitle:Compounds& hl=en&ei=wNOTTNWtEISdlgftvKmoCg&sa=X&oi=book_result&ct=result&resnum=1&ved=0CDMQ6AEwAA#v=onepage&q&f=false) in Google Books. 2. 2.0 2.1 2.2 2.3 2.4 John J. McKetta (Editor) (1997). Encyclopedia of Chemical Processing and Design, Volume 61, 1st Edition. CRC Press. ISBN 0-8247-2612-X. Available here (http://books.google.com/books?id=RNXyYP8EA2EC&printsec=frontcover&dq=0-8247-2612x&hl=en&ei=z9GTTITsJoOglAect_2qCg&sa=X&oi=book_result&ct=result&resnum=1&ved=0CDUQ6AEwAA#v=onepage&q&f=false) in Google books. 3. W.M.M. Huijbregts and R. Leferink (2004). "Latest Advances in the Understanding of Acid Dewpoint Corrosion: Corrosion and Stress Corrosion Cracking in Combustion Gas Condensates" (http://www.hbscc.nl/pdf/56%20ACMM%20Condensate%20%20SCC.pdf) . Anti-Corrosion Methods and Materials 51 (3): 173 - 188. 4. 4.0 4.1 Condensing Economizer Article (http://www.condexenergy.com/Condensing_Economizer_Article.pdf) 5. F.H. Verhoff and J.T. Banchero (1974). "Predicting Dew Points of Gases". Chemical Engineering progress 78 (8): 71 - 72. 6. R.R. Pierce (1977). "Estimating Acid Dewpoints in Stack Gases". Chemical Engineering 84 (8): 125 - 128. 7. 7.0 7.1 7.2 7.3 V Ganapathy (1993). Steam Plant Calculations Manual, 2nd Edition. CRC Press. ISBN 0-8247-9147-9. See Table 2.9 on . page 94. Available here (http://books.google.com/books?id=b_gTbFeHuiEC&printsec=frontcover& dq=intitle:Steam+intitle:plant+intitle:calculations+intitle:manual+inauthor:Ganapathy&hl=en&ei=ktKTTKuzIMLflge6h5mnCg& sa=X&oi=book_result&ct=result&resnum=1&ved=0CDMQ6AEwAA#v=onepage&q&f=false) in Google Books. 8. 8.0 8.1 8.2 Yen Hsiung Kiang (1981). "Predicting Dewpoints of Gases". Chemical Engineering 88 (3): 127.
3 of 4
11/15/2011 10:56 AM
Acid dew point - encyclopedia article - Citizendium
http://en.citizendium.org/wiki/Acid_dew_point
Retrieved from "http://en.citizendium.org/wiki/Acid_dew_point" Categories: CZ Live | Engineering Workgroup | Chemistry Workgroup | Chemical Engineering Subgroup | All Content | Engineering Content | Chemistry Content | Chemical Engineering tag
Views
Page Discussion View source History
Personal tools
Log in
Search
This page was last modified 00:37, 4 December 2010. CZ is free. All original articles are available under the Creative Commons-Attribution-ShareAlike 3.0 Unported license or any later. Articles that originated in part from Wikipedia are also available under GNU Free Documentation License 1.2. Servers and hosting generously provided by Steadfast Networks
4 of 4
11/15/2011 10:56 AM
Você também pode gostar
- Live Mechanical Labour BookDocumento68 páginasLive Mechanical Labour Bookinsult2injuryAinda não há avaliações
- Crime DetectionDocumento7 páginasCrime DetectionGlorious El Domine71% (7)
- Start Up AirDocumento2 páginasStart Up AirIsaac FloresAinda não há avaliações
- Iso 6976Documento2 páginasIso 6976Gia BaoAinda não há avaliações
- Nigeria Gas Specification - GACNDocumento1 páginaNigeria Gas Specification - GACNOlusegun OyebanjiAinda não há avaliações
- gt24 XL Gas Turbine Upgrade PDFDocumento4 páginasgt24 XL Gas Turbine Upgrade PDFkmishra80Ainda não há avaliações
- Nooter Eriksen HRSG AdvantagesDocumento3 páginasNooter Eriksen HRSG Advantagesaliscribd46Ainda não há avaliações
- An Investigation of Air-Swirl Design Criteria For Gas Turbine Combustors Through A Multi-Objective CFD OptimizationDocumento16 páginasAn Investigation of Air-Swirl Design Criteria For Gas Turbine Combustors Through A Multi-Objective CFD Optimizationperules100% (1)
- IBERDROLA GÜIRIA - SGT6-5000F(4) GENERIC CORRECTION CURVESDocumento12 páginasIBERDROLA GÜIRIA - SGT6-5000F(4) GENERIC CORRECTION CURVESENMANUELAinda não há avaliações
- 2012 Coleman Anaerobic Digestion SSO Vs WWTPDocumento16 páginas2012 Coleman Anaerobic Digestion SSO Vs WWTPcolemanpf_39910546Ainda não há avaliações
- CO2 CompressorDocumento23 páginasCO2 CompressorGaurav MishraAinda não há avaliações
- Jumag Gesamtkatalog enDocumento24 páginasJumag Gesamtkatalog enatisz333Ainda não há avaliações
- SiemensEnergy IndustrialHeatPumpsDocumento25 páginasSiemensEnergy IndustrialHeatPumpsHélder FernandoAinda não há avaliações
- Estimation Man HourDocumento241 páginasEstimation Man HourAnonymous ynJByUs52% (42)
- Predicting Dewpoints of Acid GasesDocumento6 páginasPredicting Dewpoints of Acid GasesShreyasGadkariAinda não há avaliações
- V005t15a001 88 GT 150Documento13 páginasV005t15a001 88 GT 150Antonio CilindroAinda não há avaliações
- Humidified Gas Turbines, by EderDocumento66 páginasHumidified Gas Turbines, by EderDomingo PintoAinda não há avaliações
- 20.06 - GEK110856j Steam Seal System RequirementsDocumento24 páginas20.06 - GEK110856j Steam Seal System RequirementsLIU100% (2)
- Manual Mando A DistanciaDocumento13 páginasManual Mando A DistanciaManuel SieŕraAinda não há avaliações
- PH Measurement in Tail Gas Clean-Up: Petroleum Refining IndustryDocumento2 páginasPH Measurement in Tail Gas Clean-Up: Petroleum Refining IndustryachillesntroyAinda não há avaliações
- Operating and maintenance norms for hydraulic cylindersDocumento4 páginasOperating and maintenance norms for hydraulic cylinderszsmithAinda não há avaliações
- Basic Design Principles For Gas Turbine CombustorDocumento12 páginasBasic Design Principles For Gas Turbine CombustorLeela PrasadAinda não há avaliações
- Dynamics and Stability of Lean-Premixed Swirl-Stabilized CombustionDocumento72 páginasDynamics and Stability of Lean-Premixed Swirl-Stabilized CombustionamitavjhaAinda não há avaliações
- Gas Turbine in Cairo North Power StationDocumento38 páginasGas Turbine in Cairo North Power StationAbdul Moeed Kalson0% (1)
- Asme GT2005-68799 PDFDocumento9 páginasAsme GT2005-68799 PDFJeeEianYannAinda não há avaliações
- Lefebvre 1995Documento38 páginasLefebvre 1995stepanovatvAinda não há avaliações
- Acceleration Head for Centrifugal PumpsDocumento5 páginasAcceleration Head for Centrifugal PumpsEng AlfAinda não há avaliações
- HRSG Design Tutorial - Heat BalanceDocumento3 páginasHRSG Design Tutorial - Heat BalanceRavi KasaudhanAinda não há avaliações
- Low Voltage Motor Price ListDocumento4 páginasLow Voltage Motor Price ListPARBATIAinda não há avaliações
- 6 Types of Hydrogen ElectrolyzersDocumento26 páginas6 Types of Hydrogen ElectrolyzersVishnuVarthan ImayavarambanAinda não há avaliações
- Pulsation Damper Sizing RevDocumento13 páginasPulsation Damper Sizing RevFrancesca CoattiAinda não há avaliações
- 1 s2.0 S1226086X14001221 MainDocumento6 páginas1 s2.0 S1226086X14001221 MainJorge Rodriguez HerreraAinda não há avaliações
- Lecture 28 Modeling of GTDocumento59 páginasLecture 28 Modeling of GTCindy CarvalhoAinda não há avaliações
- ISO 14532 - 2014 (En), Natural Gas - Vocabulary PDFDocumento1 páginaISO 14532 - 2014 (En), Natural Gas - Vocabulary PDFDAYAMOY APLAinda não há avaliações
- RG Integrally Geared Compressors PDFDocumento6 páginasRG Integrally Geared Compressors PDFJaswindersingh BhatiaAinda não há avaliações
- WINSEM2021-22 CHE2006 TH VL2021220501413 Reference Material I 11-03-2022 Module-5 CombustionDocumento63 páginasWINSEM2021-22 CHE2006 TH VL2021220501413 Reference Material I 11-03-2022 Module-5 Combustionswastik vijayAinda não há avaliações
- Thermo-Economic Analysis of A Heat Recovery Steam Generator Combined CycleDocumento6 páginasThermo-Economic Analysis of A Heat Recovery Steam Generator Combined CyclekouroshAinda não há avaliações
- Choked Gas Flow - Milton BeychokDocumento8 páginasChoked Gas Flow - Milton BeychokSteve WanAinda não há avaliações
- 1989 Aker Predicting Gas Turbine Performance Degradation Due To Compressor Fouling Using Computer Simulation TechniquesDocumento8 páginas1989 Aker Predicting Gas Turbine Performance Degradation Due To Compressor Fouling Using Computer Simulation TechniquesArgenomSaubiAinda não há avaliações
- Long Weld Necks 600Documento1 páginaLong Weld Necks 600Nice OrioleAinda não há avaliações
- DETC2009-86080: The Use of Interference Diagrams To Avoid Impeller Resonance: An Application To Igv DesignDocumento8 páginasDETC2009-86080: The Use of Interference Diagrams To Avoid Impeller Resonance: An Application To Igv DesignRajesh KachrooAinda não há avaliações
- LMF-GP LX15-8 - 10 - 13Documento2 páginasLMF-GP LX15-8 - 10 - 13MAZEN0% (1)
- Constant Settable Droop Design Standard of GEDocumento4 páginasConstant Settable Droop Design Standard of GEPraveen PeethambaranAinda não há avaliações
- Gas Turbine ControlDocumento9 páginasGas Turbine ControlHBNBILAinda não há avaliações
- Peppers Catalogue PDFDocumento64 páginasPeppers Catalogue PDFStone123456789Ainda não há avaliações
- Diesel Engine For 1990sDocumento165 páginasDiesel Engine For 1990schandru_82Ainda não há avaliações
- Kurzke Turbine Map ExtensionpdfDocumento14 páginasKurzke Turbine Map ExtensionpdfzzzAinda não há avaliações
- Books: Cryogenic Engineering, 2 Edition Distillation Theory and Its Application To Design of Separation UnitsDocumento1 páginaBooks: Cryogenic Engineering, 2 Edition Distillation Theory and Its Application To Design of Separation UnitsRaul tejadaAinda não há avaliações
- Centrifugal CompressorDocumento14 páginasCentrifugal CompressorridanormaAinda não há avaliações
- HP Steam Turbine StageDocumento8 páginasHP Steam Turbine StageTarun ChoudharyAinda não há avaliações
- Non-Dimensional Aerodynamic Design of A Centrifugal Compressor ImpellerDocumento12 páginasNon-Dimensional Aerodynamic Design of A Centrifugal Compressor ImpellerSubhash PadmanabhanAinda não há avaliações
- Harbin Electric InternationalDocumento14 páginasHarbin Electric Internationalawais khanAinda não há avaliações
- Ariel Calculation MethodDocumento6 páginasAriel Calculation MethodSubrata Mukherjee100% (1)
- TB KeyPerformanceEvaluation PDFDocumento2 páginasTB KeyPerformanceEvaluation PDFvcharles100% (1)
- Gas Turbine CombustionDocumento9 páginasGas Turbine Combustionvarun kumarAinda não há avaliações
- Norsok Standard I-106Documento6 páginasNorsok Standard I-106Boureghda FayçalAinda não há avaliações
- Wong Sandler (1992)Documento10 páginasWong Sandler (1992)Anonymous PO7VwbBnAinda não há avaliações
- ATB Calculation - COB09-0320Documento10 páginasATB Calculation - COB09-0320blueyes78Ainda não há avaliações
- Site Performance Review - Gas TurbineDocumento10 páginasSite Performance Review - Gas TurbinemishraenggAinda não há avaliações
- The Effects of Two Shaft Gas Turbine Operating Conditions On The Overall PerformanceDocumento16 páginasThe Effects of Two Shaft Gas Turbine Operating Conditions On The Overall PerformanceAhmad Mohammad Abdul-Aziz100% (1)
- International Thermodynamic Tables of the Fluid State: Propylene (Propene)No EverandInternational Thermodynamic Tables of the Fluid State: Propylene (Propene)Ainda não há avaliações
- DP TableDocumento8 páginasDP TablejnmanivannanmechAinda não há avaliações
- HBE Anti FlashDocumento3 páginasHBE Anti Flashinsult2injuryAinda não há avaliações
- Adiabatic Water Cooling TowerDocumento4 páginasAdiabatic Water Cooling Towerinsult2injuryAinda não há avaliações
- English Download FanProtectionDocumento14 páginasEnglish Download FanProtectionSubhankar UncertainityAinda não há avaliações
- NAECI 2016 2018 v2Documento142 páginasNAECI 2016 2018 v2insult2injuryAinda não há avaliações
- Cost AnalysisDocumento2 páginasCost Analysisinsult2injuryAinda não há avaliações
- Facilities Dynamics Engineering-ProposalDocumento3 páginasFacilities Dynamics Engineering-Proposalerkamlakar2234Ainda não há avaliações
- EconomicsDocumento16 páginasEconomicsinsult2injuryAinda não há avaliações
- Estimating With Microsoft ExcelDocumento9 páginasEstimating With Microsoft ExcelBert EngAinda não há avaliações
- LSI & Max Cycles Calculator: Tower LSI Based On Calc PH 2.50Documento2 páginasLSI & Max Cycles Calculator: Tower LSI Based On Calc PH 2.50thanh_79Ainda não há avaliações
- Seawater Property TablesDocumento17 páginasSeawater Property TablesdiespinoAinda não há avaliações
- F-12102 Rules of CalculationDocumento9 páginasF-12102 Rules of Calculationinsult2injuryAinda não há avaliações
- Rotary Selection GuideDocumento16 páginasRotary Selection GuidecamenitaAinda não há avaliações
- Typical EPA M5 PM CalculationDocumento4 páginasTypical EPA M5 PM Calculationinsult2injuryAinda não há avaliações
- Symposium On Cooling Water IntakesDocumento477 páginasSymposium On Cooling Water Intakesinsult2injuryAinda não há avaliações
- Prefab Pumping StationsDocumento20 páginasPrefab Pumping Stationsinsult2injuryAinda não há avaliações
- Metalliferous Mining Cyclone Processing GuideDocumento10 páginasMetalliferous Mining Cyclone Processing Guidechris.mwabaAinda não há avaliações
- Filtering Hydrocyclone Performance AnalysisDocumento10 páginasFiltering Hydrocyclone Performance AnalysisBenjamin Cortés EAinda não há avaliações
- CFD SGDocumento46 páginasCFD SGinsult2injury100% (1)
- Cat Srca9000Documento52 páginasCat Srca9000insult2injuryAinda não há avaliações
- Darcy Friction Factor For Pipe Flow: Reynolds NumberDocumento1 páginaDarcy Friction Factor For Pipe Flow: Reynolds Numberinsult2injuryAinda não há avaliações
- Manual Valves - Flow CoefficientsDocumento4 páginasManual Valves - Flow Coefficientspal_stephenAinda não há avaliações
- A Complete Guideline For Blocked-In Condition PDFDocumento3 páginasA Complete Guideline For Blocked-In Condition PDFNacho.pAinda não há avaliações
- Guide To Interchangeable LubricantsDocumento12 páginasGuide To Interchangeable LubricantsJavier Jesus Gonzalez QuinaAinda não há avaliações
- A Look at Centrifugal Pump Suction Hydraulic - Part 1Documento4 páginasA Look at Centrifugal Pump Suction Hydraulic - Part 1Said Ahmed SalemAinda não há avaliações
- FMDS0504 0713 Transformers PDFDocumento41 páginasFMDS0504 0713 Transformers PDFOrlando E Cabrera RojasAinda não há avaliações
- Guideline - Pump - System - Designers (Pump Fundamentals) PDFDocumento16 páginasGuideline - Pump - System - Designers (Pump Fundamentals) PDFyyukin1100% (1)
- Materials Selection For Seawater PumpsDocumento8 páginasMaterials Selection For Seawater PumpsIjabiAinda não há avaliações
- 16.1.1 Furnaces For Roasting and Calcining: 16.1.1.1 Rotary KilnsDocumento66 páginas16.1.1 Furnaces For Roasting and Calcining: 16.1.1.1 Rotary Kilnshisem100% (1)
- Computer Simulated Plant Design For Waste Minimization & Pollution PreventionDocumento178 páginasComputer Simulated Plant Design For Waste Minimization & Pollution PreventionAndreea Florea100% (1)
- Stanley Stoves Introduction GuideDocumento13 páginasStanley Stoves Introduction GuidePatrick HealyAinda não há avaliações
- Fuels & Combustion - Emission & Control Technology TYBE COEP DR Vora 040422Documento93 páginasFuels & Combustion - Emission & Control Technology TYBE COEP DR Vora 040422Amey gaikwadAinda não há avaliações
- Fuel Processing Technology: Attila Kun-Balog, Krisztián SztankóDocumento8 páginasFuel Processing Technology: Attila Kun-Balog, Krisztián SztankóSubhadip DasAinda não há avaliações
- IJER Editorial: The Future of The Internal Combustion EngineDocumento8 páginasIJER Editorial: The Future of The Internal Combustion EngineAPAinda não há avaliações
- Ash Manipulation: Manipulate Burnt MatterDocumento2 páginasAsh Manipulation: Manipulate Burnt MatterSunčica NisamAinda não há avaliações
- MSDS Palm Tree DustDocumento13 páginasMSDS Palm Tree Dusterik mulyanaAinda não há avaliações
- Ram SeminarDocumento17 páginasRam SeminarAnonymous 1grFXHMGAinda não há avaliações
- Gap 17.12.1Documento15 páginasGap 17.12.1Cristobal A. Mir G.100% (1)
- Physical ChemistryDocumento5 páginasPhysical ChemistryLynnden CastilloAinda não há avaliações
- A MEMS-based Solid Propellant Microthruster With Au/Ti IgniterDocumento11 páginasA MEMS-based Solid Propellant Microthruster With Au/Ti IgniterTing Yui ChanAinda não há avaliações
- Singapore fire safety standard updateDocumento24 páginasSingapore fire safety standard updateKim SanAinda não há avaliações
- Eenrgy Audit and Management Books-GuideDocumento750 páginasEenrgy Audit and Management Books-GuideAnonymous LHGQqOB100% (1)
- 9A21505 Aerospace Propulsion-IDocumento4 páginas9A21505 Aerospace Propulsion-IsivabharathamurthyAinda não há avaliações
- Don't Gamble With Safety On Chemical TankersDocumento14 páginasDon't Gamble With Safety On Chemical TankersHenryAinda não há avaliações
- Methocel E5lv MSDSDocumento7 páginasMethocel E5lv MSDSOladimejiAinda não há avaliações
- Patio Furniture & Accessories Assembly Instructions Stop - ImportantDocumento17 páginasPatio Furniture & Accessories Assembly Instructions Stop - Importantoccupant12Ainda não há avaliações
- Internal Combustion EngineDocumento16 páginasInternal Combustion EngineHamzeh Al-QaisiAinda não há avaliações
- PC300 - 8Documento52 páginasPC300 - 8Mg Roy100% (3)
- PU85452T 2 Report EN45545 Bel+Power PDFDocumento10 páginasPU85452T 2 Report EN45545 Bel+Power PDFbekbek12Ainda não há avaliações
- GS (Global Studies 302) Summary Chapter 1-4 IFLDocumento36 páginasGS (Global Studies 302) Summary Chapter 1-4 IFLKay Chan Sotheara100% (2)
- GT Operation (Nanjin)Documento146 páginasGT Operation (Nanjin)Ady FardyAinda não há avaliações
- Homework #1 SolutionsDocumento13 páginasHomework #1 Solutionsvalerieweeaini100% (3)
- Piercing Rod Training: Understanding Hazards of PRB Coal DustDocumento33 páginasPiercing Rod Training: Understanding Hazards of PRB Coal DustDSG100% (1)
- Thermo Chemistry of Fuel-Air MixturesDocumento34 páginasThermo Chemistry of Fuel-Air Mixturesmahmudul adilAinda não há avaliações
- 0-2103471 Introduction To Engine DesignDocumento85 páginas0-2103471 Introduction To Engine DesignJulian DavidAinda não há avaliações