Escolar Documentos
Profissional Documentos
Cultura Documentos
Diagrama FeC - Curvas de Resfriamento - 2013
Enviado por
ddouglas123Título original
Direitos autorais
Formatos disponíveis
Compartilhar este documento
Compartilhar ou incorporar documento
Você considera este documento útil?
Este conteúdo é inapropriado?
Denunciar este documentoDireitos autorais:
Formatos disponíveis
Diagrama FeC - Curvas de Resfriamento - 2013
Enviado por
ddouglas123Direitos autorais:
Formatos disponíveis
Diagrama Fe-C Curvas de Resfriamento
Desenvolvimento: Antonio Carlos Gomes Jr.
Ref.: Metals Handbook / www.key-to-steel.com
Pag. 1
FACENS
Diagrama Fe-Fe3C / TTT / CCTde Tratamento Trmico
Os aos podem apresentar uma grande variedade de propriedades dependendo da composio, bem como as fases e micro-constituintes presentes, que por sua vez dependem do tratamento trmico. A base para a compreenso do tratamento trmico dos aos o diagrama de fase Fe-C. Na verdade, inclui dois diagramas; o diagrama ferro grafite estvel e o diagrama Fe-Fe3C metaestvel. A condio estvel normalmente leva um tempo muito longo para desenvolver, especialmente na gama de baixa temperatura e baixo teor de carbono, portanto, o diagrama metaestvel de maior interesse FACENS
Pag. 2
Diagrama Fe-Fe3C / TTT / CCTde Princpios Tratamento Trmico de Tratamento Trmico
O ao geralmente definido como uma liga de ferro e carbono com o teor de carbono entre centsimo at cerca de 2% em peso. Outros elementos de liga pode chegar no total a cerca de 5% em peso em aos denominados de baixa liga e para os aos mais alta liga, tais como aos ferramenta, aos inoxidveis (> 10,5%) e resistentes ao calor aos CrNi (> 18%) a porcentagem elevada. Aos podem apresentar uma grande variedade de propriedades dependendo da composio, bem como as fases e microconstituintes presentes, que por sua vez dependem do tratamento trmico como j citado, refora-se este ponto.
FACENS
Pag. 3
Diagrama Fe-Fe3C / TTT / CCTde Tratamento Trmico
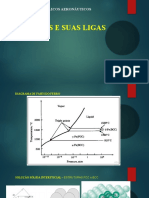
Diagrama de fase Fe-C A base para a compreenso do tratamento trmico dos aos o Fe-C diagrama de fase (Fig. 1 pg 05). A Figura 1 mostra realmente dois diagramas; o diagrama de ferro fundido-estvel (linhas tracejadas) e o metaestvel diagrama Fe-Fe3C. A condio estvel normalmente leva um tempo muito longo para se desenvolver, especialmente em baixa temperatura e baixo teor de carbono, sendo portanto, o diagrama metaestvel de maior interesse. O diagrama de Fe-C mostra que as fases so esperadas no equilbrio (ou de equilbrio metaestvel) para diferentes combinaes de concentrao de carbono e da temperatura.
Pag. 4
FACENS
Diagrama Fe-Fe3C / TTT / CCTde Tratamento Trmico
Pag. 5
FACENS
Diagrama Fe-Fe3C / TTT / CCTde Princpios Tratamento Trmico de Tratamento Trmico
No ao baixo carbono, a ferrita (-ferro) pode, no mximo, dissolver C 0,028%, em 727 C (1341 F) e a austenitaferro, o que pode dissolver 2,11 C% em peso a 1148 C (2098 F). No lado do diagrma que rico em carbono encontramos cementita (Fe3C). De menor interesse, exceto para os aos de alta liga, a existente -ferrita em temperaturas mais elevadas. Entre os campos monofsicos so encontrados regies com misturas de duas fases, tais como ferrita + cementita, austenita + cementita, e ferrita + austenita. Nas temperaturas mais elevadas, o campo fase lquida pode ser encontrado e abaixo deste so os dois domnios de fase lquida + austenita, lquido + cementita, e lquidos + -ferrita.
Pag. 6
FACENS
Diagrama Fe-Fe3C / TTT / CCTde Tratamento Trmico
Em tratamento trmico de aos, a fase lquida sempre evitada por motivos bvios. O teor de carbono em que a temperatura mnima de austenita atingido chamado ponto eutetide (0,77 C% em peso). A mistura da fase de ferrite-cementita desta composio formada durante o resfriamento tem uma aparncia caracterstica e chamado perlita e pode ser tratada como uma entidade microestrutural ou microconstituinte. um agregado de lamelas alternadas de ferrita e cementita.
Pag. 7
FACENS
O diagrama de Fe-C na Fig. 1 de origem experimental. O conhecimento dos princpios termodinmicos e dados termodinmicos modernos agora permitem que clculos muito precisos desse diagrama. Isto particularmente til quando os limites de fase deve ser extrapolados e a baixas temperaturas, onde o equilbrio experimental so extremamente lentos para se desenvolver. Se os elementos de liga so adicionados liga de ferrocarbono (ao), os limites de solubiliade e da composio eutetide so alteradas. Todos os elementos de liga importantes diminuem o ponto eutetide.Os elementos de liga adicionados tais como Mangans promovem uma estabilizao da Austenita e de maneira geral o Nquel, Tungstnio, Molibdnio e o Cromo aumentam a temperabilidade (a ser detalhado nas curvas CCT) Pag. 8 FACENS
Diagrama Fe-Fe3C / TTT / CCTde Tratamento Trmico
Diagramas de Transformao
Os aspectos de cintica de transformaes de fase so to importantes como os diagramas de equilbrio para o tratamento trmico dos aos. As fases metaestvel martensita e a bainita so microconstituintes morfologicamente metaestaestvies que so de extrema importncia para as propriedades dos aos, geralmente podem se formar com o resfriamento rpido at a temperatura ambiente.
Pag. 9
FACENS
Diagrama Fe-Fe3C / TTT / CCTde Tratamento Trmico
Bainita uma decomposio eutetide que uma mistura de ferrite e cementita. Martensita, ocorre com a austenita supersaturada e mais frgil e dura. Sua dureza aumenta com teor de carbono at cerca de 0,7% em peso. O resfriamento rpido onde conseguimos este produto ou fase denominado de tmpera e ser motivo de discusso detalhada em nosso curso de processo de fabricao.
Pag. 10
FACENS
Diagrama Fe-Fe3C / TTT / CCTde Tratamento Trmico
A transformao de uma estrutura em martensita ou otras fase, tais como a perlita e ou suas combinaes tambm de importncia no tratamento trmico de ao. Pode-se descrever convenientemente o que est acontecendo durante a transformao com diagramas de transformao. Quatro tipos diferentes de tais diagramas podem ser distinguidas: diagramas de transformao isotrmica que descrevem a formao da austenita (diagramas de ITH); contnuos de aquecimento de transformao (CRT); tempo-temperatura-transformao (TTT) contnuo resfriamento de transformao (CCT)
Pag. 11
FACENS
Diagrama Fe-Fe3C / TTT / CCTde Tratamento Trmico
Destacamos inicialmente os TTT e CCT devido uma questo prtica quando falamos de resfriamento e obteno de suas estruturas e/ou fases. A determinao dos mesmos pode ser feita atravs de dilatometria e mais comum (e mais acurado) por anlise microscpica
O desenvolvimento da microestrutura com o tempo pode ser obtido processando seguidamente, tratanado espcimes pequenos em um banho de chumbo um de cada vez aps aumentar tempos de reteno e medindo a quantidade de fases formadas na microestrutura com o auxlio de um microscpio. Pag. 12 FACENS
Diagrama Fe-Fe3C / TTT / CCTde Tratamento Trmico
Diagramas CCT importante para indicar claramente o tipo de curva de resfriamento (derivado do diagrama de transformao). A utilizao de uma taxa de resfriamento constante muito comum na prtica experimental. No entanto, este regime apesar de fcil visualizao, raramente ocorre em uma situao prtica. Para uma aplicao prtica, aliam-se estas curvas CCT com uma anlise bem mais prxima realidade com um ensaio denominado Jominy. H neste caso uma simulao do comportamento no interior de uma pea, tais como a velocidade de resfriamento a uma certa distncia da extremidade temperada.
Pag. 13
FACENS
Diagrama Fe-Fe3C / TTT / CCTde Tratamento Trmico
Cada diagrama CCT contm uma famlia de curvas que representam as taxas de arrefecimento a diferentes profundidades de um cilindro com 300 mm (12 polegadas) de dimetro. A taxa mais lenta de resfriamento representa o centro do cilindro. Quanto mais intensa a troca de calor do meio as curvas em forma de C so deslocados.
Pag. 14
FACENS
D I A G R A M A S T T T E
C C T Pag. 15 d
FACENS
Princpios de Tratamento Trmico
Pag. 16
FACENS
BACK UP TTT/CCT ORIGINAL INGLSde Tratamento Trmico
When carbon steel is quenched in the baths at constant temperatures, the velocity of austenite transformation is found to depend on temperature. The time for the beginning and completion of the transformation of the austenite is plotted against the temperature to give the Bain "S-curve", shown in Fig. 1, now called TTT-curve (time-temperaturetransformation).
Pag. 17
FACENS
Abstract: Princpios de Tratamento Trmico The structures formed during the continuous cooling of steel from above Ac3 can be understood best by studying the constant-temperature (isothermal) transformation of austenite, thus separating the two variables: time and temperature.One method consists of heating small specimens above Ac3 to form austenite, then quenching into a suitable bath (e.g. liquid tin) at some constant subcritical temperature. After holding for selected periods of time, the specimens are withdrawn from the bath and rapidly quenched in cold water. This converts any untransformed austenite into martensite the volume of which can be estimated microscopically. Another method consists in measuring length changes caused by the decomposition of austenite at the constant temperature by means of a dilatometer. Pag. 18 FACENS
Princpios de Tratamento Trmico
The logarithmic scale of time is used to condense results into a small space. Ae1 and Ae3 lines represent the equilibrium transformation temperatures. Austenite is completely stable above Ae3 and partially unstable between Ae3 and Ae1. Below Ae1 austenite is completely unstable and transforms in time. Two regions of rapid transformation occur about 550 and 250C. The form of each of the curves and their positions with respect to the time axis depend on the composition and grain size of the austenite which is transforming.
Pag. 19
FACENS
The TTT-curve is most useful in presenting an overall picture of the transformation behaviour of austenite. This enables the metallurgist to interpret the response of steel to any specified heat-treatment, to plan practical heat-treatment operations and to control limited hardening or softening and the time of soaking. The decomposition of austenite occurs according to three separate but sometimes overlapping mechanisms and results in three different reaction products: pearlitic, bainitic, martensitic.
Princpios de Tratamento Trmico
Pag. 20
FACENS
Princpios de Tratamento Trmico
Pearlitic The upper dotted curve in Fig. 1 represents the beginning of the formation of ferrite. The curve just below it indicates the beginnings of the breakdown of the austenite remnant into a ferrite-carbide aggregate. In the high-temperature pearlitic range in Fig. 1 the process resembles the solidification of crystals from a liquid by the formation and growth of nuclei of carbide followed by ferrite by side nucleation with side and edge growth, Fig. 2a and b.
Pag. 21
FACENS
Princpios de Tratamento Trmico
At 700C the formation of nuclei is slow (i.e. incubation period), then growth proceeds rapidly to form large pearlite colonies covering several austenite grains in some cases. As the transformation temperature is lowered to 500C the incubation period decreases and the pearlite becomes increasingly fine. Large numbers of nuclei form in the austenite boundaries, but growth is slower and this produces nodular troostite, Fig. 2a. In the case of medium carbon steels the excess ferrite decreases in volume and begins to show an acicular or Widmansttten type of distribution. The relative amounts of free ferrite to be expected after a given heat-treatment is indicated by the size of the "austenite and ferrite" field and by the temperature interval between Ae1 and Ae3.
Pag. 22
FACENS
Bainitic Princpios de 350 Tratamento Trmico Between about 500 and C initial nuclei are ferrite which is coherent with the austenite matrix. Cementite then precipitates from the carbon-enriched layer of austenite, allowing further growth of the ferrite as shown in Fig. 2c. The carbides tend to lie parallel to the long axis of the bainite needle to form the typical open feathery structure of upper bainite. Below 350C coherent ferrite, supersaturated with carbon, forms first and is then followed by the precipitation of carbide within the ferrite needle, transversely at an angle of 55. A proportion of the carbide is Fe2,4C and the ferrite contains a little dissolved carbon. This lower bainite structure is somewhat similar to lightly tempered martensite (Fig. 2d). Pag. 23 FACENS
Martensitic In quenching down to about 250C, the temperature Princpios de Tratamento Trmico drops rapidly through the interval in which "nucleation" could take place, to a temperature so low that the molecular mobility, i.e. diffusion, becomes too small for the formation of nuclei. In the third stage, therefore, the austenite changes incompletely into a distorted body-centred structure, with little or no diffusion of the carbon into particles of cementite, to form martensite the plates of which are formed at a high speed (less than 0,002 sec). This suggests that the mechanism of formation of this structure is not nucleation and growth but a shearing process. This resembles the process of mechanical twinning and involves very little atomic movement, but considerable internal stress due to the shear and to the position of the carbon atoms. Pag. 24 FACENS
Princpios de Tratamento Trmico As the temperature decreases the elastic energy increases and eventually causes a shear in a part of the matrix, which stabilises the rest. Further shear can only occur when the temperature is lowered and more energy gained. The amount of martensite formed, therefore, is practically independent of time and depends principally on the temperatures at which the steel is held. Hence a proportion of austenite is usually retained in quenched steel which can be reduced in amount by a decrease in temperature. This fact is used in sub-zero quenching.
Pag. 25
FACENS
The temperature at which martensite begins to form (Ms) is progressively lowered as the carbon content of the steel increases, e.g.
Princpios de Tratamento Trmico
0,2 490 0,4 420 0,8 250 1,2 150
C% Ms C
0,02 520
The temperature is also affected by the alloy content, but the following empirical formula (Steven and Haynes) can be used for calculating Ms from the chemical analyses, provided all carbides have been dissolved in the austenite: Ms in C = 561 - 474 (% C) - 33 (% Mn) - 17 (% Ni) - 17(% Cr) - 21 (% Mo). Mf is about 215C below the Ms. Plastic and elastic stresses promote the formation of martensite, but it is retarded when cooling is interrupted. When cooling is resumed after such a stabilisation arrest martensite only begins to form again after cooling to a lower temperature. The rate and extent of stabilisation (depression) depend on the temperature and time of holding, amount of prior transformation and alloy content.
Two forms of martensite have been identified depending on carbon content. In low carbon steels laths containing many dislocations are found, while in high carbon steels the plates are heavily twinned, Fig. 3(a) and (b).
Pag. 26
FACENS
Você também pode gostar
- Parte elétrica e dispositivos elétricos da NR 12: conceitos básicos e fundamentais para a compreensão da normaNo EverandParte elétrica e dispositivos elétricos da NR 12: conceitos básicos e fundamentais para a compreensão da normaNota: 5 de 5 estrelas5/5 (3)
- Apostila Tratamentos TérmicosDocumento273 páginasApostila Tratamentos TérmicosBárbara DiasAinda não há avaliações
- 07 Processo de Têmpera e RevenimentoDocumento6 páginas07 Processo de Têmpera e RevenimentoEmerson Castro100% (1)
- FundiçãoDocumento20 páginasFundiçãoeliseuhenrique100% (1)
- #00 - MCM - 300 (Metal - SENAI) PDFDocumento213 páginas#00 - MCM - 300 (Metal - SENAI) PDFProfessor Reginaldo GomesAinda não há avaliações
- ZincoDocumento8 páginasZincodanielAinda não há avaliações
- Procedimento ReparoDocumento4 páginasProcedimento ReparoThais SchwinderAinda não há avaliações
- Diagrama TTT Cap 3 PDFDocumento8 páginasDiagrama TTT Cap 3 PDFEsthevenAinda não há avaliações
- Aula 02Documento39 páginasAula 02Vitor CavalcantiAinda não há avaliações
- Cap 10 - Transformaçoes de Fases em MetaisDocumento71 páginasCap 10 - Transformaçoes de Fases em MetaisEltonAinda não há avaliações
- Aula 10-Diagrama TTTDocumento47 páginasAula 10-Diagrama TTTGuilherme VieiraAinda não há avaliações
- Diagrama de Equilibrio Fe-CDocumento19 páginasDiagrama de Equilibrio Fe-Ctcosta88Ainda não há avaliações
- Introdução Ao Estudo Dos Aços-Parte 3Documento21 páginasIntrodução Ao Estudo Dos Aços-Parte 3kffgkgigl24Ainda não há avaliações
- Aula 9-Tempera e Revenido - Curvas TTT - IT - CCT ReduzidoDocumento43 páginasAula 9-Tempera e Revenido - Curvas TTT - IT - CCT ReduzidoJoão Asmar JuniorAinda não há avaliações
- Aula 4 - Diagrama TTTDocumento82 páginasAula 4 - Diagrama TTTLorena FerreiraAinda não há avaliações
- Têmpera e Revenido e MartêmperaDocumento4 páginasTêmpera e Revenido e MartêmperaIzak SilvaAinda não há avaliações
- Aços - Tratamentos TérmicosDocumento9 páginasAços - Tratamentos Térmicossergioeq2000Ainda não há avaliações
- Processos de Fabricação I Aula 3 Diagrama de TransformaçõesDocumento35 páginasProcessos de Fabricação I Aula 3 Diagrama de TransformaçõesJose Roberto dos SantosAinda não há avaliações
- Apresentação - TermodinamicaDocumento21 páginasApresentação - TermodinamicaThiago MoreiraAinda não há avaliações
- Tratamento Térmico e Ligas Metálicas - PetrobrasDocumento12 páginasTratamento Térmico e Ligas Metálicas - PetrobrasIgor Jefferson Cabral AraújoAinda não há avaliações
- Estudo Das Curvas TTTDocumento6 páginasEstudo Das Curvas TTTlucianafigueiro100% (1)
- Trabalho de CaeDocumento3 páginasTrabalho de CaeEdgarx SilvaAinda não há avaliações
- Curvas TTT AulaDocumento36 páginasCurvas TTT AulaAmanda Victória Luz da RosaAinda não há avaliações
- Curvas TTTDocumento11 páginasCurvas TTTChico GondimAinda não há avaliações
- Aula 3 - Diagrama de Equilibrio Fe-CDocumento64 páginasAula 3 - Diagrama de Equilibrio Fe-CLorena FerreiraAinda não há avaliações
- Recozimento Ligas FerrosasDocumento10 páginasRecozimento Ligas FerrosasHeberAinda não há avaliações
- MinicursofundicaoDocumento71 páginasMinicursofundicaoCesar Alencar MendesAinda não há avaliações
- 13 Diagrama Ferro-CarbonoDocumento2 páginas13 Diagrama Ferro-CarbonoAristides Alves Reis100% (1)
- Estudo Das Curvas TTT PDFDocumento6 páginasEstudo Das Curvas TTT PDFEsthevenAinda não há avaliações
- Diagramas TTT e TACDocumento3 páginasDiagramas TTT e TACJotaAinda não há avaliações
- curvaTTTDocumento39 páginascurvaTTTFelipe Torres BarrosAinda não há avaliações
- Diagramas Tratamentos Térmicos e TTT PDFDocumento39 páginasDiagramas Tratamentos Térmicos e TTT PDFMasd GorouseiAinda não há avaliações
- Metalurgia Física Dos AçosDocumento11 páginasMetalurgia Física Dos AçosHugo Carvalho AmorimAinda não há avaliações
- Acendino. Diagrama de Fases Fe-CDocumento10 páginasAcendino. Diagrama de Fases Fe-CAcendino Notholias de SousaAinda não há avaliações
- 1687062155120+introdução TeoricaDocumento5 páginas1687062155120+introdução TeoricaLuan MoraesAinda não há avaliações
- TratamentDocumento197 páginasTratamentLidiane MunizAinda não há avaliações
- Aula 9 Tratamento TermicoDocumento35 páginasAula 9 Tratamento TermicoRodrigo Del Guerso SoaresAinda não há avaliações
- Mudanças Microestruturais em Ligas Fe-CDocumento40 páginasMudanças Microestruturais em Ligas Fe-CBrunoRamosAinda não há avaliações
- Engenharia Dos Materiais Metálicos - Parte 2Documento175 páginasEngenharia Dos Materiais Metálicos - Parte 2Nick AaronAinda não há avaliações
- Aula 11 Transformacoes de Fases em MetaisDocumento32 páginasAula 11 Transformacoes de Fases em MetaisSullivam PrestesAinda não há avaliações
- Diagrana Fe CDocumento4 páginasDiagrana Fe CRaquel MarraAinda não há avaliações
- Exercicios de CienciasDocumento13 páginasExercicios de CienciasMarilis MaguengueAinda não há avaliações
- Lista 2 de PCMDocumento6 páginasLista 2 de PCMLeonardo MedeirosAinda não há avaliações
- Diagrama de FasesDocumento20 páginasDiagrama de FasesDenilsonRobertoAinda não há avaliações
- Diagrama Ferro CarbonoDocumento3 páginasDiagrama Ferro CarbonoCamilla Souza MenezesAinda não há avaliações
- Diagramas de Transformação Por Resfriamento ContínuoDocumento1 páginaDiagramas de Transformação Por Resfriamento ContínuoJessica CardosoAinda não há avaliações
- Aula 1 Curva TTTDocumento19 páginasAula 1 Curva TTTRenan GuerraAinda não há avaliações
- AustêmperaDocumento3 páginasAustêmperaLucas ChiminAinda não há avaliações
- MCM II - RevisãoDocumento19 páginasMCM II - RevisãoEdimilson RodriguesAinda não há avaliações
- 20945-Apostila Introdução Tratamentos TérmicosDocumento23 páginas20945-Apostila Introdução Tratamentos TérmicosJames DewarAinda não há avaliações
- AulasDocumento9 páginasAulascarpolotesAinda não há avaliações
- 5-Metalurgia EFIDocumento43 páginas5-Metalurgia EFIdfinspecaoAinda não há avaliações
- THERMOCALDocumento14 páginasTHERMOCALsdfvdsfAinda não há avaliações
- Metalurgia Da Soldagem ProminpDocumento19 páginasMetalurgia Da Soldagem ProminpmaiconAinda não há avaliações
- Modulo 6 PreparadoDocumento34 páginasModulo 6 PreparadoGilberto BadeAinda não há avaliações
- 1 - Diagramas TTTDocumento35 páginas1 - Diagramas TTTVitoria MarianaAinda não há avaliações
- Especulações Da Prova de MCM 22Documento4 páginasEspeculações Da Prova de MCM 22Guilherme MonteiroAinda não há avaliações
- 2 - Aços 1Documento53 páginas2 - Aços 1Canal News/NovidadesAinda não há avaliações
- Análise das tensões em estruturas de concreto devido a variações de temperatura entre os anos de 2009 e 2018No EverandAnálise das tensões em estruturas de concreto devido a variações de temperatura entre os anos de 2009 e 2018Ainda não há avaliações
- Vestibular2012 Grupo2 Prova Dia0611Documento15 páginasVestibular2012 Grupo2 Prova Dia0611QUIMICAMILAAinda não há avaliações
- Lopas Catalogo 2022Documento108 páginasLopas Catalogo 2022Thiago SilvaAinda não há avaliações
- 6-Alto Forno EspecializacaoDocumento40 páginas6-Alto Forno EspecializacaoGeraldo Magela PereiraAinda não há avaliações
- Homologacão Das Normas ABNT PDFDocumento8 páginasHomologacão Das Normas ABNT PDFJuliana MedeirosAinda não há avaliações
- Catálogo Ligas Especiais - Specialty AlloysDocumento9 páginasCatálogo Ligas Especiais - Specialty AlloysVinicius Martins PedrosaAinda não há avaliações
- Corrosão em Metais em Alcool PDFDocumento117 páginasCorrosão em Metais em Alcool PDFMarceloPauloAinda não há avaliações
- Catalogo-Digital-Dia Das Maes-Silvia EstimaDocumento26 páginasCatalogo-Digital-Dia Das Maes-Silvia EstimaCris FragaAinda não há avaliações
- Tratamentos Térmicos Aço 5160 Revisão 05-07-11Documento7 páginasTratamentos Térmicos Aço 5160 Revisão 05-07-11lolzera100% (3)
- 05 - Acos InoxDocumento8 páginas05 - Acos InoxAllan DiasAinda não há avaliações
- Principais Ligas, Formatos, Características e Aplicações Do Bronze - ShockmetaisDocumento2 páginasPrincipais Ligas, Formatos, Características e Aplicações Do Bronze - ShockmetaisVITOR RENAN SILDA DA CONCEIÇÃOAinda não há avaliações
- Exercicios de Concurso Diagramas (COM Gabarito)Documento8 páginasExercicios de Concurso Diagramas (COM Gabarito)Jailson Nóbrega100% (2)
- Metalurgia Do PóDocumento15 páginasMetalurgia Do PóamandabraunAinda não há avaliações
- Condutores Eletricos de Cobre e Aluminio para Instalaçoes Industriais Ate 1kV PDFDocumento6 páginasCondutores Eletricos de Cobre e Aluminio para Instalaçoes Industriais Ate 1kV PDFvaldirmiranda83Ainda não há avaliações
- Processo Produtivo METALOMECÂNICADocumento15 páginasProcesso Produtivo METALOMECÂNICAHenrique BarrosAinda não há avaliações
- Fundição em CoquilhaDocumento35 páginasFundição em CoquilhaVitor CavalcantiAinda não há avaliações
- Folder Condutores de AluminioDocumento4 páginasFolder Condutores de AluminioLu CoeAinda não há avaliações
- Algo NovoDocumento45 páginasAlgo NovoSajane MassadaAinda não há avaliações
- Catálogo de Tubos - Aço InoxDocumento3 páginasCatálogo de Tubos - Aço InoxGileno DiasAinda não há avaliações
- AWR14Documento4 páginasAWR14cristinainoueAinda não há avaliações
- Fabricação Do MG Fundido e Suas LigasDocumento42 páginasFabricação Do MG Fundido e Suas LigasMatheus LopesAinda não há avaliações
- 2-Lista de Exercícios Estrutura de Solidos CristalinosDocumento6 páginas2-Lista de Exercícios Estrutura de Solidos CristalinosDaniboyAinda não há avaliações
- ACIARIA - DessulfuraçãoDocumento59 páginasACIARIA - DessulfuraçãoMarduarte8383% (6)
- Ferneto-Manual Dough Divider DMFDocumento2 páginasFerneto-Manual Dough Divider DMFMohammed Abu HawashAinda não há avaliações
- EStanhoDocumento36 páginasEStanhoCelso SousaAinda não há avaliações
- ManualPratico FCCDocumento12 páginasManualPratico FCCRaquel Beatriz SilvaAinda não há avaliações
- Brinco de Zirconia Masculino - Pesquisa GoogleDocumento1 páginaBrinco de Zirconia Masculino - Pesquisa GoogleThalyson BenoneAinda não há avaliações