Escolar Documentos
Profissional Documentos
Cultura Documentos
Alkanes and Alkenes
Enviado por
Agus LugrahaDescrição original:
Direitos autorais
Formatos disponíveis
Compartilhar este documento
Compartilhar ou incorporar documento
Você considera este documento útil?
Este conteúdo é inapropriado?
Denunciar este documentoDireitos autorais:
Formatos disponíveis
Alkanes and Alkenes
Enviado por
Agus LugrahaDireitos autorais:
Formatos disponíveis
Alkanes and Alkenes
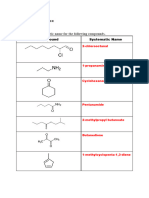
1. Complete the table below
Alkanes
Alkenes
1.
Similarities
2.
Differences
2. Complete the table below
Name
Ethane
Propene
Trichloromethane
2 Dimethyl Penthane
Hex-3-ene
1,4 Dicloro Hexane
2 Cloro 3 Floro Penthane
1.
Molecular structure
1.
2.
Reactivity
2.
3.
Type of reaction
3.
4.
With Aqueous Bromine
4.
Molecular Formula
Structural Formula
3. The substance X, Y and Z shown in the following reactions are alkanes
X + chlorine
uv light
CH3CH2Cl + HCl
Y + chlorine
uv light
CH3CH2Cl + HCl
CH3
Z + chlorine
uv light
CH3CHCH2CH3 + HCl
Cl
Identify and draw the full structural formulae of X, Y, and Z
_______________________________________________________________
_______________________________________________________________
_______________________________________________________________
4. Write the chemical equations for the following reactions
a. Combustion of propene
_______________________________________________________________
_______________________________________________________________
_______________________________________________________________
b. Reaction of propene with bromine
_______________________________________________________________
_______________________________________________________________
_______________________________________________________________
c. Reaction of butene with hydrogen
_______________________________________________________________
_______________________________________________________________
_______________________________________________________________
5. The hydrocarbon J is an alkane. The relative molecular mass of J is 72
a. What is the molecular formula of J
_______________________________________________________________
_______________________________________________________________
_______________________________________________________________
b. J reacts with chlorine in the presence of sunlight.
i. What type of reaction occurs when J reacts with chlorine
____________________________________________________________
ii. Write an equation for the reaction between J and chlorine
____________________________________________________________
iii. Describe what you will observe when J reacts with bromine vapour.
____________________________________________________________
6. Below is diagram showing cracking of an alkane
Octane, C8H18
Decane, C10H22
X
a. Name subtance X
_______________________________________________________________
b. Describe how alkenes are cracked
_______________________________________________________________
7. a. Name the hydrocarbon shown below
HH H
H-C-C=C-H
H
_______________________________________________________________
c. Is this molecule unsaturated? Explain your answer.
_______________________________________________________________
d. Write the chemical equation to represenr the complete combustion of this
hydrocarbon belongs
_______________________________________________________________
e. i. Name the homologous series to which this hydrocarbon belongs
_______________________________________________________________
ii. Draw full structure formula of the hydrocarbon with one more carbon atom in
the same homologous series.
8. The table below shows the properties of some alkenes
Alkene
Molecular Formula
Ethene
C2H4
.........
C3H6
Butene
.........
Pentene
C5H10
Hexene
C6H12
a. Complete the table above
Boiling Point ( oC)
-104.0
-47.7
-6.2
30.3
64.0
b. Describe how alkenes are manufactured
_______________________________________________________________
_______________________________________________________________
_______________________________________________________________
c. Draw a dot and cross diagram to show teh bonding of ethene
d. What is the value for x with alkene with molecular formula of CxH16
_______________________________________________________________
e. Identify the compound in the reaction
Ethene + X
Ethanol
i. Using full structural formula, write the equation for the reaction
_______________________________________________________________
ii. What name is given to this type of reaction?
_______________________________________________________________
9. The relative molecular mass of an unsaturated hydrocarbon Y is 54. It is found that
13.5 g of Y required 0.5 mol of chlorine for complete reaction.
i. How many carbon-carbon ( C=C ) are present in one molecule of Y?
___________________________________________________________________
___________________________________________________________________
___________________________________________________________________
ii. Suggest the structural formula of
_______________________________________________________________
10. Three gases, X, Y and Z are collected in the test tube.
Gas
Mr
X
2
Y
42
Z
58
Suggest the identities of X, Y and Z
Reaction with Bromine
No reaction
Decolourise bromine
No reaction
____________________________________________________________________
____________________________________________________________________
Você também pode gostar
- The Complete Organic Chemistry WorksheetDocumento8 páginasThe Complete Organic Chemistry WorksheetRishab Khandelwal100% (1)
- Schaum's Easy Outline of Organic Chemistry, Second EditionNo EverandSchaum's Easy Outline of Organic Chemistry, Second EditionNota: 3.5 de 5 estrelas3.5/5 (2)
- Organic Chem Practice Exam by Solomon CH 1-4Documento12 páginasOrganic Chem Practice Exam by Solomon CH 1-4Natasha Moo100% (1)
- Chem Igcse2Documento2 páginasChem Igcse2bonolomphaAinda não há avaliações
- Apch11 EstersDocumento2 páginasApch11 Estersloly62006Ainda não há avaliações
- Ujian 2 Form 5Documento9 páginasUjian 2 Form 5Nazreen NashruddinAinda não há avaliações
- Intro To Organic Exam Q No MechanismsDocumento14 páginasIntro To Organic Exam Q No Mechanismsarychan418Ainda não há avaliações
- Final-Module 5-The AlkynesDocumento5 páginasFinal-Module 5-The Alkynesjohncarlodc99Ainda não há avaliações
- Intro To Organic and Alkanes Revision QuestionsDocumento19 páginasIntro To Organic and Alkanes Revision Questionsarychan418Ainda não há avaliações
- 202-DCP Problem Set-6 With-Solutions RadicalsDocumento12 páginas202-DCP Problem Set-6 With-Solutions Radicalsmichelmanirakiza591Ainda não há avaliações
- Organic Chemistry: Section 1Documento8 páginasOrganic Chemistry: Section 1LiliAinda não há avaliações
- Fractional Distillation QDocumento9 páginasFractional Distillation Qarychan418Ainda não há avaliações
- Hydrocarbons. Section 21.1 Introduction To HydrocarbonsDocumento5 páginasHydrocarbons. Section 21.1 Introduction To HydrocarbonsAhmad asaAinda não há avaliações
- A Teaching - Learning Package in General Chemistry I: Tel - Nos: 487-8927/ 487-7670/ 09562796609Documento6 páginasA Teaching - Learning Package in General Chemistry I: Tel - Nos: 487-8927/ 487-7670/ 09562796609andy gamingAinda não há avaliações
- Chem 241 Final ExamDocumento4 páginasChem 241 Final ExamHerya EssaAinda não há avaliações
- Year 13 Practical Techniques and Data Analysis Past Paper PackDocumento51 páginasYear 13 Practical Techniques and Data Analysis Past Paper PackXx Jasmine XxAinda não há avaliações
- Igcse Chemistry Pasco ADocumento32 páginasIgcse Chemistry Pasco AGodfred WelbeckAinda não há avaliações
- More QuestionsDocumento86 páginasMore QuestionssuccesshustlerclubAinda não há avaliações
- Organic 1Documento6 páginasOrganic 1Jaspar GlagovsAinda não há avaliações
- OrganicNotes TolamateDocumento36 páginasOrganicNotes Tolamateraghava123456Ainda não há avaliações
- BiochemistryDocumento34 páginasBiochemistryVade ZenAinda não há avaliações
- Organic Chemistry Student Booklet 2Documento15 páginasOrganic Chemistry Student Booklet 2Mirjeta ZymeriAinda não há avaliações
- Practice PacketDocumento20 páginasPractice PacketTheresa Rebullos BileAinda não há avaliações
- CHEMISTRYWSF Organic ChemistryDocumento51 páginasCHEMISTRYWSF Organic ChemistrySneha JainAinda não há avaliações
- Practice Orgo ExamDocumento7 páginasPractice Orgo ExamDoris GrimaldiAinda não há avaliações
- Exp Carbon Compound Form 5Documento10 páginasExp Carbon Compound Form 5Zalilahismail IsmailAinda não há avaliações
- Carboxylic Acids and Derivatives QPDocumento14 páginasCarboxylic Acids and Derivatives QPRahbot Wolde-MichaelAinda não há avaliações
- Isomerism in Alkanes, Cycloalkanes, and Alkenes Using Molecular ModelsDocumento4 páginasIsomerism in Alkanes, Cycloalkanes, and Alkenes Using Molecular Modelsalbertvdatu278Ainda não há avaliações
- CalorimetryDocumento99 páginasCalorimetry/ “Nu” /Ainda não há avaliações
- 3.1.1 Naming Practice QuestionsDocumento33 páginas3.1.1 Naming Practice QuestionsAyaan RaufAinda não há avaliações
- Basic Organic Nomenclature Packet Honors Chemistry: Name: - BlockDocumento12 páginasBasic Organic Nomenclature Packet Honors Chemistry: Name: - BlockJamaica Calamno SalvadorAinda não há avaliações
- 3.3.9.2 Acylation (A-Level Only)Documento95 páginas3.3.9.2 Acylation (A-Level Only)jason smtihAinda não há avaliações
- Science Grade 9: Quarter 2 - Module 4-5 The Carbon CompoundsDocumento16 páginasScience Grade 9: Quarter 2 - Module 4-5 The Carbon CompoundsAaron Asne100% (1)
- Organic Chem Final ExamDocumento7 páginasOrganic Chem Final ExamAurora corpuzAinda não há avaliações
- STPM May Evaluation Chemistry 2Documento7 páginasSTPM May Evaluation Chemistry 2Ventus TanAinda não há avaliações
- Chem 4 Classwork 1 & Assignment 1Documento4 páginasChem 4 Classwork 1 & Assignment 1Lordsfavour AnukamAinda não há avaliações
- Organic Chem ExDocumento15 páginasOrganic Chem Exwan zhouAinda não há avaliações
- CHE 332 Final Review NameDocumento11 páginasCHE 332 Final Review Nametlyons1188Ainda não há avaliações
- Haloalkane Revision 3 QuestionsDocumento14 páginasHaloalkane Revision 3 Questionsarychan418Ainda não há avaliações
- CHM202 F12 HE1blankDocumento9 páginasCHM202 F12 HE1blankEric MclaughlinAinda não há avaliações
- 3.3.1 Introduction To Organic Chemistry ASDocumento20 páginas3.3.1 Introduction To Organic Chemistry ASOliver JonesAinda não há avaliações
- WS 11Documento4 páginasWS 11ratuadhwaAinda não há avaliações
- Halogenoalkanes Alcohols and Modern Analytical TechniquesDocumento17 páginasHalogenoalkanes Alcohols and Modern Analytical TechniquesDaniel KirovAinda não há avaliações
- Midterm ReviewDocumento15 páginasMidterm Reviewapi-295101311Ainda não há avaliações
- Intro To Organic POGILDocumento6 páginasIntro To Organic POGILzfmwj2db47Ainda não há avaliações
- WS READING Physical Chemical ChangeDocumento3 páginasWS READING Physical Chemical ChangeAnas TasyaAinda não há avaliações
- Carbon Compound 1Documento9 páginasCarbon Compound 1Kym LowAinda não há avaliações
- Physical Sciences p2 Last PushDocumento54 páginasPhysical Sciences p2 Last Pushmonkbons15Ainda não há avaliações
- Chem 0000Documento5 páginasChem 0000chikondikosamu24Ainda não há avaliações
- Sample Exam 1Documento3 páginasSample Exam 1Joseph CatiisAinda não há avaliações
- SCH4 Organic Unit TestDocumento6 páginasSCH4 Organic Unit TestMariiam CiiAinda não há avaliações
- Organic C CCCC CCCCDocumento88 páginasOrganic C CCCC CCCCKugan KishurAinda não há avaliações
- Carbon CompoundDocumento61 páginasCarbon CompoundhaslimiAinda não há avaliações
- Organic Chem Review With ANSWERSDocumento16 páginasOrganic Chem Review With ANSWERSRyan Christian PatriarcaAinda não há avaliações
- Las Balancing Chemical EquationsDocumento8 páginasLas Balancing Chemical EquationsPrince Dave NievesAinda não há avaliações
- Organic Diploma QuestionsDocumento16 páginasOrganic Diploma QuestionsRyan Christian PatriarcaAinda não há avaliações
- Practice Makes Perfect in Chemistry: Organic Chemistry with AnswersNo EverandPractice Makes Perfect in Chemistry: Organic Chemistry with AnswersAinda não há avaliações
- Practice Makes Perfect in Chemistry: Organic ChemistryNo EverandPractice Makes Perfect in Chemistry: Organic ChemistryNota: 3 de 5 estrelas3/5 (1)
- College Organic Chemistry Semester II: Practice Questions with Detailed ExplanationsNo EverandCollege Organic Chemistry Semester II: Practice Questions with Detailed ExplanationsAinda não há avaliações
- Molecular Devices: An Introduction to Technomimetics and its Biological ApplicationsNo EverandMolecular Devices: An Introduction to Technomimetics and its Biological ApplicationsAinda não há avaliações